Commentary: The Many Faces and Futures of 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene
Historical Development
Chlorinated hydrocarbons capture a complex history tied deeply to mid-century efforts to boost agricultural productivity and manage pests. 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-tetrahydro-4,7-methanoindene, a mouthful by any standard, surfaced through the drive to maximize crop yields with relentless efficiency. A substance born in an era where environmental concerns routinely took a backseat to chemical innovation now stands at a crossroads, facing scrutiny for the baggage it carries: persistence, toxicity, and regulatory pressure. Exploring its legacy offers insight into how well-meaning science sometimes writes checks that society struggles to cash generations later. The demand for cheap, potent, long-lasting pesticides set the stage. Manufacturers, prioritizing field results over downstream effects, poured these molecules across fields and into water supplies. Tracing this trajectory highlights more than product evolution—it exposes shifts in scientific responsibility and public perception. People once hailed organochlorines for their miracles. Today, the same families of chemicals feature prominently on lists of environmental concerns and are often the subject of heated debate at scientific conferences and around regulatory tables.
Product Overview and Synonyms
In labs and among regulatory bodies, this compound typically goes by its systematic name, but workers and suppliers might call it by synonyms like chlordene derivative or heptachloro norbornene. While technical literature often wraps its true identity in jargon and abbreviations, the core structure—seven chlorines studded onto an indene backbone—does much of the heavy lifting. If you’ve spent time with technical manuals, you see right away how consistently synonyms and abbreviations can trip up even experienced hands. Beyond names, the compound’s reputation rides less on clever chemistry than on its physical punch and environmental stubbornness.
Physical & Chemical Properties
Thanks to its structure, the molecule handles most storage and shipping conditions without flinching. High chlorine count delivers density, reduced solubility in water, and a pronounced tendency to hang around long after application. While this seems like a win for users counting on residual pest control, it also throws a wrench into ecosystem restoration efforts. I remember my first real encounter with persistent organochlorines. The expectation was that rain and sunlight would eventually wash away or degrade what we put into the fields—nature’s cleanup crew. This stuff proves far more stubborn. Its low volatility and high soil binding keep it in place, creating problems for farmers, water managers, and neighbors downwind alike.
Technical Specifications & Labeling
Strict rules cover the labeling and concentration limits for 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-tetrahydro-4,7-methanoindene, especially in countries that track persistent organic pollutants. Regulations reflect mounting pressure from environmental science and health advocacy. Labels make explicit reference to acute and chronic hazards, warning of the need to shield applications from waterways, animals, and people. From my own time working on extension programs, clarity in labeling can make or break safe practice. Confusion or neglect in this sphere leads to more than regulatory fines—it erodes trust between industry, regulators, and the public. A robust technical label serves as both legal shield and practical guide, parts of a contract between user and maker.
Preparation Method
Unlike green chemistry innovations that embrace renewable feedstocks and minimal by-products, the synthesis of these halogenated indenes is a holdover from less restrictive times. Industry relies on sequential chlorination of hydrocarbon precursors under controlled, often aggressive, conditions. For anyone raised in the safety-aware generations of chemists, these methods raise eyebrows—not just for their inherent risks, but for the volumes of hazardous waste and emissions they generate. Synthesis, purification, and packaging demand tightly sealed environments and an experienced workforce. My time in chemical plants revealed the strain on teams who balance productivity with strict waste handling and accident prevention.
Chemical Reactions & Modifications
Since the core indene skeleton is heavily chlorinated, options for chemical modification are limited. Most efforts focus on slight tweaks to improve formulation stability or reduce volatility. The chemical’s resistance to breakdown—one reason for its original commercial draw—now stands as its major liability. Incineration at specialized facilities can dismantle the molecule, but partial combustion risks forming even more problematic byproducts. Discussions with remediation professionals often circle back to the limited toolbox available for safe disposal or degradation, a testament to the need for smarter molecular design from the start.
Safety & Operational Standards
Conversations about operational safety start with the basics: gloves, goggles, protective clothing, and enough ventilation to clear any airborne residues. Beyond equipment, culture and training make the real difference. One misstep or corner cut, and the risks extend to entire communities, especially where outdated equipment or poor oversight dominate. I’ve seen firsthand how committed teams can drive down exposure incidents to near zero, but it takes ongoing investment and vigilance. Regulators increasingly pressure companies to track, record, and publicly release accident rates, exposure levels, and remediation efforts.
Application Area
Originally, the wide-ranging insecticidal power pulled this compound into agriculture, pest control, and even structural protection for timber. The same stubbornness that delighted users also cues headaches for neighbors and future property owners. Long soil half-lives limit its continued use, with many growers favoring newer chemistries that break down more quickly and leave less trace. Attempts to rehabilitate contaminated sites remain costly and technically challenging, creating a tension between past benefits and future liabilities. In urban settings, even trace residues can spark debates over land reuse and redevelopment.
Research & Development
Once a star in the pesticide research pipeline, the compound now attracts scientific attention for its environmental footprint, transport, and breakdown. Modern work focuses on soil and water remediation, analytical detection at lower and lower limits, and improved risk assessment models. I sat in seminars where research teams outlined new microbial treatments or catalytic destruction pathways, and the sense of urgency felt real. Science now measures progress not by sales volume or persistence, but by detection avoidance, risk minimization, and successful cleanup.
Toxicity Research
No modern commentary can skip past the health side. Evidence from animal and epidemiological studies links exposure to disruptions in neurological development, hormonal imbalance, and increased risk of cancer. The chemical’s tendency to bioaccumulate—gathering in fatty tissues, working up food chains—complicates the story for both consumers and wildlife. Past experience shows that delays in coupling usage restriction to these findings can leave long shadows on community health. What emerges from hundreds of published studies and field reports is clear: oversight needs to stay in front, not behind, our understanding of risk.
Future Prospects
With mounting restrictions and intense scrutiny, the market for 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-tetrahydro-4,7-methanoindene continues to shrink outside heavily regulated legacy uses. Inventing cleaner synthesis and greener alternatives stands as the most promising path forward. Incentives and support for innovation—both at the university and company level—can speed the arrival of tools that balance effectiveness with low persistence. Experience suggests integrated pest management, biocontrol agents, and rapidly degradable compounds promise better harmony between productivity and sustainability. For regulators, focusing on legacy site cleanup and exposure monitoring remains key. Honest communication, grounded in what we know and what we still need to learn, underpins any responsible path forward.
What is 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene used for?
What’s in a Name?
That name is a mouthful, but dig a little deeper and it gets even more tangled. This chemical actually goes by another more common name—chlordane. People working on their lawns and gardens, and folks raised on farms, probably remember it from decades ago as that strong-smelling bug killer. Chlordane once held a spot as a big-league pesticide, showing up everywhere from cornfields to suburban crawlspaces in the hopes of knocking out termites, ants, and pretty much anything else that might munch on wood or crops.
Why Did People Use It?
Farmers turned to chlordane to keep crops protected from rootworms and other pests ready to devour their hard work. Homeowners hired pest control crews who sprayed it in the soil around new houses, chasing out termites or trying to keep them out in the first place. In a world where people want picturesque front yards and full pantry shelves, you can understand the appeal. Chlordane seemed to last a long time in soil. It tackled a broad range of insect problems. Efficiency sold it. Companies produced and sold a ton of this stuff right up through the late ‘80s in the United States.
Big Problems Nobody Saw Coming
Sometimes a solution spawns an even bigger headache. That’s the story with chlordane. This chemical refuses to break down in dirt and water. Scientists found it building up in riverbeds and even in the bodies of fish and birds miles from the original spray site. Chlordane ended up in human bodies, too. Tests showed it in milk and blood. After several years, research linked chlordane exposure to everything from breast cancer to nervous system disorders to liver trouble. Kids, especially, felt the risks since their smaller bodies can’t handle the toxic hits as well.
No Free Passes for Toxic Legacy
Governments finally caught on. By 1988, the U.S. Environmental Protection Agency stopped the sale of almost all chlordane products. People working in pest control didn't have much choice but to look for safer tools. While several countries took similar action, chlordane stuck around in places with patchier regulations, and leftovers from the old days still rest in contaminated soil or crawlspaces.
What’s Actually Happening Today?
Most manufacturers no longer make or sell chlordane for regular use. Its legacy hangs around in research labs where scientists piece together toxicology puzzles or monitor wildlife for persistent chemicals. Environmental engineers get called out to clean up old dumping grounds and home sites where chlordane residues linger, especially near creeks or in neighborhoods built before the bans. A handful of highly specialized industrial processes might use it, but for everyday life, its story already hit the last page.
Facing the Fallout
The mess chlordane left behind offers more than a science lesson. It reminds anyone working with chemicals to test and ask hard questions before using new compounds on homes, farms, or public spaces. Routine soil and water testing—the kind environmental watchdogs push—helps flag old contamination. Education and transparent records give families and farmers information before they dig, plant, or buy land. Solutions aren’t perfect, but learning from the chlordane story just might keep future generations from repeating it.
Is 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene hazardous to health?
Getting a Handle on Toxic Chemicals in Daily Life
Most folks never knowingly encounter the long, tangled name of 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene, but chemical hazards often hide in plain sight. This stuff belongs to a notorious family called organochlorines. These compounds pop up in old pesticides, and once sprayed on crops or lawns, they linger for years—sometimes for generations. Think about how rain washes soil, how wind picks up particles; the journey of such chemicals doesn't end at the field’s edge. Years ago, I watched a neighbor’s concerns grow after learning that a pesticide used decades before still clung to their backyard’s dirt and made its way into the local water supply. Concerns like those aren’t just paranoia—they’re based on research connecting these compounds with big health problems.
What Research Says About Risks
Scientists don’t pull warnings out of thin air. Some organochlorines have triggered cancer in animals and stoked worries about reproduction in humans. For this specific compound, lab studies highlight how it sticks around in fatty tissues. In real life, that means animals, and possibly people, can store low doses from food, water, or even dust. Bioaccumulation leads to bigger risks higher up the food chain. Kids playing on the grass and gardeners getting soil on their hands run a higher chance of exposure compared to someone just walking by a treated field. Even small exposures—think a sip from contaminated water or breathing a puff of dirt on a windy day—can add up over months or years. The World Health Organization and the Environmental Protection Agency treat related chemicals with suspicion for a reason.
Real-World Health Concerns
Stories float around in farming families and communities near industrial sites. People mention trouble with learning and memory, mysterious headaches, or issues with fertility. The medical research backs them up in many cases. A study published by the National Institutes of Health tracked a rise in certain cancers and neurological problems in places with heavy pesticide use. More than once, I've seen farmers get worried after a higher cancer risk made local headlines. They ask how to protect their land and their kids, wondering if keeping old sheds locked is enough. Truth is, keeping an eye on air, water, and soil quality offers the closest thing to genuine peace of mind.
Routes to Safer Communities
Avoiding risky chemicals starts with full transparency from manufacturers and regulators. People deserve to know if old stockpiles of pesticides sit near their schools, gardens, or water sources. Clean-up work takes money, time, and honest measurement. Simply covering up contaminated soil won’t cut it. I remember joining a community push for better testing after some kids got sick at a local park. Pressure from parents and teachers worked—soil got tested, and cleanup began. Choices matter at home, too. Using gloves in the garden, washing produce well, and supporting organic growers add up to fewer pesticides in the body. On a larger scale, research into safe alternatives pushes the industry toward smarter and cleaner farming methods, replacing stubborn toxins for good. The path to safety isn’t simple, but experience shows ordinary people can light a fire under slow-moving systems. That’s how real change takes hold—and why talking openly about these hazards never feels optional.
What are the safety precautions when handling this compound?
Why Handling Chemicals Deserves Respect
Chemicals spark breakthroughs and shape our daily world, but there’s no getting around it: the risks are real. Even a moment’s lapse in focus can turn a routine day sideways. Years in the lab and shop have taught me that respect for chemicals isn’t just about following rules—it’s about people’s lives and health. From harsh acids to organic solvents, I’ve seen gloves eat away, fumes sting the lungs, and more than one person regret a shortcut. Safety precautions aren’t red tape; they’re essential habits that keep accidents from getting personal.
Understand Before You Touch
Most trouble starts when people skip reading safety data sheets (SDS). The SDS lays out what matters: toxicity, reactivity, flammability, storage needs, and first aid if things go wrong. It’s tempting to rush, especially if you’ve handled something similar, but chemicals can surprise you. Even familiar names sometimes hide new hazards, especially in new forms or concentrations. Since people can react differently, knowing your own sensitivities helps too—for instance, I once learned the hard way that a low level of formaldehyde is enough to trigger asthma symptoms.
Personal Protection Isn’t Optional
Forget fashion—make personal protective equipment (PPE) your closest ally. Eye protection, lab coats, and gloves will stop most injuries before they start. For nastier fumes, a proper respirator beats a cheap mask every time. Watch out for glove-breakers like phenol or methyl ethyl ketone; make sure the material matches the job. In my experience, chemical-resistant aprons can save more than a shirt during splashy tasks. Shoes matter, too—open-toed sandals belong nowhere near corrosives or caustics.
Keep Workspaces Ready for Surprises
I’ve walked into labs cluttered with books and coffee alongside beakers. It’s a recipe for spills, contamination, and headaches. A tidy, organized bench says a lot about how seriously someone takes safety. Fume hoods exist to capture vapors and keep inhalation risks down. If something smells funny outside the hood, trust your nose and pull back. Never eat, drink, or even apply lip balm in work areas—a forgotten snack easily turns into an exposure route. I recall a technician who ignored this, and an accidental hand-to-mouth move forced a hospital visit.
Storage: Where Mistakes Like to Lurk
The right bottle matters as much as what’s inside. Acids and bases go separate ways, and oxidizers keep far from organics. Labels should be crystal clear, including hazards and dates: in my early days, I mixed up two similar-looking bottles and learned why color coding isn’t just for show. Having fire extinguishers and spill kits within reach can turn a potential disaster into a brief clean-up. Inventory checks prevent chemicals from hanging around far past their prime, where they might degrade or even explode.
Don’t Work Alone—And Speak Up
Solo work feels efficient, but it leaves no backup if something goes wrong. Even after hundreds of safe sessions, the unexpected happens. Teaching newcomers matters: don’t let inexperience morph into risk for everyone. If someone flags an unsafe practice, try listening. Years of safety talks taught me that almost every incident comes with a warning sign beforehand.
Continuous Training Makes a Difference
Regulations and best practices evolve. What worked years ago could miss a hazard discovered last month. I stay updated with refresher courses, and I push colleagues to do the same. Learning from past incidents—not just your own, but industry-wide—means fewer stories that end in regret.
Better Safety, Stronger Outcomes
Safety isn’t just about fear—it's about care. When people work from accurate information, develop steady habits, and foster a culture of honesty around hazards, chemicals become powerful tools, not threats. From leadership to lab techs, everyone shares the responsibility, because every accident avoided means more time and talent for discovery.
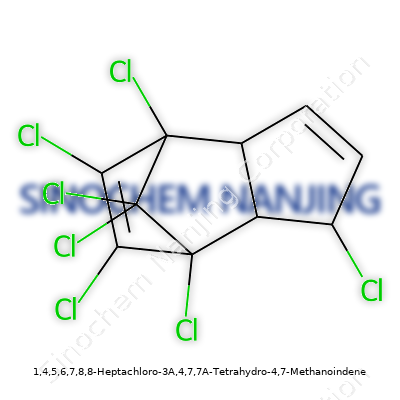
What is the chemical structure of 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene?
What’s Hiding in a Name
Peeling apart the name 1,4,5,6,7,8,8-heptachloro-3A,4,7,7A-tetrahydro-4,7-methanoindene takes a bit of patience and a willingness to look closer. This chemical sits at the center of the controversy and conversation around a notorious pesticide, chlordane. Mapping out its structure isn’t just a chemistry exercise; it shows the reason for its toxicity, tenacity, and the ripple effect on soil and water.
Structure Shapes Persistence
This molecule spins out from the basic frame of indene, a fused ring with both a benzene and a cyclopentene portion. Imagine that core, then patch on a methano bridge that binds carbons four and seven, turning the structure into a rigid, almost cage-like form. Now, that’s just the bones of it. Seven chlorine atoms drape onto specific carbons across this structure, replacing hydrogen atoms that might otherwise make it more biodegradable.
Chemically, every chlorine glued to the ring protects it from breaking down. Chlorines on adjacent carbons crowd together, raise its stability and make the molecule resistant to attack by microbes or sunlight. You can bet this layout fuels its tendency to stick around in soil and fat, more than just about any regular hydrocarbon cousin. That structure explains why decades after application, residues of this compound linger in ground samples and in local wildlife.
History Carved in Carbon and Chlorine
Chlordane, with this structure as a building block, hit the market as a multifaceted pest killer in the 1940s and 1950s. Farmers and pest control companies saw results right away—no more termites, no more crop destroyers. On paper, it looked ideal: bugs dropped, crops survived, and it didn’t easily wash away with the next rain. With insight from modern toxicology, that indestructible design shows a downside. Its hydrophobic tail means it clings to fatty tissues in animals and works through the food chain. The EPA eventually pulled the plug in the United States in 1988 after evidence of both environmental and health risks piled up.
Environmental and Health Impact
Unlike quick-fading weed killers or household bleaches, 1,4,5,6,7,8,8-heptachloro-3A,4,7,7A-tetrahydro-4,7-methanoindene’s structure makes it stick in soils, streambeds, and even in fish and mammals. The tightly packed, chlorine-heavy rings don’t give enzymes an easy entry point. Studies show this compound disrupts the nervous system, builds up as a persistent organic pollutant (POP), and can cross the placenta in mammals. Over time, those who live in areas with heavy past use have seen damage to both local ecosystems and sometimes to human health as well.
Paths Forward: Clean Up and Prevention
Addressing this legacy chemical calls for focused approaches. Research on bioremediation turns toward specialized bacteria and fungi that can edge into breaking down these stubborn rings. Moving away from persistent pollutants in pesticide development marks another step. Better monitoring and tighter regulations on imported produce or legacy-contaminated soils help limit ongoing exposure. Sharing knowledge about these molecules’ unique architecture can drive smarter choices, not just for chemists but also for public health, law, and farming.
The shape of this molecule doesn’t change, but our response to it can. Clearheaded study and action—applied at every step from the lab to the field—hold the key.
Where can I purchase 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene?
What Stands Behind the Name Heptachlor?
Talking about 1,4,5,6,7,8,8-Heptachloro-3A,4,7,7A-Tetrahydro-4,7-Methanoindene hits a nerve among anyone who spends time reading about environmental health and agricultural practices. Known best as heptachlor, this chlorinated cyclodiene has left its mark on farms and in environmental policy. Chemists, toxicologists, and environmental watchdogs have wrestled with its legacy for decades. The question about purchasing it is not about routine chemical supplies — it’s about understanding the landscape shaped by regulatory policies set in place because of legitimate safety fears.
The Reason Behind the Tight Controls
Heptachlor played a big role as an insecticide in mid-twentieth-century farming. Watching its effectiveness at killing pests on crops, many growers welcomed it for making food production look easy. With time, science caught up. Studies revealed a dark side: heptachlor’s persistent behavior in soil and water, long breakdown time, and links to health problems including cancer, liver damage, and developmental issues. In the food web, residues trickle into milk and meat, affecting folks who might never have touched a pesticide container in their lives.
Regulators couldn’t ignore the mounting evidence. Agencies like the U.S. Environmental Protection Agency and equivalent bodies worldwide classified heptachlor as a persistent organic pollutant (POP), squarely placing it on the list of chemicals that needed phasing out. Sales for agricultural and household use stopped, and strict controls followed for storage, handling, and disposal.
Looking for Heptachlor? You’ll Hit Regulatory Walls
Ask experienced lab techs or even the most connected chemical suppliers about purchasing heptachlor, answers always include the same themes: legal restrictions, tracking, and the reason for the demand. Inventories only exist for research and monitoring, almost always tethered to government labs or highly regulated academic studies. Commercial chemical marketplaces don’t openly list it. Reputable suppliers keep rigorous vetting in place, checking credentials and the end-use before a single ounce gets shipped.
Why These Barriers Matter
These roadblocks serve a real purpose. Chemical safety isn’t just a matter for regulators; it’s about neighbors, animals, children, and the ecosystem. Nobody wants pesticides building up in rivers, soil, or dinner plates. Unsafe use or release could mean permanent harm that money can’t fix. These hard lessons stem from disasters in the past, where weak oversight led to long-term community exposure. So, for anyone looking to get their hands on heptachlor, it’s important to reflect on intent, responsibility, and compliance.
How Research Can Move Forward Responsibly
Curiosity about how chemicals interact with the environment or human bodies doesn’t vanish after bans go into effect. Science sometimes requires careful review of legacy pollutants. That means applications for legal access still exist, but they run through institutional oversight, ethical review boards, and government permits. Reputable scientists submit proposals, describe methods, and agree to secure handling. Only then does a trusted chemical supplier proceed — typically under contract and traceable delivery.
Safer Alternatives and the Push for Innovation
Rather than revisiting old dangers, many researchers and businesses invest in safer substitutes. Integrated pest management approaches, selective biological products, and targeted synthetic chemicals now fill roles heptachlor once held. Crop scientists and environmental cleanup teams keep exploring green chemistry for new tools, sidestepping risks locked into persistent chemicals.
Bottom Line
Heptachlor isn’t something you can buy easily or freely. Regulations reflect the collective lessons learned from real-world harm. If acquisition is ever needed, it demands oversight, transparency, and purpose that extends far beyond commercial convenience.
| Names | |
| Preferred IUPAC name | 1,4,5,6,7,8,8-heptachloro-4,7-methano-3a,4,7,7a-tetrahydroindene |
| Other names |
Aldrin HHDN 1,2,3,4,10,10-Hexachloro-1,4,4a,5,8,8a-hexahydro-1,4-endo,exo-5,8-dimethanonaphthalene Isodrin |
| Pronunciation | /ˌhɛp.təˈklɔːr.oʊ ˌθriˈeɪ ˌtɛtrəˈhaɪdroʊ ˌmɛθ.ə.noʊ.ɪnˈdiːn/ |
| Identifiers | |
| CAS Number | 1024-57-3 |
| Beilstein Reference | 3118737 |
| ChEBI | CHEBI:34629 |
| ChEMBL | CHEMBL601485 |
| ChemSpider | 157357 |
| DrugBank | DB11123 |
| ECHA InfoCard | 03fa0c825f9d-42e7-4c7b-9b18-346130a1034e |
| EC Number | 212-486-9 |
| Gmelin Reference | 82577 |
| KEGG | C11214 |
| MeSH | DDT |
| PubChem CID | 9586786 |
| RTECS number | GV9810000 |
| UNII | Q7V7WQ03D1 |
| UN number | UN2761 |
| CompTox Dashboard (EPA) | DTXSID1020099 |
| Properties | |
| Chemical formula | C10H3Cl7 |
| Molar mass | 406.326 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.89 g/cm³ |
| Solubility in water | insoluble |
| log P | 4.88 |
| Vapor pressure | 1.67E-5 mmHg at 25°C |
| Acidity (pKa) | 4.60 |
| Magnetic susceptibility (χ) | -79.5·10^-6 cm³/mol |
| Refractive index (nD) | 1.5990 |
| Viscosity | 5000 cP (25°C) |
| Dipole moment | 2.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 377.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3352 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Pesticides |
| Hazards | |
| Main hazards | Harmful if swallowed. May cause damage to organs through prolonged or repeated exposure. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P308+P313, P312, P314, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-4-0 |
| Flash point | Flash point: 200°C |
| Autoignition temperature | Autoignition temperature: 430 °C |
| Lethal dose or concentration | LD50 oral rat 38 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 947 mg/kg |
| NIOSH | NA0150000 |
| PEL (Permissible) | 0.5 mg/m3 |
| REL (Recommended) | 0.5 mg/m³ |
| Related compounds | |
| Related compounds |
Cis-chlordane Trans-chlordane Heptachlor Aldrin Dieldrin |

