An Editorial Look at 1,4-Dihydroxy-2-Butyne: More Than a Chemical Formula
Historical Development
A deep dive into 1,4-Dihydroxy-2-Butyne reveals a timeline built on curiosity and the pursuit of richer organic chemistry. Sitting at a crossroad between old-school synthetic routes and modern application-driven research, this compound speaks to the journey of chemical discovery that borrows both from the theoretical groundwork of the mid-20th century and the hunger for new functionality in pharmaceuticals and materials science. Chemists in early decades understood simple alkynes and diols within the scope of basic functional groups, but 1,4-Dihydroxy-2-Butyne pushed boundaries with its dual reactivity, offering two hydroxyl groups and a reactive triple bond in a minimal four-carbon frame. Its story is filled with incremental efforts in improving selective synthesis, usually tied to the ambitions of synthetic organic labs stretching into new classes of target molecules for drug development and specialty polymers.
Product Overview
Defining 1,4-Dihydroxy-2-Butyne isn’t just about spelling out its skeletal structure. It’s about recognizing a small molecule packed with reactive potential, thanks to both hydroxyl groups and the alkyne. These features turn it into a workhorse intermediate — sometimes a building block on the way to more complex targets. Sparking the interest of industrial chemists and academic researchers alike, it represents the kind of molecular simplicity that manages to avoid obsolescence, sticking around year after year because it earns its keep in the shops and processes that rely on versatile molecules.
Physical and Chemical Properties
In the lab, 1,4-Dihydroxy-2-Butyne shows off a modest molecular weight, with a colorless to pale yellow appearance, and an ability to dissolve in water and common polar solvents. Both water solubility and reactivity make it easier to use in aqueous-phase reactions, which goes a long way in reducing the messiness that often plagues organic processes. Its odor is typically on the harmless side, not the pungent or alarming sort you get from reactive alkynes like propargyl alcohol. The melting and boiling points fit comfortably in the range needed for liquid-phase synthesis, yet the substance packs enough volatility and sensitivity that careful storage keeps it stable. These details might seem minor, but even in the best-equipped lab, the difference between ease and hassle often comes down to everyday handling.
Technical Specifications and Labeling
Market offerings regard purity above almost anything else, with commercial suppliers typically specializing in high-purity lots and measured water content. For those working on biomolecular synthesis, trace metals or peroxides spell trouble, so reputable sources pay attention to residual contamination. Detailed labeling goes beyond just hazard information, aiming for batch traceability and storage instructions that keep shelf-life from slipping away unnoticed. I have come across cases where analytical chemists appreciate data on residual solvents and UV spectra, offering an extra layer of confidence in method development. Accurate labeling doesn’t just tick regulatory boxes — it reduces the risk of ruining multi-step syntheses, which can cost weeks of effort with a simple mislabel.
Preparation Method
The synthesis of 1,4-Dihydroxy-2-Butyne usually leans on organic transformations starting with simple precursors like formaldehyde and acetylene, fused together through addition reactions and controlled oxidation. These routes often need strong bases and careful attention to temperature and purification, since the compound doesn’t take kindly to sloppy techniques. Over-oxidation and side-product formation stay as ever-present risks, especially in large-scale runs where batch consistency suddenly gains importance. Many in academia have struggled through glassware nightmares with scaling up the classic approaches, and any process tweaks that save time or avoid hazardous reagents have always gotten a warm welcome from process chemists.
Chemical Reactions and Modifications
Both the terminal alkyne and vicinal diol moieties open up a wide range of chemistry. The triple bond allows for well-known coupling or addition patterns and clicks neatly into cycloadditions. Hydroxyl functions invite protection and esterification, with easy toggling between more or less reactive partners. Synthetic strategies sometimes target this molecule for making complex molecules with multiple chiral centers due to its straightforward structure and manageable reactivity. Cross-couplings and further oxidations can turn 1,4-Dihydroxy-2-Butyne into everything from flavor precursors to enzyme inhibitors, highlighting its draw as a reliable launchpad for bespoke chemistries.
Synonyms and Product Names
People in chemical supply business and researchers in the lab bounce between various product names, including Butynediol, 2-Butyne-1,4-diol, and Butynediol-1,4. Catalog codes and systematic IUPAC names hover in the background, though for quick conversations everyone seems to settle on the shorter, punchier synonyms. Understanding which name to search in literature or which name to order from a supplier can trip up even seasoned professionals, especially with translation or regional differences sneaking into play. I’ve seen younger chemists frustrated by mismatches between labels and research papers, a reminder to double-check registry numbers before launching a synthesis.
Safety and Operational Standards
No room here for shortcuts. Handling 1,4-Dihydroxy-2-Butyne takes common-sense PPE — gloves, goggles, and good ventilation stay non-negotiable in any serious lab. The molecule’s toxicity is not considered extreme compared to stronger alkynes or industrial solvents, though exposure limits and the potential for skin or eye irritation call for respect. Reactions conducted under nitrogen or argon avoid unwanted side reactions with air. Waste management procedures focus on quenching reactive intermediates and managing water-miscible organics, with spill cleanups often featuring activated charcoal or similar neutralizing measures. For those in larger-scale production, adherence to workplace safety standards keeps minor accidents from turning into reportable incidents.
Application Area
The uses of 1,4-Dihydroxy-2-Butyne reach across several industries. In specialty polymers, it introduces rigidity and precise crosslinking thanks to its combination of hydroxyls and an alkyne. Pharmaceutical teams prize the molecule as a synthetic intermediate, allowing for the construction of active pharmaceutical ingredients where linear or conjugated dials and alkynes mean something critical for bioactivity. Research into new bioactive molecules or next-generation materials both lean on this compound’s reactivity, leveraging its promise as a springboard to more complex molecules. Paints, resins, and electrochemically active films sometimes gain improved characteristics from its presence due to its clean, persistent structure.
Research and Development
Academic labs and industrial R&D both find ways to push 1,4-Dihydroxy-2-Butyne into new chemical territory. Recent work often explores new catalysts for its functionalization, looking to simplify once-painful steps and cut solvent use. Stereoselective methods, greener synthetic routes, and asymmetric synthesis all see steady attention, especially as regulations and costs begin squeezing process efficiency harder and harder. For anyone who’s slogged through multi-step syntheses where inconsistent reagents or harsh conditions sap morale and budget, improvements in 1,4-Dihydroxy-2-Butyne chemistry serve as a welcome relief. Each advance brings another handshake between synthetic efficiency and practical application, a trend that has not lost steam since the molecule first found its place in chemical supply catalogs.
Toxicity Research
Research teams have logged toxicity data over the years, building a picture that sits between mild irritant and moderate risk depending on exposure level and route. Oral and dermal data in animal models show clear dose-response effects. Risk assessment for workplace exposure focuses on minimizing inhalation and contact with eyes or mucous membranes. Disposal and spill handling rely on avoiding environmental release, as aquatic toxicity rises along with concentration. Compared to bulk commodity chemicals, 1,4-Dihydroxy-2-Butyne rarely causes severe problems in routine handling, but it still deserves solid discipline in labeling, storage, and PPE adherence. The drive for safer analogues or handling procedures comes from the same impulse that has always guided responsible lab workers.
Future Prospects
The ongoing search for more efficient chemical processes and better functional molecules shows no signs of slowing down. As green chemistry principles guide future research, the combination of water solubility, manageable reactivity, and versatility in 1,4-Dihydroxy-2-Butyne keeps it in the game. The push for new pharmaceuticals and advanced polymers means this compound’s unique mix of functionality still matters. I have seen how incremental improvements stack up quickly in the context of coordinated R&D efforts, allowing old reagents like this to evolve and meet new demands. Regulatory, environmental, and cost pressures all play a part in shaping which chemicals stand the test of time, but 1,4-Dihydroxy-2-Butyne holds onto its appeal as a reliable, adaptable tool for modern chemists keen to shape things at the most basic structural level.
What is 1,4-Dihydroxy-2-Butyne used for?
Everyday Chemistry That Shapes Industry
Plenty of people never hear about 1,4-Dihydroxy-2-Butyne, though its roots run straight through modern industry. The chemical, often written as DHB, stands as a building-block in labs across the world. One look at its molecular structure, with those two reactive “hydroxy” ends, reveals why people in the know reach for it. That dual reactivity gives chemists a way to add specific properties to products we all use—without turning to harsh or unstable chemicals.
Why Synthetic Chemistry Leans on DHB
Chemists value DHB because it opens new doors in organic synthesis. Its structure gives them power to create new compounds. Taking DHB, a chemist can make everything from pharmaceuticals to materials with unique electrical properties. For example, research into cancer-fighting drugs sometimes starts with structures built around DHB, thanks to its ability to form strong bonds in core molecular rings. The real challenge is finding chemicals that react well and predictably, and this compound steps up.
During my time as a research assistant in a university lab, we turned to DHB for making intermediates in dye production. The compound’s ability to participate in cross-linking meant that new shades and properties became available, right at the bench. That hands-on experience taught me that choosing the right backbone for a synthesis changes everything—yield, purity, and even the safety of a process.
Applications in Polymers and Materials
Polymer chemists love DHB for another reason—it acts as a flexible linker. In practice, this means they can adjust the physical properties of plastics and synthetic rubbers, working with the hydroxy groups to improve compatibility or change durability. In the electronics world, researchers experiment with DHB when they want to make materials that handle electricity differently than standard plastics. These changes can help build better batteries and more efficient solar panels.
I once saw a startup testing several additives for flexible electronics, and DHB made the shortlist because its central triple bond creates rigidity and stability in polymer chains. Building blocks like these give designers more control over final products, whether that's a bendable display or a strong protective coating.
Safeguarding Health and the Environment
With any industrial chemical, attention turns to health and environmental impact. Handling DHB calls for caution, since that kind of reactivity doesn’t always stop where you want it. It can irritate the skin and eyes—some studies show respiratory issues with frequent contact. Regular safety protocols work well: gloves, goggles, lab ventilation. Safe disposal matters too, since reactive chemicals sometimes linger in the local ecosystem if poured down the drain.
Plenty of green chemists now look for replacements or alternative routes that skip over problematic compounds. Industry-wide, progress grows by pairing innovation with responsibility, aiming to create new materials that don’t pose hidden risks down the line.
The Road Ahead: Rethinking Uses and Risks
New methods keep arriving that make use of DHB’s strengths while reducing waste and improving worker safety. One promising approach involves better catalyst systems during synthesis, cutting down on energy use and side-products. For companies making electronics or textiles, keeping up-to-date on safer procedures and alternatives matters—for both business and public trust.
The world changes as technology and chemistry move forward. DHB serves as a small but vital cog in those changes, giving industries tools to innovate while reminding everyone involved about careful stewardship from start to finish.
What is the molecular formula and weight of 1,4-Dihydroxy-2-Butyne?
Inside the Molecule: What Makes Up 1,4-Dihydroxy-2-Butyne?
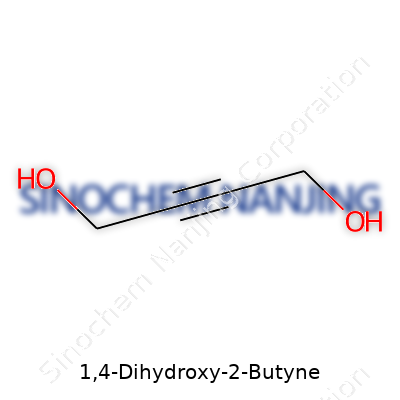
The molecular formula for 1,4-Dihydroxy-2-Butyne is C4H6O2. Beneath the surface, this means you’re looking at a molecule with four carbon atoms, six hydrogens, and two oxygens. Think of it as a butyne skeleton—the “but” comes from its four carbons—transformed by hydroxyl groups (-OH) on both ends and a triple bond tucked between the second and third carbons.
This layout isn’t just a tidy chemical notation. Hydroxyl groups turn basic skeletons into far more reactive, interesting compounds. Organic chemists, especially those eyeing the synthesis of new plastics, complex polymers, or even pharmaceuticals, pay close attention to how these groups influence reactivity. The triple bond isn’t just a line on a piece of paper; it changes the way this molecule behaves in a lab or manufacturing setting.
Molecular Weight Matters: C4H6O2
Science keeps things practical. Adding up the weights of each part brings us to a molecular weight of 86.09 g/mol for 1,4-Dihydroxy-2-Butyne. Here’s how you can check the math:
- Carbon (C): 4 atoms × 12.01 = 48.04
- Hydrogen (H): 6 atoms × 1.008 = 6.048
- Oxygen (O): 2 atoms × 16.00 = 32.00
Pull those together, and you get 48.04 + 6.048 + 32.00 = 86.09 grams per mole—a vital number any chemist tracks before heading into the lab or the factory floor.
Why Does This Matter?
Molecular formulas act like blueprints, but the importance goes past structures or weights. On a factory floor, the quality of the end product often boils down to knowing exactly what’s going into your beaker. If mistakes creep into calculations, the final product might not meet safety standards, or worse, might end up dangerous. In my experience, even small deviations in chemical makeup led to reactions stalling or, sometimes, explosions no one saw coming.
In pharmaceutical development, the purity and exact structure of a molecule set the tone for downstream safety. If you’re looking at potential anti-cancer agents or fine-tuning antifreeze, you rely on formulas and weights to make calculations that keep batches consistent. Without accuracy, even the most promising research ends up worthless.
Staying Accurate with Information
With digital databases now easier to reach than ever, keeping chemical records straight turns less on memory and more on cross-checking facts. Experienced chemists value sources like PubChem or ChemSpider. I’ve seen new faces in labs make avoidable errors from grabbing data off blogs or questionable websites. Sticking close to peer-reviewed literature or major chemical catalogues guarantees that the basis for research, process optimization, or product formulation stays rock solid.
Relying on shared, accepted chemical values doesn’t just help avoid embarrassing lab mishaps; it also underpins the trust customers put into industrial and pharmaceutical products. If regulations tighten, as they have for many solvents and chemical intermediates lately, being a stickler for accuracy saves both time and reputation.
Ways Forward: Reducing Risk and Misunderstanding
Chemistry does best when transparency and precision lead the process. Standardizing how information about substances like 1,4-Dihydroxy-2-Butyne travels from researchers to engineers sets up stronger systems for quality and safety. Lab teams can help by double-checking molecular weights and formulas during research and before running full batches.
Investing in ongoing training for young chemists and quality assurance professionals pays off. My teams saw better outcomes when we rehearsed calculations, shared peer-reviewed reference tools, and gave everyone the right to question unclear specs. The stakes in chemical manufacturing and research make those routines, not afterthoughts, but essentials.
So knowing the molecule and its weight keeps far more than numbers straight—it keeps research, products, and daily work safe and reliable.
Is 1,4-Dihydroxy-2-Butyne hazardous or toxic?
Understanding the Compound
1,4-Dihydroxy-2-butyne doesn’t win any popularity contests outside of chemical engineering circles, but it tends to pop up from time to time in specialty manufacturing and research labs. Its structure packs both alcohol and alkyne groups—giving it interesting reactivity but also demanding some respect during handling.
Safety Concerns in Everyday Handling
My first interaction with a compound like 1,4-dihydroxy-2-butyne reminded me how much even a small chemical can impact lab safety. The compound isn’t well-known in households, but in a research environment, you see the safety data sheets flagged with warning codes. People often overlook that chemicals, even outside the big-name toxins or corrosives, can carry significant health risks.
Direct skin contact or inhalation tends to be the main concern. The molecule can irritate eyes and skin, and high enough doses could lead to respiratory issues. Like many alkynes, this compound holds some potential for toxicity—not as dramatic as something like cyanide, but not benign either.
Evidence From Research and Regulations
Studies haven’t painted an extremely vivid picture of long-term toxicity, partly because it isn’t widely used outside labs. Still, manufacturers and safety bureaus treat it with caution. The National Center for Biotechnology Information lists it with hazard warnings, pointing out its ability to irritate mucous membranes and the potential, with certain exposures, for more serious harm.
In industrial environments, material safety data sheets often recommend working under fume hoods, using gloves and eye protection, and sometimes even added ventilation. OSHA and similar regulators don’t always set formal exposure limits for every chemical, but experienced chemists rarely skip precautions with reactive organics like this one.
Why Caution Matters
For those who might dismiss lesser-known compounds as “safe enough,” history offers plenty of reminders. Lab workers sometimes assume lower-volume chemicals aren't as big a deal, but risk adds up over time. My own lab experience showed that the difference between a routine day and a hazardous situation sometimes boils down to a splash or a missed leak.
Chemical reactivity piles onto the health risk, too. 1,4-dihydroxy-2-butyne carries functional groups that can spur unwanted reactions, especially if it meets strong acids or bases. That raises both safety and environmental stakes, especially if waste ends up in the wrong place.
Pushing for Safer Practice
The easiest way to dodge regret with chemicals: respect the warnings and use protective equipment every single time. Even outside the strictest labs, things like gloves, goggles, and real ventilation make a big difference. Training can’t just be the quick slide deck at orientation—it takes on-the-job reminders, questions, and time spent learning from mistakes.
Disposal deserves more attention than it gets. Compounds with reactivity or lingering toxicity can’t simply flush away. Labs should lean on chemical waste services and double-check local rules, because even a few grams sent down the drain can cause headaches for water treatment or environmental regulators later on.
Making Better Choices
Chemistry advances by solving pesky problems, not just discovering new reactions. Investigating safer substitutes or scaled-down synthetic routes keeps people and the environment out of danger. Asking tough questions about a compound’s true risk—especially one like 1,4-dihydroxy-2-butyne—pushes responsible research forward. My experience says that those extra questions and careful habits save people from real harm, every single year, even with the small-batch compounds.
What are the storage and handling recommendations for 1,4-Dihydroxy-2-Butyne?
Why Proper Storage Matters
1,4-Dihydroxy-2-butyne can be found in chemical labs and some industrial sites. It isn’t just another bottle on the shelf—those working with it recognize quickly that its instability can pack a punch. Over the years, I’ve learned that the fear of a chemical spill or unwanted decomposition isn’t overblown. Even small amounts of moisture or contamination become troublemakers inside storage areas. Those worries aren’t just lab legend; it only takes one mix-up to see how things can go sideways. Data from safety sheets warn about the risk of spontaneous reactions or even fires if this compound gets cozy with the wrong substances or sits under the wrong conditions. That’s enough to give anyone second thoughts about sloppy storage.
Understanding the Risks
Leaving 1,4-Dihydroxy-2-butyne in an open container doesn’t spell good news. The liquid is not only flammable but reacts with air and moisture. Vapors may drift, and if the environment isn’t right, the stuff starts breaking down faster. If you’ve ever seen the crusty crystals that can form at a bottle’s lip, you’ll know why tightly sealed containers make a difference. This isn’t just about cost: The health hazards are real, including skin and eye irritation or even more severe toxic effects. There’s no upside to cutting corners on containment. Even small leaks, over time, can contaminate a whole storage area and make cleanup work a nightmare. The ripple effect, from fire risk to health scares, extends beyond one person at the bench.
Simple Fixes that Help
Solid habits beat fancy technology. I’ve put a heavy focus on keeping these chemicals in airtight glass bottles. You don’t want plastic here, as this compound doesn’t always play nice with softer materials. Keep containers in a cool, dry spot—low humidity and away from sunlight. Sunlight can change the game, triggering reactions in the liquid that nobody asked for. A good rule: stick temperature controls on the room and keep the thermostat steady below room temperature, if possible. Even on hectic days, double-check every lid and label.
Separate Storage Saves Trouble
Too many labs stash chemicals side by side without reading up on which can set each other off. Acids, bases, and certain oxidizers never belong near 1,4-Dihydroxy-2-butyne. Chemical inventory systems exist for a reason; they help everyone avoid surprises. If the bottles start to sweat or show pressure on the cap, that’s a warning shot – time to clear out, ventilate the room, and get safety team eyes on it. Quick reactions save property and people alike. Fire-resistant cabinets don’t just tick a box on inspections, they honestly reduce real-world risks. Following strict separation rules cuts down on accidental mixing, especially for folks new on the job or moving between projects.
Training Cuts Down on Incidents
Not everyone in a lab or storage facility has years under their belt. Refreshers on chemical compatibility and emergency steps save headaches. Bring in hands-on drills using the right protective gear—goggles, gloves, and full coverage clothing. Those moments stick in people’s minds far better than dry slideshows. For cleanup, sorbent pads keep accidental drips in check; waste containers marked “hazardous” speed up safe disposal. Picking up a habit of recording every movement of high-risk chemicals creates a culture where everyone watches out for the next person.
Commitment, Not Shortcuts
Safe handling shows up as routines built day by day. In every lab I’ve worked, the best results come from stubborn attention to detail. Use glass containers, control temperature, crack open the binder before introducing unfamiliar chemicals, and treat every bottle like it isn’t your own. If something feels off, call for backup. Good habits become the backbone of safety—not just for your own desk, but for the crew that picks up where you left off. That’s how you stop preventable accidents before they start.
Where can I purchase 1,4-Dihydroxy-2-Butyne and what is its typical purity?
Pursuing Specialty Chemicals — The Search Isn't Always Simple
Anyone who’s tried to buy 1,4-Dihydroxy-2-Butyne knows specialty chemicals don’t just land in your lap. This molecule, often used in organic synthesis, cross-coupling, or the electronics field, shows up more rarely in general catalogs than common lab reagents. Most chemists have to reach out to established suppliers like Sigma-Aldrich, TCI America, or ChemShuttle. Price varies based on quantity, grade, and your location. Not every supplier will ship internationally, and some may want written assurance of your intended use if the compound falls under regulatory flags.
Purity: Why Clean Stock Matters More Than Ever
Labs should carefully check the purity listed for 1,4-Dihydroxy-2-Butyne, since reactions can nose-dive if it doesn’t meet published specifications. Most catalog suppliers offer this compound at 95% or higher. Sigma-Aldrich, for example, lists their material at ≥98%. TCI America hovers close to 97%. Some custom synthesis groups or smaller shops may provide 90–95% if that's all you need. Yet, for reactions headed toward pharmaceuticals, advanced polymers, or microelectronics, chancier stocks can mess things up fast. Impurities sometimes block key catalyst sites or sneak through to final products with expensive consequences.
Why E-E-A-T Principles Matter for Buyers and Sellers
Practical folks with real lab or purchasing experience know that not all claims of “high purity” stand up when the flask hits the stirring plate. Reliable chemical companies must work transparently–publish safety data, show typical NMR or LC-MS evidence, and provide certificates of analysis on request. This trust comes from living up to a track record. In my own work running pilot organic synthesis campaigns, I learned the value of checking every COA and talking plainly with sales chemists. Bad batches cost projects weeks or send teams scrambling for purification tools. Some of the best advice came from colleagues who’d burned time or budget on cheap, inconsistent stock.
Online Marketplaces: Legit or Risky?
People turn to platforms like Alibaba or eBay for rare chemicals, but these sites bring real risk. There’s the issue of mislabeling, substitution, or underplaying hazard categories. Without a direct relationship or verifiable track record, small-lot buyers now see supply chain issues and scams. If you’re experimenting or scaling up, stick to suppliers who publish traceable data and have experienced tech support. Sometimes the premium pays off when your project depends on reliable yields and trusted MSDS documents.
Regulatory Watch: Staying Above Board
Countries watch over the movement of chemicals like 1,4-Dihydroxy-2-Butyne. Buyers face legitimate background screenings or end-use checks, since misuse could invite regulatory issues. Established suppliers guide customers through paperwork and storage advice—an invaluable help, especially for early-career researchers or companies without a full-time compliance officer.
Solutions: Vet, Verify, Communicate
Instead of casting a wide net, I recommend reaching out to trusted chemical distributors or speaking with technical contacts at established catalog companies. Request the most current COA, ask about impurities, and put your questions in writing to track the supplier’s answers. Peer advice in industry forums or from collaborators often highlights which vendors stay responsive and reliable over time. If price is a factor, negotiate for small-lot pricing based on your research budget, but walk away from vague purity claims.
Final Take: Trust Matters as Much as the Molecule
In buying 1,4-Dihydroxy-2-Butyne, nothing replaces due diligence or open dialogue with suppliers. Trustworthy partners keep projects running, and knowing how to vet chemicals and purity sets professionals apart in research and industry alike.
| Names | |
| Preferred IUPAC name | but-2-yne-1,4-diol |
| Other names |
1,4-Butynediol But-2-yne-1,4-diol 2-Butyne-1,4-diol Butynediol 1,4-Dihydroxybut-2-yne |
| Pronunciation | /ˈwʌn, fɔːr daɪˈhaɪ.drɒk.si tuː ˈbjuːtaɪn/ |
| Identifiers | |
| CAS Number | 110-65-6 |
| Beilstein Reference | 1209377 |
| ChEBI | CHEBI:51886 |
| ChEMBL | CHEMBL151838 |
| ChemSpider | 61906 |
| DrugBank | DB03766 |
| ECHA InfoCard | 03f872d1-287c-445e-8bfa-bd684c548d45 |
| EC Number | 205-653-7 |
| Gmelin Reference | 66804 |
| KEGG | C13583 |
| MeSH | D003994 |
| PubChem CID | 79080 |
| RTECS number | EK8225000 |
| UNII | L5I6X43H2P |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C4H6O2 |
| Molar mass | 86.09 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Odorless |
| Density | 1.133 g/mL at 25 °C (lit.) |
| Solubility in water | Soluble |
| log P | -0.34 |
| Vapor pressure | 1.74E-3 mmHg at 25 °C |
| Acidity (pKa) | pKa = 8.01 (for OH group) |
| Basicity (pKb) | 3.98 |
| Magnetic susceptibility (χ) | -37.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.483 |
| Viscosity | 3.87 cP (25°C) |
| Dipole moment | 2.4361 debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –162 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -983.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 1-2-2-W |
| Flash point | Flash point: 137 °C |
| Autoignition temperature | 130°C |
| Explosive limits | 1-6.3% |
| Lethal dose or concentration | LD50 (oral, rat): 20 mg/kg |
| LD50 (median dose) | LD50 (median dose): 40 mg/kg (intravenous, mouse) |
| NIOSH | DA0875000 |
| PEL (Permissible) | PEL (Permissible): Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
But-2-yne 1,4-Butynediol 2-Butyne-1,4-diol 1,4-Butanediol |

