1,3-Difluoro-2-Propanol: The Unheralded Building Block Every Chemist Should Know
Historical Development
Chemical innovation rarely comes from the spotlight. Take 1,3-difluoro-2-propanol—a modest molecule, overlooked by anyone who doesn’t keep an eye on the nuts and bolts of synthetic organic chemistry. As fluorinated chemicals began to unravel doors in pharmaceuticals and material sciences through the 20th century, the demand for small, functionalized intermediates with unique reactivity patterns grew. Early studies in the 1960s and 1970s started cracking open methods for selective fluorination, and the synthetic protocols matured throughout the following decades. Researchers recognized that difluorinated alcohols offered a rare combination: a polar, hydrophilic moiety with strong C-F bonds—a formula that attracted attention in specialty chemical labs and industrial pipelines.
Product Overview
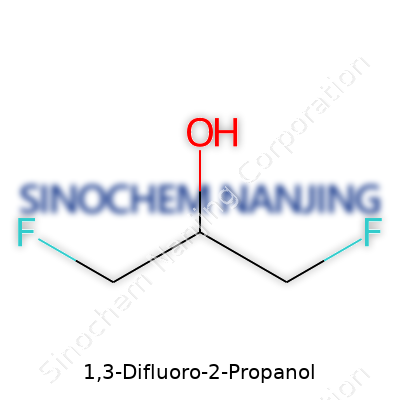
Take a closer look at this compound, and you’ll notice its tight structure: a three-carbon chain, bearing fluorine atoms at the first and third carbon with a hydroxyl sitting at the second. This simple motif makes the molecule both a synthetic intermediate and a potential end-product in specific formulations. Over the years, industries from pharma to agrochemicals decided it holds more value than most would guess from its name alone. Difluorinated alcohols of this sort played roles both as reagents and as parts of more extensive, often bioactive scaffolds.
Physical & Chemical Properties
Sitting in a bottle, 1,3-difluoro-2-propanol comes as a clear, colorless liquid. It blends the usual volatility of small alcohols with the higher density and lower reactivity you’d expect from fluorine substitution. Direct experience in labs backs up literature reports: you’re dealing with something that boils well below water, dissolves well in polar solvents, and resists oxidation better than its non-fluorinated cousin. Those C-F bonds don’t give up easily, and anyone who’s ever tried to hydrolyze a gem-difluoromethylene group knows this is more than textbook thumbing—it forces chemists to think ahead about waste and treatment.
Technical Specifications & Labeling
Specifications for 1,3-difluoro-2-propanol depend on how and where it’s used. Reagent grade stock commonly reports purity in the upper ninetieth percentile, usually above 98 percent by gas chromatography. Labels call out the CAS number along with structural formula, but anyone experienced with chemical procurement recognizes the need to check for trace impurities—especially halide contaminants or remaining starting materials from synthesis. Shipping and storage instructions align with those for volatile, moderately toxic organofluorines: sealed bottles, away from incompatible oxidants, and clearly marked “Flammable” owing to the low flash point.
Preparation Method
Reliable preparation methods grew up alongside demand for this class of structures. Direct fluorination of 1,3-dihydroxypropane doesn’t get you there easily—selectivity isn’t trivial. Instead, practiced chemists lean on halogen exchange, starting from 1,3-dichloro-2-propanol or similar halogenated precursors, treated with reagents like potassium fluoride under phase-transfer conditions. These reactions avoid the hazardous conditions associated with elemental fluorine, favoring safer, more tunable approaches. One-pot syntheses with minimal byproducts often become the goal, and the push for greener chemistry means more reactions move to less toxic solvents or lower temperatures.
Chemical Reactions & Modifications
1,3-difluoro-2-propanol stands out because its structure invites both nucleophilic and electrophilic attacks—but only on the right terms. The hydroxyl group opens a path to form esters, ethers, or even more complex acyclic derivatives, yet the fluorines guard their carbon neighbors, making displacement reactions tricky. In their own experience, seasoned synthetic chemists turn to this molecule when looking to introduce both polarity and stability in a new scaffold—especially since the difluoromethylene segment tends to resist metabolic breakdown. Modifications often include transformations at the oxygen: oxidations to form the ketone, or subtle conversions under catalytic conditions for target analogues.
Synonyms & Product Names
Across literature and laboratory stockrooms, 1,3-difluoro-2-propanol takes on aliases: 2-hydroxy-1,3-difluoropropane, or by its registry number. Some suppliers sell it under generic “difluoropropanol” tags. Consistency matters—confusion with monofluorinated or trifluorinated versions can derail whole synthetic projects. I’ve seen more than one new researcher order the wrong bottle, expecting results that never come. Cross-checking with CAS numbers and structured datasheets cuts down these headaches.
Safety & Operational Standards
Direct handling of this alcohol means respecting its volatility, its flammability, and the moderate toxicity associated with many small-molecule fluorinated compounds. Personal protective equipment—nitrile gloves, splash goggles, and solid ventilation—should be considered non-negotiable. Accidental inhalation or skin contact brings irritation risks, and spills, although not highly persistent in the environment, deserve careful cleanup to avoid workplace exposure. Facilities usually mandate local exhaust and spill kits nearby, echoing broader chemical safety rules aimed at organic solvents.
Application Area
Flexible reactivity makes 1,3-difluoro-2-propanol valuable in synthesis of specialty pharmaceuticals, agrochemicals, and even polymer building blocks—mainly when researchers want to test effects of fluorine substitution on biological or material properties. For example, placing difluorinated units into drug candidates alters both metabolic stability and bioavailability; in many cases, the goal is to slow down unwanted degradation or to fine-tune the shape and polarity of a molecule. In polymer chemistry, modifications with this molecule open routes to more weather-resistant, chemically inert end products. First-hand, one hears stories from medicinal chemistry teams about how a swap for a -CF2 group transforms an average candidate into something new.
Research & Development
Ongoing research digs into more selective synthesis and greener routes to this compound. Laboratories across academia and industry push for catalytic, low-energy methods, using recyclable fluorinating agents. There’s a constant search for ways to expand the molecule’s reactivity, tailoring it for cross-coupling, for asymmetric syntheses, or for late-stage functionalization in complex scaffolds. As part of multidisciplinary teams, I’ve seen R&D projects hinge on whether small intermediates like this are commercially available at reasonable purity and price. As more tools develop—automation, flow chemistry reactors, continuous purification—access to these “niche” alcohols will only grow.
Toxicity Research
Fluorinated alcohols come with a mixed safety record. While not the most dangerous in their class, compounds like 1,3-difluoro-2-propanol still pose risks through inhalation, ingestion, or skin absorption. Toxicological data remains somewhat limited, especially compared to extensively studied analogues. Animal studies suggest moderate acute toxicity, with primary risks related to central nervous system depression and potential for cumulative organ impacts at higher exposures. Experienced handlers know to advocate for real-time monitoring, and ongoing improvement in detection methods has helped limit workplace incidents. Calls for full toxicological profiles echo across regulatory tables, especially as downstream metabolites or waste streams may pose unexpected hazards.
Future Prospects
Chemistry advances fastest when simple ideas breed new opportunities. 1,3-difluoro-2-propanol, humble as it seems, could find fresh life as demand for tailored fluorination continues to rise. Applications in pharmaceuticals, material sciences, and next-generation polymers all hinge on reliable access to flexible, well-documented building blocks. With pressure growing for green chemistry and safer industrial protocols, more sustainable synthesis and effective recycling routes are set to become standard. The push for deeper toxicity data and better environmental controls promises a future in which molecules like this earn new trust from industry and regulators alike. If my experience building out synthetic libraries has taught anything, it's that innovation relies on the right “small” choices—and among these, difluorinated intermediates are increasingly hard to ignore.
What is the chemical formula of 1,3-Difluoro-2-Propanol?
Breaking Down the Basics
1,3-Difluoro-2-propanol seems like something straight out of a dense college textbook, but this compound brings a few real-world implications. Its chemical formula—C3H6F2O—tells a short story about structure, reactivity, and potential impact.
Molecular Story Unfolded
Diving into its name, "propanol" means a three-carbon backbone topped with an alcohol group. The “1,3-difluoro” part means that there are fluorine atoms bonded to the first and third carbon atoms. This changes how the molecule acts in a reaction or inside the body. Tossing two fluorine atoms on the ends can turn a simple alcohol into a chemical with different uses and risks. For someone with chemistry experience in a lab, seeing fluorine substitutions on a short-chain alcohol signals a change in both toxicity and chemical properties. Fluorine atoms love to hold onto their electrons, making bonds tight and hard to break—something that matters both in research and in environmental settings.
From Structure to Safety
Most compounds with added fluorine have a stronger metabolic kick compared to non-fluorinated relatives. Some are even more persistent if released into soil or waterways. Industries often use related fluorinated compounds to increase lifespan in products, or as specialty solvents. But this persistence leads to questions about safe handling and disposal. Struggling to properly contain these chemicals puts workers at risk and could threaten water sources.
Studying chemicals like 1,3-difluoro-2-propanol doesn’t just rest on the shoulders of chemists or students cramming for exams. As more fluorinated chemicals circulate through labs, factories, and supply chains, accidental exposures become more likely. The US Environmental Protection Agency and other agencies track these types of chemicals for a reason. The tricky part is staying updated on safe thresholds and disposal practices. Missteps sometimes leave a long legacy—polluted wells, harmed wildlife, and costly cleanups. Mistakes made around fluorinated alcohols can stick around a long time, costing communities and businesses far more than a little extra caution up front.
Practical Solutions: Mind the Molecule
Paying attention to the chemical formula does more than solve a test question; it gives clues for better choices on storage and waste. My time working in research taught me the value of simply keeping up with labels, double-checking storage instructions, and not skipping safety training. Spills or misuse often result from small gaps in attention or shortcuts, not big acts of recklessness.
For industries and labs using chemicals like this, clear labeling and accessible information go a long way. Creating routine check-ins and updating safety sheets helps teams avoid guesswork. Collaboration with environmental health experts can lead to smarter ways of phasing out persistent compounds, or switching to alternatives when possible.
Everyday Lessons from a Simple Formula
Science doesn’t live in a vacuum. Even the humble formula C3H6F2O holds weight outside academic circles. Understanding what each part stands for in practical terms makes a difference in how we work, what risks we accept, and how we protect people and places down the line.
What are the main uses or applications of 1,3-Difluoro-2-Propanol?
A Closer Look at a Powerful Building Block
Anyone who’s dipped their toes into chemical research will tell you, specialty compounds set the tone for what’s possible in science and manufacturing. 1,3-Difluoro-2-propanol proves this point. With its two fluorine atoms and a single hydroxyl group sitting on a three-carbon backbone, this molecule turns up where precision chemistry matters most. It's not as famous as Teflon or other household fluorinated names, but dig beneath the surface and you’ll spot its fingerprints in medicines, specialty materials, and laboratory tweaks that keep science moving forward.
Paving the Way in Drug Discovery
Medical chemists constantly hunt for new scaffolds and fragments that help build novel drug molecules. 1,3-Difluoro-2-propanol enters the frame for its ability to introduce fluorine atoms precisely where they're needed, tuning “drug-like” properties in unique ways. Fluorine improves stability and changes how molecules fit inside the body, often increasing the success rate for drugs in development. You see this with small-molecule antivirals, some cancer treatments, or even certain imaging agents used in diagnostics. It’s satisfying to know that such a small molecule can change the odds for medicines that reach the world’s clinics.
Shaping Specialty Polymers and Performance Materials
Material scientists love tools that allow them to customize polymers for challenging uses. Fluorinated monomers, like 1,3-difluoro-2-propanol, fit the bill. They often give rise to polymers resistant to heat, acids, and even the stickiest solvents. The flexible backbone and fluorine content make it easier to design materials for electronics, membranes, or coatings that outlast those based on older formulas. I’ve walked through labs where researchers carefully pick their monomers, looking for just the right mix to solve a material challenge—fluorinated alcohols appear on those shortlists more than you’d guess.
Reactivity and Chemistry with a Purpose
Chemists searching for molecular “handles” know the hydroxyl group in this molecule invites a range of reactions. It reacts with acids, acyl chlorides, and even metallic catalysts, kicking off synthesis routes that let labs build bolder, more functional compounds. This versatility means it’s not just a standalone product, but a bridge for inventing new compounds—sometimes the only way to explore fresh chemical territory.
Meeting Real-World Challenges and Responsible Use
Along with its strengths, fluorinated compounds tend to stick around in the environment. Scientists and regulators scrutinize their lifecycle and impact, especially with the surge of interest in “forever chemicals.” Manufacturers and research teams who rely on 1,3-difluoro-2-propanol focus on responsible sourcing, safe handling, and mindful waste disposal. I recall a training session where experienced lab managers drilled into us that safety and stewardship don’t just tick boxes—they protect health, reputation, and trust within the scientific community.
Building a Stronger Chemical Toolkit
1,3-Difluoro-2-propanol may never become a household name, but those who rely on specialty chemicals know its worth in developing better drugs, stronger materials, and smarter chemistry. By prioritizing quality sourcing and safe use, the benefits can shine without leaving a problematic legacy—just as any cutting-edge technology should.
What are the safety precautions when handling 1,3-Difluoro-2-Propanol?
Getting to Know the Risks
Working with chemicals like 1,3-Difluoro-2-Propanol means facing hazards many people outside the lab never think about. This liquid has a knack for causing skin and eye irritation. Breathing in its vapors can leave your lungs burning. It’s a small molecule, so it sneaks through the air and finds cracks under goggles and gloves if you aren’t careful. Some of my earliest chemistry work taught me how quickly a moment’s distraction with these kinds of fluorinated alcohols can turn into weeks of itching, hacking, or worse.
Suit Up: Personal Protective Gear
Every time you reach for the bottle, the basics matter—nitrile gloves (not latex, which breaks down), a lab coat buttoned closed, well-fitted chemical splash goggles, and a face shield when things might splash. Good shoes keep spills from soaking through socks. Long sleeves and pants cut the risk of droplets burning your skin. Pushing sleeves back or skipping gloves always tempts fate. Once, during a late-night session, a colleague ignored a small tear in her gloves—her skin paid for it with stinging welts the next day.
Ventilation and Air Flow
Never trust your nose to warn you about fumes from 1,3-Difluoro-2-Propanol. Most labs use chemical fume hoods for a reason. Strong airflow pulls vapors away before you ever breathe them in. Never lean your head into a hood to “get a closer look”; stay behind that sash. Setting up proper ventilation at a working height and keeping the sash as low as possible shields both lungs and eyes. For anyone working in a small lab with aging infrastructure, portable filtration units become essential backup.
Spill Ready: Quick Actions Matter
Accidents don’t wait until you’re ready. Spills happen fast. Put absorbent pads nearby and know the nearest shower location. Small spills get covered right away and cleaned with appropriate neutralizing agents (a mix of sodium bicarbonate and water works better than wishful thinking). I once saw a grad student freeze in panic with a broken bottle—training and quick muscle memory turn that fear into action. Practice matters: run through what you’d do in a spill before it ever happens.
Storage and Labeling: No Guesswork
Good labeling saves headaches and hospital trips. Every container should spell out what’s inside—no mystery bottles. Flammable cabinets keep this liquid away from both heat and light. Sealing caps tightly keeps vapors from drifting out. Forgetting to chill the bottle or store with desiccants can lead to dangerous pressure buildup. Double-check seals, date your containers, and never store near acids or oxidizers.
Training and Real-World Smarts
Rules mean less if nobody follows them. Initial safety classes lay the groundwork, but regular refreshers stop protocol from becoming background noise. From my own experience, the labs with the fewest accidents talked safety often—every meeting started with reminders or close-call stories. People look out for each other.
Handling Waste: Don’t Cut Corners
Dumping leftover 1,3-Difluoro-2-Propanol in the sink risks community health and runs afoul of the law. Special containers marked for halogenated alcohols keep everything separated until professional disposal. I’ve seen new staff try shortcuts out of convenience, only to spark big problems down the line. Taking five extra minutes for proper waste holds everybody’s trust.
The Bottom Line
Respect for chemicals like 1,3-Difluoro-2-Propanol builds from daily habits. Suit up with the right gear, keep spaces well-ventilated, prepare for accidents, label everything, push for safety culture, and handle waste responsibly. Each of these steps means fewer close calls and healthier colleagues. In the end, safety becomes less about fear and more about respect. That’s what keeps people coming home at the end of each shift.
How should 1,3-Difluoro-2-Propanol be stored?
Simple Steps Save Trouble
Anyone working in a lab knows chemicals with names like 1,3-Difluoro-2-Propanol do not play around. This isn’t the type of stuff you want to stick next to your orange juice, but that’s where common sense starts and stops. From my time in the field, learning the right way to store a chemical means protecting people, equipment, and your own peace of mind. I’ve seen more than a few messy labs, and trust me, shortcuts bite back.
Keep It Cool, Keep It Dry
This compound needs a space that stays cool—ideally under 30°C. Excess heat can make chemicals volatile, raising the odds of leaks or pressure build-up. Humidity lets water sneak in, leading to possible reactions or even decomposition. At a university lab, years ago, humidity warped seals and caused a leak in a reagent bottle, and that set off the incident log for months. Don’t give moisture a foothold; store in a dry place away from sinks or open windows.
Avoid Sunlight and Ignition Sources
Direct light or heat speeds up chemical breakdown. Fluorinated compounds can get unstable, especially under harsh lighting. At one industrial site, a coworker once left a bottle near a window. The label faded and the liquid darkened. We learned the importance of tucking stuff into closed, shaded cabinets. Simple metal or polymer cabinets do the job—no fancy setup necessary, just out of the sun.
Use Proper Containers: Safety First
Never skimp on containers. Glass works for some chemicals, but a good, fluoropolymer-lined bottle gives an extra barrier. These bottles resist chemical corrosion and hold up better if you need long-term storage. Seal tight! Any whiff of vapor or liquid means a waste of money and a risk to your health. Years back, I saw corrosion turn aluminum shelving black around a bottle that leaked. That’s not just unsightly, it’s unsafe.
Separation Stops Surprises
Storing everything together is inviting disaster. 1,3-Difluoro-2-Propanol belongs in its own spot, away from bases, acids, and oxidizers. Mixing storage means a spill or break could spark a bigger chemical reaction. I remember an incident in a teaching lab—one careless student stacked acids too close to organics, and that mistake led to an evacuation. Mark shelves clearly. Include hazard labels and instructions, so even a new hand knows the drill.
Ventilation Means You Breathe Easy
Good airflow is the best insurance. Fumes from some chemicals rise fast and spread farther than you think, often before your nose picks them up. I’ve always trusted chemical fume hoods and local exhaust systems. Keeping bottles outside high-traffic areas reduces exposure risk. If the smell spreads, that’s already too late for prevention.
Training Shuts Down Problems Before They Start
Workers can’t guess their way through chemical safety. At every job I’ve held, hands-on training made the difference—labels, datasheets, and protocols posted in plain sight keep mistakes to a minimum. I’ve seen coworkers jump into action because they knew right where spill kits and emergency eyewash sat.
Proper Disposal Wraps Up the Cycle
No chemical is forever. Outdated or unneeded bottles bring a risk no one needs. Routinely check expiry dates and disposal protocols. Chemical waste programs make sure old stock doesn’t stick around to surprise anyone later.
What is the boiling point and physical properties of 1,3-Difluoro-2-Propanol?
Understanding the Nature of 1,3-Difluoro-2-Propanol
1,3-Difluoro-2-propanol isn’t something many people casually keep in their garage. This compound grabs attention for its unique structure, sitting somewhere between simple alcohols like ethanol and the punchier, highly reactive chemicals that only show up in a controlled lab setting. The presence of two fluorine atoms on either end of the propanol backbone changes things in significant ways.
Boiling Point Facts and Practical Impact
Precision matters in chemistry, so let’s get the numbers right. The boiling point of 1,3-difluoro-2-propanol sits at about 104-106°C (219-223°F). This is higher than regular propanol, which boils off closer to 97°C. Adding fluoride into a molecule raises its boiling point — not by accident. Fluorine’s pull makes molecular bonds tighter and the resulting intermolecular forces stronger, so more heat is needed to send the liquid into a vapor.
Knowing the boiling point isn’t just trivia. If you’re working in a lab, you want a precise margin for safety, especially since higher boiling points often mean slower evaporation, but not always less volatility. It changes how you’d set up your experiments and what kind of ventilation or closed systems matter most.
What Else Sets the Physical Properties Apart?
It’s not just about temperature. The two fluorine atoms on the ends bring other quirks. Take density. Compared to regular 2-propanol, 1,3-difluoro-2-propanol is denser, floating closer to 1.24 g/cm³ even at room temperature — regular propanol sits around 0.79 g/cm³. That means, in practice, the same volume weighs a lot more, and storage containers need to be labeled and handled with care.
Colorless, clear, and giving off a bit of a biting odor, this liquid doesn’t alert the nose quite as strongly as ammonia, but nobody would confuse it for water, either. Its molecular formula — C3H6F2O — signals that a shift in reactivity might sneak up on a chemist expecting regular alcohols. Fluorine’s presence often makes many chemicals more resistant to breakdown by light or heat, but that doesn’t automatically mean safe. Inhalation, skin contact, or accidental mixing can still create trouble, so routine glove-and-goggles policy stands.
Why Anyone Should Pay Attention to These Properties
Few people outside chemical plants or research labs will ever handle this compound, but knowing how 1,3-difluoro-2-propanol behaves matters for those creating specialty solvents or certain pharmaceutical intermediates. It’s not about curiosity — it’s about predictability and safety. Handling, shipping, or storing fluids with strong fluorine-carbon bonds often means extra rules for hazardous materials transport. There’s a reason we separate out these compounds from the everyday alcohols used for cleaning or medical use: unexpected reactions can happen.
Extra care in chemical labeling, storage away from incompatible materials, and robust spill management form the minimum standards. Emergency protocols aren’t just paperwork. Having worked in environments with fluorinated chemicals before, I’ve seen what happens when ventilation falls short or a label gets missed. The consequences run from minor skin burns to more lasting trouble.
Considerations for Use and Handling
The technical specifics — boiling point, density, reactivity — aren’t just charts for specialists. Anyone involved in lab safety, chemical supply, or industrial processing ought to recognize how small tweaks in a molecule create big changes in risk. Design storage based on real measurements, keep clean-up materials at hand, and continuously train anyone likely to work with rare compounds like this one.
Fluorinated alcohols often play roles that no other chemical fills, but with those benefits come the demands of deeper expertise, better gear, and steady vigilance.
| Names | |
| Preferred IUPAC name | 2,2-Difluoropropan-1-ol |
| Other names |
1,3-Difluoropropan-2-ol 1,3-Difluoroisopropanol 2-Hydroxy-1,3-difluoropropane |
| Pronunciation | /ˈwʌnˌθri daɪˈfluːəroʊ tuː ˈproʊpənɒl/ |
| Identifiers | |
| CAS Number | 454-10-8 |
| 3D model (JSmol) | `load chemical:CC(O)CF` |
| Beilstein Reference | '1738736' |
| ChEBI | CHEBI:36960 |
| ChEMBL | CHEMBL517679 |
| ChemSpider | 52211 |
| DrugBank | DB01852 |
| ECHA InfoCard | 100.013.802 |
| EC Number | 206-934-6 |
| Gmelin Reference | 137132 |
| KEGG | C19238 |
| MeSH | D023442 |
| PubChem CID | 123227 |
| RTECS number | UE9275000 |
| UNII | 44B5F1A7A3 |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C3H6F2O |
| Molar mass | 98.05 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet odor |
| Density | 1.29 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | -0.18 |
| Vapor pressure | 0.5 mmHg (at 20 °C) |
| Acidity (pKa) | 15.54 |
| Basicity (pKb) | Basicity (pKb): 8.96 |
| Magnetic susceptibility (χ) | -47.6e-6 cm³/mol |
| Refractive index (nD) | 1.353 |
| Viscosity | 1.338 cP (25°C) |
| Dipole moment | 2.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.7 J/mol·K |
| Std enthalpy of formation (ΔfH⦵298) | -585.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1249.1 kJ/mol |
| Hazards | |
| Main hazards | Toxic if swallowed. Toxic in contact with skin. Causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS02, GHS05, GHS07 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P280, P301+P310, P304+P340, P311, P312, P321, P330, P363, P405, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 103 °C (217 °F; 376 K) |
| Autoignition temperature | 460 °C |
| Lethal dose or concentration | LD50 Oral Rat 1650 mg/kg |
| LD50 (median dose) | LD50 (median dose): 315 mg/kg (rat, oral) |
| NIOSH | UE4825000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1,3-Difluoro-2-Propanol: "No OSHA PEL established |
| IDLH (Immediate danger) | IDLH: Unknown |

