1,3-Dinitrobenzene: The History, Challenges, and Opportunities Behind a Niche Compound
A Glance at the Historical Pathway of 1,3-Dinitrobenzene
Getting to know 1,3-Dinitrobenzene means looking back at the roots of modern organic chemistry. This compound has woven itself into scientific research since the late nineteenth century, evolving alongside industry’s deeper understanding of aromatic chemistry. Early chemists were fascinated by the reactivity of benzene rings, especially once they saw how strong acid mixtures could pack not just one, but two nitro groups onto a single benzene ring. Industrial-scale nitration grew from research like this. By the dawn of the twentieth century, 1,3-Dinitrobenzene had slipped into labs, finding roles in explosives research, dyes, and chemical analysis. Many researchers, including myself, first learned of its utility from classic organic synthesis textbooks, not glossy brochures, hinting at its presence in foundational chemical recipes.
Physical and Chemical Realities
Holding a vial of 1,3-Dinitrobenzene, you notice its solid, yellowish crystalline appearance. There’s a sharp, pungent odor, unforgettable and unmistakable, that reminds me of high school chemistry stores. Its melting point sits well below 100°C, and chunky crystals break easily as they tumble from the bottle. Water never dissolves it well; instead, organic solvents like acetone or ethanol offer better luck. The molecule itself looks somewhat symmetrical, which impacts how it reacts and survives in the environment. Compared to some of its cousins, like 1,2- or 1,4-dinitrobenzene, the meta orientation shapes both its chemistry and its hazards. It won’t ignite as unpredictably as trinitrobenzene or TNT, but it still carries a risk that pushes folks to handle it with gloves and goggles, even outside of formal lab settings.
Technical Touchpoints and Labeling
The technical details draw lines that determine how labs and factories use 1,3-Dinitrobenzene. Most containers highlight its purity level, often above 98 percent for analytical work. Long chemical names, like m-Dinitrobenzene, share space with standard hazard symbols, typically the GHS toxic mark. In my time teaching undergrad labs, visible, detailed labels made all the difference in avoiding confusion, especially with other nitroaromatics floating nearby. Honest, upfront technical information can prevent unwanted accidents and guide safe storage, like keeping Dinitrobenzene away from strong reducing agents or open flames.
Production: More Sweat Than Glamour
Scaling up production of 1,3-Dinitrobenzene takes more grit than glamour. The basic method relies on carefully controlled nitration of benzene, using nitric and sulfuric acids as the main workhorses. Adjusting acid ratios and temperatures controls whether the process adds one or two nitro groups, not more, not less. In a teaching lab, small-scale batch runs are noisy, smelly, and a chore to clean up afterward. Large plants invest in elaborate cooling, venting, and acid-recycling systems. High yields and consistent purity never come by accident; they come from trial and error, constant monitoring, and sometimes, a bit of singed clothing.
Chemical Reactions and Modifications
Working with 1,3-Dinitrobenzene challenges both beginners and seasoned chemists. Its two nitro groups make it both demanding and rewarding. Reducing conditions shift it toward diamines, while nucleophilic attack can swap out nitro groups or add substituents to open new possibilities. In some projects, I used 1,3-Dinitrobenzene as a stepping stone to build more complex pharmaceuticals or polymers, watching small structural changes create huge effects further down the line. Reactivity comes at a price. Mishandling strong acids and bases during modification can spell trouble quickly, often telegraphed by a sudden change in color or fumes that sting the eyes.
Behind the Names: Synonyms and Naming Quirks
Confusion sneaks in through the doors of naming. Over the years, textbooks and suppliers alike have labeled 1,3-Dinitrobenzene in a handful of ways. “m-Dinitrobenzene” tips its hat to the meta orientation, while older treatises sometimes stick to Roman numerals or more technical descriptors. For anyone on a research team, checking the literature twice helps dodge any mix-up with 1,2- or 1,4-isomers—a mistake more common than many admit. The names may sound fussy, but they separate safe, informed experimentation from accidents that can ruin a week’s work.
Workplace Safety and Running a Tight Ship
No one handles 1,3-Dinitrobenzene without a healthy dose of respect. Nearly every manual, and my own memory of lab mishaps, stresses tight protocols. Gloves and goggles are non-negotiable. Well-ventilated hoods and chemical-resistant containers keep inhalation and skin contact at bay. Spill response must be quick and decisive—witnessing a careless colleague knock over a bottle and scramble to contain the mess left an impression I won’t forget. Treat every handling session as a fresh chance for error, and double-check containers for leaks or cracks, because toxic exposure disrupts more than just lab schedules.
Where It Ends Up: Real-World Applications
Despite living in the shadow of more famous nitro compounds, 1,3-Dinitrobenzene carves out a steady market. Its largest roles pop up in explosives research, though not frontline use, but rather as a benchmark or intermediate for synthesizing more potent stuff. In the dye industry, it has triggered the creation of deep, vivid pigments that colored everything from textiles to printer inks. Occasionally, academic teams rely on its well-understood reactivity for probing reaction mechanisms or calibrating instruments. In my early career, watching simple reactions with this compound formed a rite of passage, but seeing it at work in dye vats or explosives labs brought out a new level of respect for bench-scale chemistry shaping entire industries.
Research and Development: Still Churning
Even as newer aromatic compounds crowd the shelves, academics and industrial chemists squeeze more knowledge out of 1,3-Dinitrobenzene’s core structure. Every couple of years, someone unveils a more efficient synthetic route, perhaps cutting down acid waste or bumping up yields. Environmental chemists push for cleaner, closed-loop methods, while analytical chemists use its stability and reactivity to refine sensors or monitor environmental contamination. Over the years, I’ve seen funded grants drift toward greener processes or medical derivatization, hinting that under-the-radar compounds still offer fresh ground for development.
Toxicity Research: The Less Glamorous Side
Few topics in chemical research test patience and resources like toxicity studies. 1,3-Dinitrobenzene holds a spot on regulatory lists due to its effects on humans and wildlife. Exposure can target the nervous system, red blood cells, and reproductive health. I spent months wading through old animal studies showing dose-dependent toxicity and chasing fresh studies on environmental persistence. Today, research teams lean on LC-MS techniques, in vitro models, and even genomics to unravel exposure pathways. Public safety remains a top concern, urging researchers to balance productive applications with transparent, data-driven risk assessment.
Where 1,3-Dinitrobenzene Might Head Next
The future for 1,3-Dinitrobenzene dodges easy predictions. With increasing pressure for green chemistry, sustainable manufacturing, and non-toxic alternatives, its traditional market may shrink. Ongoing exploration of biodegradable dyes and safer intermediates trims demand, but core research keeps 1,3-Dinitrobenzene in circulation. Regulatory tightening, especially around worker safety and community health, shapes operational standards more than ever. I expect up-and-coming chemists to inherit a landscape dotted with stricter rules, but also smarter sensors and better containment systems. Its story, from nineteenth-century curiosity to modern workhorse, reminds us that progress in chemistry comes balanced on the edge of utility, safety, and human responsibility.
What is 1,3-Dinitrobenzene used for?
More Than Just a Chemical Compound
Walk into any factory relying on industrial chemicals, and the odds are good you’ll find 1,3-dinitrobenzene on a spreadsheet or a shelf. Known to chemists as m-dinitrobenzene, this yellow crystalline solid carries a reputation built on its role in making other things, not on the end of a production line. I’ve come across it most often while talking to folks working in explosives, dyes, and even pharmaceutical research labs. Its uses paint a picture of both industry demand and the push for safety in handling potentially hazardous materials.
Building Blocks for Explosives and Polymers
1,3-dinitrobenzene is no stranger to energetic materials. Workers in explosives manufacturing use it as an intermediate when making more complex chemical agents, including trinitrotoluene (TNT). The chemical structure allows for a predictable reactivity, which is vital in controlling the delicate steps needed for safer, more reliable explosives. Years ago, I toured a facility centered on blasting agents; the staff discussed the importance of carefully managing temperature and ventilation, because even slight negligence with such compounds can spell trouble.
Industrial chemists also rely on 1,3-dinitrobenzene to create other nitroaromatic compounds and plastics. In particular, it serves as a starting point to produce m-phenylenediamine, a molecule woven into polymers and aramid fibers. Think bulletproof vests—aramid is at the core of Kevlar’s strength. The route from dinitrobenzene to these durable fibers is a showcase of chemistry turning basic materials into life-saving gear.
Connection to Dyes and Pharmaceuticals
Many of the bright, stable dyes in textiles or inks trace their origins back to dinitrobenzene derivatives. Color chemists mention needing compounds that will anchor pigment to fabric or paper and resist fading. m-Dinitrobenzene, with its nitro groups in symmetrical positions, can be chemically manipulated to create complex dye molecules.
While not as common, some pharmaceutical projects use 1,3-dinitrobenzene for synthesizing specialty molecules. New drugs often require multiple steps, and intermediates like this one give researchers flexibility. I've spoken to synthetic chemists who stress that choosing the right intermediate can mean less waste, more efficiency, and improved safety in research environments.
Health Concerns and Safer Practices
Anyone spending time around chemicals knows the mantra—safety first. There’s no sugarcoating it: 1,3-dinitrobenzene is toxic. Inhalation or skin contact can cause serious health issues, including effects on the blood and nervous system. Factory workers, lab techs, and even shipping handlers get regular training on how to store, transfer, and dispose of this compound.
Strict regulations, including acceptable exposure limits and environmental controls, shape how industries operate. Following OSHA and EPA rules protects workers and limits pollution. Improvements in containment, personal protective equipment, and routine health monitoring build a culture of respect for what these chemicals can do—for good and for harm.
Rethinking Chemical Processes
Many companies now invest in greener synthesis techniques. One research group I met highlighted progress in reducing hazardous waste and using less water during production. They explained that sustainable practices not only meet regulatory standards but often save money and spark creativity within R&D teams.
1,3-dinitrobenzene remains a workhorse of chemical manufacturing, but its risks prompt constant vigilance. Every time I meet an old colleague in the industry, the conversation returns to a single thread: how to harness its utility, protect workers, and move towards safer, smarter chemical processes.
What are the safety precautions when handling 1,3-Dinitrobenzene?
Understanding the Risks
Walking into a lab or factory where 1,3-Dinitrobenzene waits on the shelf means facing real risks. Even people with chemistry experience sometimes overlook how nasty this yellow, crystalline substance can get. Touching it with bare hands can lead to skin rashes. Breathing it in or swallowing a tiny amount introduces risks far greater than many would guess—nervous system damage, anemia, and effects as serious as permanent hearing loss or blue fingers from a lack of oxygen. The chemical doesn’t play favorites, either. Repeated exposure can catch even the most seasoned technician off guard.
Common-Sense Preparation
Nobody should think rubber gloves alone offer magical protection. Splash-proof goggles, nitrile gloves, a fitted lab coat, and—most importantly—a tight-sealing respirator stand between you and the hospital. Cheap cotton won’t stop the substance from soaking through. It can be tempting to skip a step when you just need to weigh out a few grams, but no job is routine enough to make chemical burns worth the shortcut. Personally, I learned to triple-check my sleeves and neck protection after seeing a seasoned chemist deal with nerve tremors for months after a small spill.
Ventilation and Spills: Sweat the Small Stuff
Fume hoods save more than just headaches. Just turning on the fan isn’t enough: keep all work with 1,3-Dinitrobenzene inside the hood, not at the edge. Airflow matters because this chemical lingers in dust form, waiting to stick to unsuspecting skin. Any powder on benches or in the wrong trash can shifts the risk from you to the next person. So spill kits should always be within reach. Never sweep this powder dry. Use damp towels or specialized absorbents, throwing them away in clearly marked hazardous waste containers every single time.
Hygiene and Storage Habits
Every expert agrees: eating or drinking near 1,3-Dinitrobenzene is asking for trouble. Even if snack breaks feel harmless, dust on fingers or sleeves can find its way to your mouth. Wash hands with real soap. Skip the waterless sanitizer, since it doesn’t break down tough chemicals. If any chemical lands on skin or clothes, shower with cool water, and don’t tough it out hoping irritation will fade.
Storing this compound calls for locked cabinets, clear hazard labels, and distance from open flames. It decomposes and gives off toxic fumes when heated above 250°C. Keep it below that and far from oxidizers or strong bases—common sense, but details slip when supplies pile up.
Long-Term Solutions: Training and Attitude
Experience helps, but only so far. Everyone from students to supervisors needs ongoing, real-world training. Practicing emergency cleanups using mock spills builds that muscle memory I’ve relied on more than once. Signs should sit at eye level, not tucked near the floor. Companies owe their workers regular refresher courses. If someone’s unsure about a safety step, they should speak up, no matter the pressure to finish a task fast. No degree or years on the job change the fact that safety gear only works if used every single time.
Making safety habits routine doesn’t come by accident. It takes leaders who don’t just post rules—they model them. If the newest intern feels safe asking questions or double-gloving, everyone benefits. Mistakes with 1,3-Dinitrobenzene hurt more than just one person. Respect for this chemical starts with small details and grows through practice.
What is the molecular formula of 1,3-Dinitrobenzene?
The Backbone of Organic Chemistry
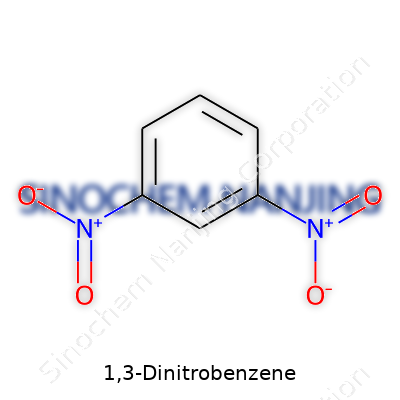
Chemistry had this way of grabbing my attention back in school with names that sounded more like puzzles than answers. 1,3-Dinitrobenzene falls right into that category—a name packed with meaning, not just for chemists, but for anyone interested in how small changes in structure can make big differences in use and safety. 1,3-Dinitrobenzene might not show up in everyday conversation, but it affects industries from dyes to munitions. Its molecular formula—C6H4N2O4—tells its story in shorthand.
What’s Behind Those Letters and Numbers?
Chemists learn the language of molecules early on, seeing that every carbon, hydrogen, nitrogen, and oxygen tells part of the tale. In 1,3-Dinitrobenzene, six carbons form a ring, a structure called benzene, with four hydrogens clinging to that backbone. Two nitro groups (NO2) take the places usually held by hydrogen—one sits across from the other on that ring, which is why we call it 1,3-. That structure—C6H4N2O4—points to its role in chemical processes that need stability, and sometimes, the power to release energy fast.
Beyond the Lab: Safety and Industry Uses
My first brush with the nitro group wasn’t in a textbook but in a discussion on explosives. That sticks—1,3-Dinitrobenzene plays a quiet role in manufacturing explosives and propellants, where chemical stability under normal conditions meets the potential for power under the right trigger. The same chemical pops up in making dyes, rubber chemicals, and pesticides. These uses need respect: handling dinitrobenzenes asks for careful work, because these substances aren’t people-friendly. Exposure can threaten the blood’s ability to carry oxygen, something most workers in related industries already know far too well.
The value of experience in the lab shows in how researchers and safety teams manage, store, and transport chemicals like 1,3-Dinitrobenzene. Wearing gloves and eye protection, working with proper ventilation, and following storage guidelines keeps workers from getting sick. For companies, thorough training makes the difference between safe industry and workplace injuries. Government agencies like OSHA set limits on exposure for a reason. Worker health draws attention, especially in chemical plants, and it's often the people closest to these materials who spot risks before they become headlines.
Safer Alternatives and Environmental Responsibility
Keeping everyone safe means more than rules in a binder. The search for safer replacements for hazardous chemicals has become part of the chemical industry’s future. Some companies push to move away from compounds like 1,3-Dinitrobenzene, especially if they can find less toxic ways to get the job done. There’s no simple solution, but momentum grows as green chemistry becomes more than a slogan. Choices in research steer how tomorrow’s factories and labs operate, and watching new options emerge shows progress rooted in both science and care for people and the environment.
Knowing the molecular formula—C6H4N2O4—goes beyond mere trivia. It opens conversation about why such compounds matter, where they fit in modern life, and what it takes to handle them responsibly. My time spent flipping through reference books reminds me: chemistry always comes back to detail, safety, and the real impact on people. Keeping that in mind serves more than just scientists—it shapes better workplaces and, hopefully, a safer world.
How should 1,3-Dinitrobenzene be stored?
Understanding the Risks of 1,3-Dinitrobenzene
1,3-Dinitrobenzene isn’t just another chemical on the lab shelf. The stuff might not explode like TNT, but it brings its own dangers if people don’t treat it with respect. It’s toxic, and inhaling even small amounts or letting it soak into skin can turn into a health scare—causing problems like headaches, dizziness, or methemoglobinemia, which messes with how blood moves oxygen. Years ago, a few colleagues I knew got sick just from cleaning up a small spill. Their experience made me realize good habits in chemical storage keep everyone safer at work.
Keep It Cool, Keep It Dry
Some folks assume that just sticking containers on a shelf works for every kind of chemical, but 1,3-dinitrobenzene changes the rules. Heat starts speeding up chemical reactions, sometimes causing decomposition or, even worse, an unexpected fire. Dry conditions cut down on the odds of hydrolysis and also keep corrosion away from metal containers. Open bins or shelving near a radiator or sunny window sets up a recipe for trouble. Based on what I’ve seen in university and industrial labs, a spot with reliable ventilation, below 25°C, and out of direct light supports safer handling over the long haul.
Containers and Labeling Make a Difference
Glass or tightly-sealed polyethylene containers resist chemical attack and stand up against leaks. Rusty or dented cans let chemicals seep out or let water sneak in. It sounds simple, but proper labeling prevents mix-ups. A faded marker or missing label leads to confusion, and more than one lab accident started with someone grabbing the wrong bottle. I learned to always double-check containers for labels and seals before every use. Time spent on clear labeling reduces future headaches and helps emergency responders know what they’re dealing with if something spills.
Separate Hazardous Substances
One mistake I’ve seen is storing 1,3-dinitrobenzene next to acids, bases, or flammable solvents. The wrong mixture could release hazardous gases or set off an unexpected reaction. Even after working with chemicals for years, I keep a habit of splitting up organics by hazard class. Flammable cabinets work for some, but 1,3-dinitrobenzene likes its own secure, lockable spot away from storerooms with all-purpose storage. Wall-mounted shelving with clear signage prevents crowded bench tops or accidental cross-contact. This kind of separation stops a small mishap from becoming a full-on incident.
Emergency Gear and Training Reduce Harm
Spill response kits, eyewash stations, gloves, and proper ventilation all play a part in safe storage. A fancy storage system doesn’t matter if nobody knows what to do in an emergency. Regular training updates remind everyone what gear to grab or how to move away from a spill. People who work around 1,3-dinitrobenzene benefit from knowing more than just what the label says; they’ve got to understand why certain steps prevent chaos.
Don’t Ignore Inspections
Tucking 1,3-dinitrobenzene on a shelf and forgetting about it never works out for long. Respiratory hazards, leaks, or corrosion show up where you least expect them. Monthly walkthroughs, paired with written logs, catch those problems before they get out of hand. In my experience, teams that take inspections seriously see fewer accidents and spot bad habits before they become routine. Good storage habits grow out of paying attention and putting safety first, every day.
Is 1,3-Dinitrobenzene hazardous to health or the environment?
A Close Look at a Banned Chemical
You don’t hear about 1,3-dinitrobenzene every day, but it keeps popping up in stories tied to industrial accidents and old manufacturing waste. Most folks aren’t familiar with the name, but this stuff can cause real problems in the wrong hands or the wrong place. Having worked a stint in a plant that handled specialty chemicals, I saw firsthand how fast things could go sideways if hazardous materials slipped in through a backdoor in safety protocols. 1,3-dinitrobenzene always landed near the top of the worry list.
What Happens to People
Exposure to 1,3-dinitrobenzene doesn’t quietly pass by. At work, even the whiff of unusual smells sparked alarms. This chemical’s vapors and dust can hurt red blood cells, leading to a type of anemia that starves tissues of oxygen. Shortness of breath and headaches set in—then worse symptoms like cyanosis. I’ve heard OSHA investigators talk about past exposure cases with lasting nerve and reproductive issues. It’s not just about feeling sick one afternoon; these effects can stick to a person for life.
Direct contact causes skin irritation, and there are reports of eye damage too. In some research studies, test animals breathed in or swallowed small amounts, and changes showed up in tissues and on blood tests. The World Health Organization flags nitrobenzenes—including 1,3-dinitrobenzene—as chemicals needing careful scrutiny. That’s not fear-mongering—it’s rooted in medical records and years of accident reports.
How It Lingers in the Environment
Don’t count on 1,3-dinitrobenzene breaking down quickly on its own. Poured out during manufacturing or improper disposal, it can hang around in soil and groundwater. Once in water, aquatic life stumbles—fish and insects struggle with toxic effects, and entire food webs can destabilize. It’s not just theoretical. In places where old factories dumped waste, water samples downriver often show persistent traces of these compounds for years.
The movement of 1,3-dinitrobenzene through soil creates another headache. Drinking water wells become vulnerable if this chemical sneaks past shallow containment. I remember cases from community health studies where small towns faced “do not drink” orders after old chemical tanks leaked. It’s easy to ignore these risks until the tap water changes color or taste, but by then, the cleanup costs multiply fast.
What Can Actually Be Done?
Prevention proves cheaper and safer than trying to clean up after things go wrong. Industry leaders rarely volunteer for expensive changes, but real oversight forces hands. Stronger regulations stop 1,3-dinitrobenzene from getting into the air and water in the first place. At my old job, workplace testing caught leaks early. Employees had to wear good protective equipment, and training taught everyone to treat every unknown chemical with caution.
Waste disposal lines up as the next battlefront. High-temperature incineration of hazardous waste keeps substances like 1,3-dinitrobenzene out of landfills and water. New detection methods can trace faint traces in groundwater, helping environmental scientists pinpoint a spill before it spreads. Community pushback also works. People living near old plants need clearer information—and access to testing kits and medical support if pollution turns up down the block.
The lesson? Trust but verify. An ounce of prevention, stronger laws, and common sense at the job site protect people and the planet from a molecule that never belonged outside lab walls to begin with.
| Names | |
| Preferred IUPAC name | 1,3-dinitrobenzene |
| Other names |
m-Dinitrobenzene Meta-Dinitrobenzene |
| Pronunciation | /ˌwaɪˌɛnˌdiːˌnaɪˌtroʊˈbɛnˌziːn/ |
| Identifiers | |
| CAS Number | 99-65-0 |
| Beilstein Reference | 120922 |
| ChEBI | CHEBI:34557 |
| ChEMBL | CHEMBL14373 |
| ChemSpider | 10468 |
| DrugBank | DB01975 |
| ECHA InfoCard | ECHA InfoCard: 100.003.080 |
| EC Number | 208-601-1 |
| Gmelin Reference | 61353 |
| KEGG | C01762 |
| MeSH | D017350 |
| PubChem CID | 7417 |
| RTECS number | CZ9625000 |
| UNII | ZN27J49A5U |
| UN number | UN2069 |
| CompTox Dashboard (EPA) | DTXSID7020629 |
| Properties | |
| Chemical formula | C6H4N2O4 |
| Molar mass | 168.11 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1.52 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.89 |
| Vapor pressure | 0.0025 mmHg (25°C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | 11.96 |
| Magnetic susceptibility (χ) | -68.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 2.13 mPa·s (25 °C) |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 178.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -44.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3072 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | G04BX10 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes damage to organs; harmful to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H319, H332, H351, H370, H372 |
| Precautionary statements | P210, P260, P264, P270, P271, P273, P280, P301+P310, P304+P340, P305+P351+P338, P311, P312, P330, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 210 °C (410 °F; 483 K) |
| Autoignition temperature | 390 °C |
| Explosive limits | 0.9–7.4% |
| Lethal dose or concentration | LD50 oral rat 240 mg/kg |
| LD50 (median dose) | LD50 (median dose): 240 mg/kg (oral, rat) |
| NIOSH | DY1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,3-Dinitrobenzene is **0.15 mg/m³**. |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | 50 mg/m3 |
| Related compounds | |
| Related compounds |
Benzene Nitrobenzene 1,2-Dinitrobenzene 1,4-Dinitrobenzene Trinitrobenzene |

