An Editorial Look at 1,4-Hexadiene: More Than a Simple Hydrocarbon
Historical Development and Discovery
Chemistry books once spent little time on 1,4-hexadiene. It’s found its way into the spotlight not through flashy headlines, but by quietly enabling progress in labs and on factory floors alike. In the 1940s and 50s, as organic chemists set their sights on diene chemistry, simple alkenes like 1,4-hexadiene gained real attention. Early experiments focused on understanding its reactivity, and from there, manufacturers learned how to scale up its synthesis, often by cracking larger hydrocarbons or carefully crafting elimination reactions from precursors found in petroleum. Its structure—a six-carbon chain with double bonds on each end—opened up opportunities for both synthesis and industrial modifications. Over the decades, the availability of petrochemical feedstocks and evolving catalytic technologies made it easier to isolate and purify. Its relevance grew not just because it existed, but because researchers and industrial chemists kept finding new uses for it, pushing what might’ve been a back-shelf molecule into newer applications.
Product Overview and Versatile Utility
Today, 1,4-hexadiene shows up in more places than most people outside of chemistry circles realize. As a building block, it brings flexibility to organic synthesis: connect two different things to those double bonds, and you’ve got options that simpler alkenes just can’t offer. Its linear structure, two unconjugated double bonds, and manageable reactivity mean that it brings unique leverage to fields like polymer manufacture, specialty chemicals, and new material design. It turns into a cross-linker, intermediary, or even a target for further functionalization in the hands of a smart chemist. For example, specialty elastomers owe a good deal of their resilience to diene units like 1,4-hexadiene, which can be meticulously woven into larger structures. Once, its uses felt narrow—now it’s an indispensable thread in a huge tapestry of synthetic work.
Physical and Chemical Properties
This colorless liquid, slightly lighter than water, evaporates with a sweet odor that sharp noses in the lab will recognize after a few attempts. It boils just over 80 degrees Celsius and has a flash point low enough to demand respect and careful storage. Its double bonds don’t sit next to each other, so it avoids some of the wild reactivity of conjugated dienes but still plays host to a range of reactions: additions, cyclizations, metathesis—each offers a lead-in to more complex building blocks or polymers. It mixes well with most organic solvents and doesn’t linger long in aqueous solutions, making cleanup and waste management an art in itself. Handling it without ventilation or gloves isn’t wisdom—it’s a shortcut to headaches, literally and figuratively. A bit of experience around volatile organics quickly teaches this lesson.
Technical Specifications and Labeling Concerns
Chemists and quality control labs have long relied on clear-labeling and technical rigor to keep things safe. For 1,4-hexadiene, purity plays a massive role; trace peroxides or moisture can spoil reactions and even trigger dangerous decompositions. Labs scrutinize its physical profile through gas chromatography and NMR, flagging impurities quickly. Standardization hasn’t come at the cost of flexibility, though. Depending on use, some processes need ultra-pure product, while others tolerate a bit of residue from isomeric or similar compounds. But labeling always makes room for hazard symbols: ignore flammability, and corners get cut in ways that regulators, fire marshals, and insurance companies have little patience for. Reading a label sometimes saves lives and always saves time.
Preparation and Synthetic Methods
My early encounters with 1,4-hexadiene came in advanced organic synthesis labs, where we pieced it together using elimination strategies. Industry often turns to catalytic dehydrogenation or selective cracking—think of turning heavier alkanes into lighter ones, then scavenging out the double bonds. Chromium or palladium catalysts come in handy for targeted transformations. Safety protocols for generating and isolating the compound can feel tedious but keep accidents at bay. Side products, especially isomers or conjugated species, threaten purity if reaction conditions slip. Even though the chemistry seems simple on the surface, actually making and handling larger batches of 1,4-hexadiene is demanding without careful engineering and robust process controls.
Chemical Reactions and Modification Pathways
Once in hand, 1,4-hexadiene opens doors. Its isolated double bonds mean it’s game for addition reactions—halogenation, hydroboration, epoxidation, you name it. I’ve used it as a starting point for making cyclic compounds, tapping into Diels-Alder cycloadditions and ring-closing metathesis to create more complex targets. Many synthetic chemists rely on it for such transformations, because its reactivity is neither too wild nor too docile. Industrial teams push it even further, tacking on functional groups or coaxing it into specialty monomers for high-performance polymers and resins. No two labs or plants treat it quite the same way; it adapts to the needs at hand, making it a kind of chemical toolkit workhorse.
Synonyms and Product Names
Finding references to 1,4-hexadiene depends on where and how you look. Some chemists call it hexadiene, while others specify 1,4-hexadiene to dodge any confusion with its 1,3- or 1,5- relatives. You might spot identifiers like “NSC 74127” or see it described in catalogs as “divinylmethane” or similar. The point is, whether you call it by the full IUPAC name or reach for a more casual moniker, you’re navigating the web of nomenclature that forms part of every chemist’s day-to-day grind. Mixing it up in a reaction is easier than sorting out a muddled bottle label.
Safety and Operational Standards
Operators in chemical plants and synthetic labs learn to respect the properties of compounds like 1,4-hexadiene. It evaporates quickly and catches fire easily, making the right ventilation and flame-proofing essential. Gloves, goggles, and fume hoods aren’t optional—they're expected protocol. Years back, before strict OSHA guidelines became the norm, incidents involving fires or inhalation popped up more frequently in the literature. Today, regular audits, air monitoring, and emergency drills keep risks reined in. The industry evolves, sometimes only after enough close calls to drive home the need for constant vigilance. Chemical spill kits sit ready, and everyone working with 1,4-hexadiene learns about its low flash point and potential to form peroxides if mishandled. Safety here isn’t a slogan, it’s a practice grounded in the real hazards people face.
Application Areas: From Industry to Innovation
For people working outside the chemical sector, it’s easy to overlook how foundational compounds like 1,4-hexadiene are to innovation. It has made its mark in creating specialized plastics, acting as a bridging molecule that introduces flexibility and durability in finished products. Polymer chemists take advantage of its ability to cross-link, making it valuable in tire manufacturing, specialty elastomers, and resins used in everything from paints to adhesives. Pharmaceutical research teams turn to such dienes as starting points for more advanced synthesis, especially where the non-conjugated geometry lets them build structures other compounds can’t offer. You’ll also find research projects chasing down new uses in specialty coatings and composite materials, eager to leverage its chemical structure for enhanced performance in demanding applications.
Research, Development, and Continued Curiosity
Over the years, research into 1,4-hexadiene has shifted from curiosity-driven projects to targeted innovation. Academic labs explore new catalysts and reaction pathways, chasing higher yields and cleaner conversions. Industrial development teams probe technologies that minimize waste and energy use, seeking greener routes that align with regulatory changes and growing attention to sustainability. I’ve sat in seminars where teams unpacked new ways to functionalize this diene, turning old chemistry into new business opportunities. Publications continue to emerge, suggesting that even a molecule with decades of history can spark modern breakthroughs. The pace of research matches the growing demand for high-performance materials, and 1,4-hexadiene sits near the center of that hunt.
Toxicity and Environmental Concerns
Toxicity research on 1,4-hexadiene echoes the caution that comes with working around volatile hydrocarbons. Prolonged inhalation or skin exposure irritates, and animal studies point to dose-dependent effects that demand careful handling and storage. Workers with direct contact always use personal protective equipment, and disposal routines follow strict environmental standards to shield air and water from contamination. While not among the most notorious pollutants, its volatility means leaks or spills still contribute to volatile organic compound emissions, something environmental regulators target for reduction. In reviewing the literature and safety data, it’s clear that knowledge of its risks has grown step by step with its applications. Repeated reminders from safety bulletins and industrial hygiene guides keep these hazards in sharp focus.
Future Prospects and Challenges Ahead
Looking ahead, the story of 1,4-hexadiene keeps evolving in step with demand for stronger, lighter, more versatile materials. As companies push for greener processes, they look for new catalytic systems and renewable feedstocks that lessen the environmental footprint of its production. Material scientists eye this molecule as a springboard for next-generation plastics, elastomers, or specialty coatings designed for extreme environments. Emerging applications, like custom monomers for advanced 3D printing resins or smart composites, underscore how foundational chemistry adapts to evolving markets. Yet the path forward bristles with challenges—tightened regulations on volatile organic compounds, safety demands from ever-more complex processes, and the race to find sustainable pathways for large-scale supply. Chemists, engineers, and regulatory experts will keep steering the field, using hard-earned experience to shape the role 1,4-hexadiene plays in tomorrow’s innovations.
What is 1,4-Hexadiene used for?
A Look at What 1,4-Hexadiene Brings to the Table
People might walk past countless objects touched by 1,4-hexadiene without realizing it. Even though this chemical sounds distant or technical, its impact shapes the products folks use daily. Chemists trust 1,4-hexadiene as a handy building block. This clear liquid plays a key role in making plastics, adhesives, and specialty chemicals that drive comfort and convenience.
Turning Simple Molecules into Everyday Comfort
The story behind 1,4-hexadiene runs straight into the heart of plastic production. In places where manufacturers turn out polyethylene or ethylene-propylene rubbers, this chemical helps link molecules together. Those bridges between chains create products that stay stretchy or tough, depending on how people want to use them. Car tires, weather-resistant seals, and even soft dashboards get their bounce and resilience from these connections.
As someone who lives in a place with wild swings in temperature, I've seen firsthand how the wrong rubber seal can crumble or freeze. Products strengthened with chemicals like 1,4-hexadiene keep wind and water where they belong—on the outside. That tight seal has saved my kitchen floor from more than one winter storm.
Behind-the-Scenes: Specialty Chemicals and Unique Uses
Some jobs call for chemical reactions that need just the right touch. 1,4-Hexadiene steps in here, too. Because this molecule has two reactive spots, it becomes a useful ingredient for making specialty coatings, flavors, and fragrances. Labs also use it to study how molecules change shape or link together. Sometimes, it's a stepping stone to more complicated products used in medicines or electronics.
Risks, Regulations, and Responsibility
Not every powerful tool comes without a cost. Years of work in manufacturing have taught me that with strong chemicals comes real responsibility. Exposure to 1,4-hexadiene in high doses can cause irritation or raise concerns about long-term health effects. Strict training and careful storage keep workers safe, yet companies need to stay updated about new science and tougher rules. The U.S. Environmental Protection Agency and OSHA both expect close tracking when 1,4-hexadiene enters a workplace.
Moving Toward Greener Solutions
People in industry are feeling the pressure to go green. Biobased alternatives aren't ready to fully replace the chemical, but labs push hard to limit any release into the air. Upgraded filters, closed systems, and tracing every drop start to make a difference. From my experience, a company’s culture shapes how seriously teams take safety and the environment. Regular training and real investment in better technology pay off—both for people and for the planet.
The Bottom Line for 1,4-Hexadiene
The reach of 1,4-hexadiene runs quietly through a wide array of everyday essentials. It gives materials the strength and stretch modern life demands. By paying real attention to safe handling and sustainability, companies not only protect their workers but also build trust with their communities. As smarter materials come along, and as science moves forward, the goal remains the same: use the right chemistry without ignoring the human cost.
What are the safety precautions for handling 1,4-Hexadiene?
Understanding the Risks
1,4-Hexadiene brings a set of challenges to anyone handling chemicals in a lab or industrial setting. This colorless liquid has a strong, unpleasant odor and is known for its high volatility. Vapors can catch fire quickly and create explosive mixtures with air. Just a small spark near an open container can start a fire. Inhalation can irritate the nose, throat, and lungs, making breathing difficult in high concentrations. On the skin, it can cause dryness, redness, and even burns.
Personal Protective Equipment (PPE)
The first thing I make sure of before touching any container of 1,4-hexadiene sits with personal protection. Nitrile gloves give a barrier between the skin and the chemical. Standard lab coats or coveralls and chemical splash goggles help keep skin and eyes out of harm's way. I always double-check the gloves for holes. A face shield gives extra safety when pouring or transferring. Anyone who has ever dealt with a chemical splash knows the value of a well-fitting pair of goggles. It saves a trip to the emergency room and saves your eyesight.
Ventilation and Engineering Controls
Ventilation in the workspace cannot get ignored. Fume hoods move dangerous vapors away from where people breathe. An open lab bench does not offer enough protection, even if the room feels big. Vapors hang close to the ground and quickly spread. Proper ventilation systems keep air moving and dilute any leaks before they cause health hazards.
Safe Storage Practices
1,4-Hexadiene should never get stored near heat, ignition sources, or direct sunlight. The material reacts easily with oxidizers and acids, making fire and explosion a very real threat. Flammable liquid cabinets give a safe location far from incompatible materials. Tight-fitting, clearly labeled containers keep the vapor where it belongs. I always use a double-containment approach: chemicals stored inside containers placed within a larger secondary tray, just in case of spills.
Spills and Emergency Response
If a spill occurs, rushing over with paper towels won't cut it. Specialized spill kits have absorbent pads, neutralizers, and disposal bags just for chemicals like this. Evacuating non-essential personnel, increasing ventilation, and using PPE are the immediate steps. The chemical quickly spreads across floors, so quick thinking and preparation prevent slips, exposure, and fire. Safety showers and eyewash stations within easy reach mean fast action is always an option.
Training and Communication
Training saves lives. Nobody should get handed a bottle of 1,4-hexadiene without proper instruction. Review safety data sheets before working — these documents cover exposure limits, symptoms, and proper disposal. Routine safety drills build muscle memory, so everyone knows where exits and safety equipment are without second-guessing in an emergency.
Proper Disposal
Chemical waste shouldn't go down the drain. Waste containers labeled for flammable organics keep used or unwanted 1,4-hexadiene separate from other lab trash. Licensed hazardous waste contractors take over once lab staff fills the containers. Bypassing disposal steps creates health and environmental problems downstream.
Handling 1,4-hexadiene demands focus, preparation, and respect for its risks. With the right habits, attention to personal protection, storage, and teamwork, the laboratory or plant environment remains safe for everyone involved.
What is the chemical formula and structure of 1,4-Hexadiene?
1,4-Hexadiene: More Than Just a Hydrocarbon
Everyday chemistry often slips through the cracks, especially with something as straightforward as 1,4-hexadiene. The chemical formula—C6H10—shows up in textbooks and lab reports. Not exactly headline-grabbing, but dig a little deeper and you find the story’s a bit more involved. Six carbons bonded in a chain, ten hydrogens attached, and what sets 1,4-hexadiene apart: two carbon-carbon double bonds at both ends of the chain. The full structure goes like this: CH2=CH–CH2–CH2–CH=CH2. Those double bonds don’t hide in the middle; they frame the molecule on either side.
Structure Defines Character
Folks with some synthetic chemistry under their belt see why the structure makes 1,4-hexadiene useful. It’s not just a string of atoms—those double bonds at the 1 and 4 positions shape everything about the molecule’s personality. These are called terminal dienes. In practice, there’s a noticeable difference in reactivity and handling compared to something like 1,3-hexadiene. Double bonds this far apart behave almost like two separate teams. That makes them interesting for reactions where you want selective reactivity—think of crosslinking, cyclization, or even forming more specialized organic molecules.
Building Blocks in Synthesis
Chemists pick 1,4-hexadiene when they need a scaffold that’s both reactive and versatile. Many industrial polymer processes use it as a starting point. As an example, creating certain plastics or specialty polymers often means getting the spacing of those double bonds just right. In my old university research days, we’d use it as a testbed for catalytic reactions—especially those looking for high selectivity over competing pathways. The terminal diene structure allowed for cleaner transformations, leading to fewer unwanted side-products.
Why Attention to Detail Pays Off
Ignoring the exact structure in chemistry leads to real mistakes. I’ve seen students lose hours of time because they mixed up 1,4-hexadiene with its 1,3 cousin. One is good for certain polymerizations; the other tends to tangle things up. The physical properties change too: boiling point, stability, and even how the molecule interacts with catalysts. That’s why research and development teams check chemical structures carefully before scaling up any process. A small mistake in the formula or structure could mean wasted batch runs, safety hiccups, and higher costs.
Choosing the Right Path with Science
Understanding 1,4-hexadiene’s formula and layout isn’t just academic nitpicking. For anyone exploring new synthetic routes or tackling ambitious materials projects, accurate chemistry knowledge sets you up for success. There’s value in slowing down to draw those structures, count those hydrogens, and run a quick reactivity check in your mind. That discipline helps catch errors before they leave the bench. Sharing experiences and clear, accurate information also supports newcomers and seasoned pros facing real-world challenges in the lab. Small molecules like 1,4-hexadiene prove that chemistry’s details matter, every single time.
How should 1,4-Hexadiene be stored?
Understanding What You’re Dealing With
Anyone who’s worked around chemicals knows there’s no room for guesswork. 1,4-Hexadiene isn’t a household name, but the risks aren’t hard to find. It’s a colorless liquid that brings flammability and reactivity issues to any space—two facts that ought to grab attention before the first drum arrives on site. The real world isn’t a lab safety poster, but experience shows that careful storage makes a difference when lives and dollars are on the line.
Flammability Comes First
Countless fires in industry start with ignoring what makes chemicals tick. 1,4-Hexadiene has a low flash point—just under 1°C. That means even a mild spring day can send vapors into the air. Vapors reach ignition sources and things go from routine to disaster in no time. Storing this chemical in a cool, well-ventilated place is non-negotiable. I remember old storage rooms packed without thought for temperature, clocks ticking until an emergency. Real oversight means keeping it away from direct sunlight, boilers, and hot pipes.
Choosing the Right Container
Metal or glass containers keep 1,4-Hexadiene stable, but not all metals are wise picks. Copper, silver, and their alloys can react and create dangerous situations. I’ve seen poorly chosen barrels corrode in a few months, leaving leaks everywhere. Stainless steel works better for the long haul. No one wants a mystery puddle growing in the dark corner of a storage room. Containers need clear labels—hazard symbols, content details, and date of storage help keep everyone in the loop.
Air and Moisture—Hidden Dangers
Water doesn’t just dilute; it reacts with some chemicals to create bigger headaches. Humidity can change the properties of stored 1,4-Hexadiene. It helps to keep containers tightly sealed and, if possible, keep inert gases like nitrogen over the liquid. This prevents air from getting in and starting slow reactions that might not show up until it’s too late. From experience, accidents often start with minor leaks and routine neglect.
Keep It Separate
In storage, proximity matters. Bleach and acids should never sit on the same shelf as 1,4-Hexadiene. Mixing with oxidizers or strong acids bumps up the risk of unforeseen reactions. I’ve worked in places where chemicals shared corners out of convenience—cleanup costs and downtime rocket after a spill or vapor release. Using dedicated storage cabinets marked for flammable liquids isn’t just compliance—it’s protection for workers.
Stay Ahead With Training and Planning
Genuine expertise means more than reading a datasheet. Staff on the floor need regular training—how to spot leaks, use protective equipment, and react if the worst happens. Emergency kits don’t belong in a locked manager’s office. Spills happen fast; a team needs both knowledge and equipment within arm’s reach.
Fixing the Gaps
Technologies like temperature monitoring and automatic venting systems add safety without much hassle. Investing in double containment is smart policy, especially in high-traffic areas. Check inventory regularly—expired or compromised containers turn small risks into major threats. Keeping tight records and practicing what-ifs with the crew go further than more signs on the wall.
Practical Steps Make All the Difference
Safe storage of 1,4-Hexadiene calls for respect for the hazards, the right equipment, and a culture that rewards speaking up. Every workplace that moves away from shortcuts helps build safety that lasts.
What are the physical and chemical properties of 1,4-Hexadiene?
Why 1,4-Hexadiene Catches Attention
Anyone spending time in a lab or working with plastics eventually hears about 1,4-hexadiene. It's a colorless liquid that might not stand out among bottles on a bench, but there’s a reason researchers keep coming back to it. You pick it up, and the first thing you notice is a sharp, sweet smell—unmistakable and not at all subtle. With a boiling point close to 64°C and a density lighter than water, it evaporates quickly. That volatility makes handling it a bit tricky. Nobody wants vapors floating around, especially since breathing it in too much can make you dizzy or give you headaches.
Chemical Nature: Those Double Bonds Aren’t for Show
There are two double bonds in a six-carbon chain, spaced apart with carbons in between. That's what gives 1,4-hexadiene its name and its punch. It isn’t just lying there, waiting. Put it with a catalyst, and those double bonds open up a range of options for building longer molecules, or cross-linking—real workhorse reactions in synthetic chemistry.
This isn’t just a fun fact for chemists. Those double bonds give manufacturers the control they want. I once helped on a project where this compound played a key role in polymer modification. That flexibility let us tweak the strength and flexibility of the end product, which was more useful than trying to reinvent everything from the ground up.
Hazards: Not Just Numbers on a Sheet
You open a bottle of this liquid, and it starts evaporating before you know it. It has a flash point below room temperature—meaning a single spark in the wrong spot can set off a fire. That’s not something you want to learn the hard way. Eye and skin irritation happen fast if you skip gloves or goggles. It feels obvious to say “work in a fume hood,” but the warning really means something here. I remember sitting through a safety debrief after a chemist spilled some on their clothes. Quick action stopped anything lasting, but that stuck with me.
The long-term risks don't get the same headlines. There’s evidence that overexposure hurts the nervous system and lungs in animals. Even if there’s not a family history of cancer, working with things that might cause tumors keeps me thinking about proper handling rather than shortcuts.
Why Properties Shape How Everyone Uses It
People use 1,4-hexadiene in plastic production, pharmaceutical research, and as a building block for specialty chemicals. That all hinges on the two double bonds. It slides into reactions that produce nylon intermediates or help craft resins with tailored flexibility. I’ve seen it used in the lab to create new catalysts—test reactions that scale up when trial batches look promising. Its clear, liquid state keeps shipping simple but only if containers stay sealed and cool.
Tougher regulations in recent years push for safer transport and handling protocols. That only feels right after watching small mistakes create big problems. Better training, labels that actually catch your eye, and automatic ventilation systems keep labs safer. Rather than cutting corners, doubling down on practical safety pays off quickly.
I see a place for greener chemistry moving forward—perhaps designing catalysts that lower the risks, improving recycling for polymer waste, or tailoring uses so less residual chemical winds up in the environment. Chemistry shapes more than bottles and beakers; it touches everyday life. Getting the basic properties right is where the difference starts.
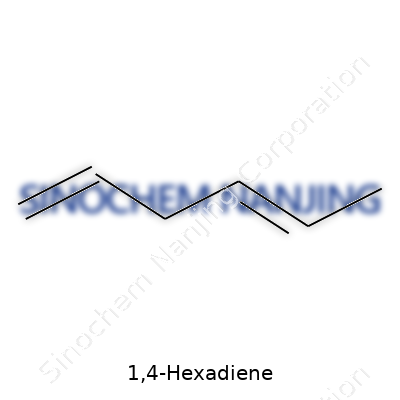
| Names | |
| Preferred IUPAC name | hexa-1,4-diene |
| Other names |
1,4-Divinyl Butadiene, 1,4-diethylidene Diallyl Hexa-1,4-diene Tetramethylene, 1,4-diene |
| Pronunciation | /ˌwʌn.fɔːrˈhɛk.səˌdaɪ.iːn/ |
| Identifiers | |
| CAS Number | 592-45-0 |
| Beilstein Reference | 1360368 |
| ChEBI | CHEBI:51170 |
| ChEMBL | CHEMBL15334 |
| ChemSpider | 63845 |
| DrugBank | DB13825 |
| ECHA InfoCard | 03d8423e-2d26-4d8e-bbba-9f6c437d4f2d |
| EC Number | 211-168-2 |
| Gmelin Reference | 821 |
| KEGG | C01938 |
| MeSH | D006526 |
| PubChem CID | 11587 |
| RTECS number | MK3850000 |
| UNII | 9B1G0I5VVR |
| UN number | UN2049 |
| Properties | |
| Chemical formula | C6H10 |
| Molar mass | 82.15 g/mol |
| Appearance | Colorless liquid |
| Odor | mild pleasant odor |
| Density | 0.675 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 2.98 |
| Vapor pressure | 3.8 kPa (20 °C) |
| Acidity (pKa) | 44.2 |
| Magnetic susceptibility (χ) | -52.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.423 |
| Viscosity | 0.642 mPa·s (20 °C) |
| Dipole moment | 0.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 276.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | ΔfH⦵298 = 85.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3724 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H315, H319, H335 |
| Precautionary statements | P210, P260, P280, P305+P351+P338, P312, P370+P378, P403+P235 |
| NFPA 704 (fire diamond) | 1-2-2-膺 |
| Flash point | -18 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | 1.4–9.7% |
| Lethal dose or concentration | LD50 oral rat 3160 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 6700 mg/kg |
| NIOSH | PA9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1,4-Hexadiene: Not established |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 750 ppm |
| Related compounds | |
| Related compounds |
1,3-Hexadiene 1,5-Hexadiene Hexene Cyclohexene 1,4-Butadiene 1,4-Pentadiene |

