1,4-Phenylenediamine: A Commentary on Its Journey, Science, and Future
Historical Development
The story of 1,4-phenylenediamine isn’t a headline grabber, yet its history quietly weaves through the world of applied chemistry. Chemists first drew up ideas around benzene derivatives back in the 19th century, eager to explore aromatic amines and their ragged possibilities. In those early days, the realm of synthetic dyes opened doors, and the likes of 1,4-phenylenediamine helped launch a revolution in textile coloring. Over time, research didn’t just stop at dyes. This compound threaded itself into pharmaceuticals, polymers, and even hair dyes. The subtle shift from basic research to large-scale industry teaches a lesson – meaningful science doesn’t always announce itself loudly, but it reshapes industries behind the scenes.
Product Overview
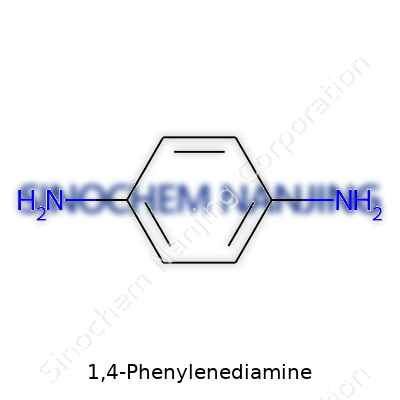
From a practical standpoint, 1,4-phenylenediamine stands as an aromatic amine with a benzene ring supporting two amino groups exactly opposite each other. Most readers recognize it as a white or pale solid, unless it darkens from oxidation. The material may look unremarkable, but its chemistry packs real punch. For those outside the lab, it feels strange to think such a substance helps color hair, improves rubber, or forms the backbone of aramid fibers used in bullet-resistant vests. When a product finds its way into so many hands and industries, it raises questions about oversight, safety, and innovation all at once.
Physical & Chemical Properties
Anyone who’s handled 1,4-phenylenediamine can recall its sharp aroma and tendency to undergo color change once exposed to air. At room temperature, it sits as a crystalline solid. It melts at a temperature above 140 degrees Celsius, holding up under heat until pushed further. Solubility lands on the moderate side in water, but rises in acid, which many operators find handy in formulation work. The key interest, though, centers on its reactivity: those amino groups invite coupling, oxidation, and substitution reactions, acting like a chemist’s playground. Not every aromatic amine behaves as energetically or as usefully, making this molecule stand out.
Technical Specifications & Labeling
Labels, purity claims, and technical sheets do their job, but they’re only the beginning. Companies dealing with 1,4-phenylenediamine keep strict controls to avoid hazardous contamination, often aiming for purity above 98%. Material safety data makes it clear—this isn’t a substance for loose handling. Labels often use names like PPD or para-phenylenediamine, alerting users to potential risks. Legislation keeps a close watch, especially in consumer goods such as hair dyes. People deserve to know what's inside the products they buy, and clear labeling marks the line between informed choice and unwanted surprise. Mistakes or oversights here bring more than regulatory slapdowns—they can turn public perception, rightly, against any company cutting corners.
Preparation Method
On the industrial scale, manufacturers usually rely on reduction routes from nitrobenzene or its dinitro analogs to produce 1,4-phenylenediamine. This process isn’t new, but it’s refined over years for yield and safety. Some routes involve catalytic hydrogenation, others use iron and acid, all aiming for efficiency and clean output. These choices reflect more than just economics—they tell you about priorities. High yield is important, but so is minimizing hazardous by-products. Waste management and emission controls stay as top concerns for anyone serious about chemical ethics.
Chemical Reactions & Modifications
Its core attraction lies in its reactivity. 1,4-phenylenediamine serves as a prime candidate for coupling reactions in making azo dyes. Those who spent time in a lab recall how quickly color changes signal reaction progress—brown, green, or blue hues marking success or trouble. Oxidative polymerization transforms it into conducting polymers, while condensation with phosgene crafts the backbone of aramid fibers. Hundreds of patented tweaks exist for its modification, but all draw on the same underlying versatility. Its chemistry keeps the material in play, letting it evolve with market needs. If a molecule can land in both fashion and warfare, it deserves attention.
Synonyms & Product Names
Names have power in chemistry, and this one branches out. ‘PPD’ in technical industries, ‘para-phenylenediamine’ among regulatory bodies, and local names in regional contexts make it hard for the casual observer to connect dots. In retail goods, especially hair dyes, labels may not even include the full chemical title—opting for coded synonyms or translated terms. Folks using these products may not notice the link to more technical domains or to occupational safety data. Building a common understanding starts by recognizing these names as more than jargon—they stand for a hidden web of risks and uses.
Safety & Operational Standards
There’s no sugarcoating the health issues tied to 1,4-phenylenediamine. Professionals know to treat it with respect, isolating it in ventilated spaces, gloves on, eye protection up, and spills cleaned right away. Allergic reactions hit some users hard, especially in hair products, with skin sensitivity even at low exposures. Regulatory bodies, including the European Chemicals Agency and the US EPA, keep it under tight focus. Guidelines lock in exposure limits in workplaces, labeling on finished goods, and prescribed testing for new uses. I’ve seen field technicians take short cuts, only to regret rashes or compromised health. Respect for operational standards isn’t a formality. It’s the invisible net that protects lives.
Application Area
The reach of 1,4-phenylenediamine stretches wider than most suspect. In the cosmetics aisle, it shows up in dark hair dye formulations, where nothing else creates color with the same depth. The textile world relies on it for synthetic fiber dyeing, running through ton after ton in busy plants. It shapes the structure of aramid fibers, like Kevlar, feeding into high-profile protective equipment. If one follows its journey further, you’ll see it pop up in rubber chemicals—enhancing strength in tires and elastic goods. Each application, from industrial textiles to something as personal as a hair color kit, raises hard questions about balance—utility versus risk, innovation versus oversight.
Research & Development
Ongoing research circles around safer alternatives and improved handling procedures. Scientists hunt for less allergenic substitutes for hair coloring and safer process catalysts for large-scale manufacturing. The search doesn’t just prioritize compliance; it aims to shrink both health risks and the environmental footprint. Teams experiment with greener synthesis methods, employing bio-based reducing agents or recyclable solvents—ambitious, yes, but necessary. R&D isn’t a box to check; it’s the line between stagnation and progress. Results in this field could spill over into countless industries, tipping the balance toward sustainability and public health without stripping away the advantages of the original molecule.
Toxicity Research
Toxicological studies have cast a long shadow over 1,4-phenylenediamine. It earned its infamy through reports of sensitization, skin irritation, and even mutagenic effects in lab settings. Epidemiological evidence ties occupational exposure to health outcomes, prompting restrictions, especially in Europe. Animal tests reinforce the need for strict thresholds—no free pass for careless operators. I remember one case from a manufacturing site, where an accidental exposure put a worker in the hospital, changing company policies overnight. These reminders ground research in the reality that industrial chemistry always walks a line between utility and harm. Regulators, companies, and consumers all need transparency and vigilance, not just in monitoring, but in honest communication of risks.
Future Prospects
The future of 1,4-phenylenediamine hinges on innovation, regulation, and public perception. Its chemistry guarantees continued presence in niche applications, especially where performance matters more than cosmetic preference. Researchers working on green chemistry and product safety hold the key to its next phase—whether through safer derivatives, closed-loop processes, or better exposure management. Industries leaning on its unique traits must keep evolving too, investing in safer technologies and transparent labels. As more consumers voice concerns over ingredient safety, product design can’t afford to ignore the lessons from past mistakes. A molecule with such reach can’t outpace its own shadow, but it can inspire a smarter, safer approach for the next wave of scientific advancement.
What is 1,4-Phenylenediamine used for?
A Key Ingredient In Everyday Products
1,4-Phenylenediamine stands out in the world of chemicals because people use products containing it almost every day, without giving it much thought. Think about dyeing your hair or changing the color of your clothes with fabric dyes. This ingredient gives vibrant, lasting color to hair dyes and textile pigments. Its effects reach far beyond just cosmetic appeal—it changes how goods look, making them more appealing to consumers and helping industries meet fast fashion trends.
The Backbone Of Hair Dye Formulas
Most permanent and semi-permanent hair dyes feature this chemical. It helps activate color through a process called oxidation. Once applied and mixed with other substances, it reacts to form shades ranging from black to brown, and even some grays. Stylists look for it in formulas because its color stays true, even after many washes. Having dyed my own hair, I remember a stylist warning about possible allergies—1,4-Phenylenediamine can cause skin reactions in some people. Those dangers affect not just customers, but also salon professionals who handle dyes daily. That’s why it’s always smart to read labels and perform patch tests, even for products sold at reputable stores.
Supporting The Textile Industry
As fashion cycles speed up, textile manufacturers lean on this ingredient to create resistant colors that last in sunlight and after several washes. I’ve worked with suppliers who chose certain dyes because customers complained about faded shirts and pants. 1,4-Phenylenediamine steps in here, keeping busy factories running and brands happy. Clothes dyed with it often hit shelves during fashion seasons, driven by the need for both bright and dark fabric tones. The problem? Factories that use it may risk worker exposure, so companies must focus on strong safety practices and keep protective equipment on hand.
Industrial And Scientific Uses
Beyond looks and fashion, this chemical has a role in factories making rubber and plastics. Engineers add it as a chemical intermediate, which means it helps synthesize other materials. It can tighten up certain chemical bonds in rubber goods, leading to tires and hoses that won’t crack under stress. Chemistry labs use it to test for certain metals, making 1,4-Phenylenediamine a staple for both research and manufacturing. From a science standpoint, I remember long days in the lab, watching solutions shift to deep purples thanks to that reaction. Its staying power in research circles explains why so many technical fields depend on it.
Balancing Benefits With Health Risks
There’s no denying 1,4-Phenylenediamine performs essential roles, but safety can’t be pushed aside. Medical reports link it to skin allergies and, with repeated exposure, possible respiratory issues. Regulatory agencies across Europe, the US, and Asia keep watch over its use, setting limits for consumer products and workplaces. After reading several case studies on occupational exposure, it’s clear employers need to offer training, gloves, and ventilation—skipping these steps risks more than productivity. As an individual who cares about safe work environments, I see room for more oversight and better consumer education.
Charting A Safer Path Forward
While alternatives do exist, few match the impact and versatility of 1,4-Phenylenediamine. Still, companies seek out newer formulations that aim to lower allergy rates and reduce environmental impact. For now, shoppers and workers stay safest when they ask questions, check product safety data sheets, and keep searching for brands committed to transparency. My experience tells me that safer manufacturing and better labeling go a long way in protecting both consumers and workers, so pushing for stronger standards often spells out real change.
Is 1,4-Phenylenediamine toxic or hazardous?
What is 1,4-Phenylenediamine?
1,4-Phenylenediamine pops up in a number of products that most people would never expect. This chemical plays a big role in some black hair dyes, rubber products, and even certain inks and polymers. Lots of households might not know it, but it sees daily use in manufacturing around the world and makes its way into the lives of many people through these routes.
Risks to Human Health
I remember working in a place that produced specialty inks, and there was always a certain caution around certain chemicals. 1,4-Phenylenediamine is one that standard safety training hones in on. Skin exposure often leads to allergic reactions for sensitive people. Redness, itching, swelling — these are more than a rare complaint. Longer exposures, especially in under-ventilated spaces, often bring respiratory irritation. Does it cause cancer? Current science doesn't pin it as a confirmed human carcinogen, but some tests in animals spark enough concern to make anyone think twice about careless handling.
The European Chemicals Agency lists it under substances of very high concern due to its suspected mutagenic effects. Companies in the European Union must label products containing this chemical clearly — that’s no small regulatory hurdle. The U.S. National Institute for Occupational Safety and Health also classifies it as a “potential occupational carcinogen.”
Environmental Impact
Disposal is another issue. At a previous job, routine checks on our drainage and waste streams revolved largely around compounds like this. 1,4-Phenylenediamine doesn't just disappear when it washes out with wastewater. It has a knack for sticking around in the environment and can harm aquatic life if it leaves the plant unchecked. Fish and other water creatures react badly to it, which means responsible management stretches well beyond the factory gate.
Why Does This Matter to Regular Consumers?
Most folks don’t handle industrial-sized buckets of this ingredient. Even so, black hair dyes and coloring kits rely on 1,4-Phenylenediamine for depth and staying power. People rarely realize a patch test on the skin before application isn’t just about allergies — it’s about avoiding a reaction that can last weeks. Hairdressers face repeat exposure: gloves, masks, and ventilation make a difference. Users should pay attention to ingredient labels on cosmetics and cleaners.
Safer Handling and Alternatives
Better ventilation, good gloves, and strict spill clean-up rules make a real difference in workplaces. I’ve seen coworkers take shortcuts, only to wind up with dermatitis and regret. Companies that keep rigorous safety policies see lower long-term health costs. On the consumer side, natural or less reactive hair colorants exist, but results might not feel quite the same. Still, the peace of mind can outweigh a marginal trade-off in product performance.
Regulators keep pushing companies toward safer alternatives. Technology moves slower than public health pressure sometimes, but change does happen. Reading safety data sheets, following labeling, and leaning on up-to-date workplace protections all contribute in ways that statistics alone can’t fully capture. People have a right to know what their products contain and how to protect themselves — and the industry holds a clear responsibility not to skirt those truths.
What safety precautions should be taken when handling 1,4-Phenylenediamine?
Understanding What We’re Dealing With
1,4-Phenylenediamine shows up most often in hair dyes and some industrial chemicals. I remember working in a lab where this stuff was common, yet a lot of folks treated it like simple food coloring. That’s a mistake. It can trigger allergic reactions, skin conditions, even asthma if you’re not careful. So, treating it with respect is a lot more important than some people realize.
Simple Steps for Personal Protection
Don’t handle this chemical with bare hands. Gloves that handle solvents, like nitrile or neoprene, offer a much better shield than typical latex. I saw coworkers end up with red, itchy hands just from a quick splash. If there’s risk of tiny airborne particles, a proper mask matters, not just those cloth face covers. Eyes need goggles, not just because of splashes, but because any irritation around the eyes gets miserable fast.
Keep Clean and Keep Air Moving
Good ventilation makes a difference. Once, a fume hood in my lab broke down, and folks noticed headaches and coughing within an hour. Even a small amount of vapor in the air can start causing problems. Staying in a well-ventilated space slashes the risk. At home or in a small shop, crack a window and use a fan at the minimum.
Handling Spills and Waste
Small spills happen. Wipe them up right away with disposable towels—just tossing them in the regular trash puts others at risk. Seal the used materials in a bag, and drop them with hazardous waste so they don’t end up somewhere they shouldn’t. Never mix this stuff down the sink. Municipal water systems can’t always break it all down, and that affects everyone’s health downstream.
Every Label Tells a Story
People skip the labels far too often. That’s risky. The symbols and warnings printed on the container can save you from learning lessons the hard way. I remember a friend who ignored the “respiratory hazard” symbol, and a cough lingered for weeks. Those markings aren’t just legal red tape; they’re there because people have gotten hurt before.
Medical Response Means Acting Fast
If skin starts burning or eyes water up, rinse fast and let a supervisor know right away. Don’t try to tough it out—chemical exposures turn nasty without warning. If someone swallows it or has trouble breathing, call emergency services without hesitating. Timely response can keep small incidents from turning into emergencies.
Building a Safety Culture Pays Off
No matter if it’s a university lab, salon, or small business, people look out for each other. New team members should watch veterans who treat chemicals with care: gloves, masks, extra ventilation, knowing what to do if something spills. Supervisors can encourage folks to stick to these steps by running short refreshers or posting reminders right near the chemicals.
Better Safe Than Sorry
Ignoring 1,4-Phenylenediamine’s dangers doesn’t make them go away. Respecting the risks keeps people out of the doctor’s office and lets everyone work more comfortably. Staying safe means thinking ahead, not looking back with regret.
How should 1,4-Phenylenediamine be stored?
Understanding the Risks
Storing 1,4-phenylenediamine calls for an approach shaped by real-world experience and solid science. This chemical earns attention because of its potential health hazards—skin irritation, respiratory issues, and even long-term effects if inhaled or handled carelessly. I remember my first job in a college lab, reading labels and obsessing over safety data sheets. Those long hours made clear that the rules exist for a reason. Special handling matters most with compounds like this.
Keep the Chemical Stable
1,4-Phenylenediamine reacts with air and light much faster than some realize. I’ve seen bottles of it turn from grayish to almost black in poorly lit, warm store rooms. To keep the chemical stable, sealed containers go a long way. Choose amber glass bottles or opaque containers to block light. Ditch flimsy snap-cap bottles for screw tops that stop air from leaking in. A cool, well-ventilated cabinet is your friend—never leave this compound sitting in a sunlit workspace or near heating vents.
In my own work, I kept a thermometer and hygrometer close to the storage cabinet so any shift got flagged before it could hurt someone or ruin a batch. 1,4-Phenylenediamine breaks down above regular room temperatures. An air-conditioned chemical storeroom or a specialized fridge, away from any acids or oxidizers, helps keep things safe. Multiple studies back up this practice, showing heat speeds up decomposition and releases more fumes.
Real Health Impacts
Unlike some chemicals that only pose a risk after years of contact, 1,4-phenylenediamine causes rashes and even dangerous allergic reactions in some people after one spill. I watched a coworker develop skin welts after a drop splashed on their wrist. Latex gloves, sturdy lab coats, and splash goggles became non-negotiable in my circle. Never store personal items or food near this chemical—a rookie mistake that cost another friend hours at urgent care.
Ventilation cannot get ignored. Open the cabinet, and that sharp, odd odor signals you to get the air moving. Smart labs have fume hoods close to storage zones, making it easier to handle without breathing in dust or fumes. Even if the workplace isn't state-of-the-art, a simple exhaust fan or cracked window helps.
Labeling, Documentation, and Emergency Prep
Clear, permanent labels matter much more than most expect. Scribbles with dull markers fade so fast. I used thick, chemical-resistant labels and wrote the storage date in bold to keep everything straight. Safety data sheets should be out in the open, not hidden in a file somewhere. Anyone grabbing a bottle should know what they’re holding and how to respond to a spill.
Spill kits lived under the sink in our lab, stacked with absorbent pads, neutralizers, and gloves. The quickest response always came from people who checked the emergency plan ahead of time—not during the crisis. In accidents involving 1,4-phenylenediamine, rapid cleanup limits health risks and protects the rest of the facility.
Working Toward Safer Practices
I trust the habits I developed through years of working around tough chemicals more than any theory. Store 1,4-phenylenediamine in sealed, lightproof containers, keep the room cool and breezy, gear up with personal protection, and keep emergency tools ready. These actions protect you, your coworkers, and anyone who might enter the storeroom. It only takes one slip to show why these routines exist.
What are the common applications of 1,4-Phenylenediamine in industry?
The Role in Dye Production
Walking through the history of synthetic dyes, you’ll always run into 1,4-Phenylenediamine (PPD). This compound pops up in the textile and hair dye business because it reacts well with oxidizing agents. Once it hits the fabric, it helps lock in dark colors that do not wash out easily. Hair coloring professionals tend to favor PPD for its ability to deliver strong, lasting shades, especially the darker tones customers ask for again and again.
Its popularity isn’t without some warnings. The CDC has flagged allergic reactions, with symptoms ranging from mild irritation to severe dermatitis. Salon workers take this seriously by using gloves and running patch tests. Regulators in the European Union and the US both set concentration limits, aiming to balance consumer safety with the demand for effective products.
Polymer and Plastic Engineering
PPD finds another job as a building block for certain engineering plastics. It reacts with isophthaloyl or terephthaloyl chloride to form aramid fibers like Kevlar. Most people think of Kevlar when talking about bulletproof vests, but it also lines up in uses like reinforced tires, protective gloves, and even aircraft parts. The advantage here comes down to its light weight and remarkable strength for resisting cuts or breaking under strain.
After spending time in a manufacturing facility, I noticed how safety teams prioritize air quality. PPD dust exposure can mean headaches, eye irritation, and other real health risks. Smart industries invest in good ventilation and require workers to wear respiratory gear, not just for compliance, but to protect workers from the long-term impact nobody wants to risk.
Rubber Industry Applications
If you go behind the scenes in tire plants, you'll see how antioxidants slow the breakdown of rubber from sun and oxygen. Certain PPD derivatives, such as 6PPD, play a huge part here by extending the life of rubber products. This means fewer tire failures and less waste—an environmental and economic win. Still, an environmental story burst onto the scene in 2020 when scientists linked 6PPD-quinone—an oxidized byproduct of PPD in tires—to coho salmon deaths in the Pacific Northwest. Now, companies and researchers dig for safer substitutes and better containment strategies, marking a fresh push for sustainable methods in chemical manufacturing.
Pharmaceutical and Specialty Chemical Synthesis
Chemists use PPD to create specialty chemicals and pharmaceuticals. It acts as an intermediate, reacting to form compounds used in everything from curing agents to corrosion inhibitors. This status as a “chemical bridge” fuels innovation, giving drug developers and materials scientists the freedom to build new molecules.
Knowing the wide reach of PPD through so many industries highlights both the utility and the responsibility that comes with its use. Companies that put safety and environmental respect at the core of their process earn trust from their workers, the public, and regulators. Advanced filtration systems, robust worker-safety training, and ongoing eco-toxicity research form the backbone of a healthy relationship with this powerful, no-nonsense chemical.
| Names | |
| Preferred IUPAC name | benzene-1,4-diamine |
| Other names |
p-Phenylenediamine 1,4-Diaminobenzene PPD p-Diaminobenzene para-Phenylenediamine |
| Pronunciation | /ˌwʌn.fɔːr fəˈnɪliːnˌdaɪəˌmiːn/ |
| Identifiers | |
| CAS Number | 106-50-3 |
| Beilstein Reference | 1209242 |
| ChEBI | CHEBI:17617 |
| ChEMBL | CHEMBL1401 |
| ChemSpider | 5467 |
| DrugBank | DB00783 |
| ECHA InfoCard | 03b250735901-45d0-894e-d49c26ba10ba |
| EC Number | EC 203-584-7 |
| Gmelin Reference | 84138 |
| KEGG | C06180 |
| MeSH | D010634 |
| PubChem CID | 1019 |
| RTECS number | SS8050000 |
| UNII | 9FQ1KG01LI |
| UN number | UN1673 |
| Properties | |
| Chemical formula | C6H8N2 |
| Molar mass | 108.15 g/mol |
| Appearance | White to grayish or purple solid |
| Odor | Odorless |
| Density | 1.06 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.03 |
| Vapor pressure | 0.001 mmHg (25 °C) |
| Acidity (pKa) | 25.0 |
| Basicity (pKb) | 6.26 |
| Magnetic susceptibility (χ) | -37.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.643 |
| Viscosity | 1.06 mPa·s (80 °C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 85.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 44.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3306 kJ/mol |
| Pharmacology | |
| ATC code | D08AE17 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes skin irritation, causes serious eye irritation, may cause an allergic skin reaction, may cause respiratory irritation, suspected of causing cancer. |
| GHS labelling | **GHS labelling string:** `GHS02, GHS05, GHS06, GHS08` |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H317, H319, H331, H341, H351, H410 |
| Precautionary statements | P264, P280, P302+P352, P304+P340, P305+P351+P338, P312, P332+P313, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 149°C |
| Autoignition temperature | 644°C |
| Lethal dose or concentration | LD50 oral rat 315 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 80 mg/kg |
| NIOSH | SW4380000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 60 mg/m³ |
| Related compounds | |
| Related compounds |
p-Aminoaniline para-Phenylenediamine 1,2-Phenylenediamine 2,5-Diaminotoluene Benzidine |

