1,3-Dichloroacetone: An In-Depth Commentary
Historical Development
The story of 1,3-Dichloroacetone traces back to the early investigations into halogenated ketones during the expansion of organic chemistry in the late 19th century. At the time, chemists looked for simple ways to enhance reactivity for various syntheses. Adding chlorine atoms to an acetone molecule led to a product with a sharp bite, setting it apart from its non-halogenated cousins. Researchers in industry soon recognized the utility offered by chlorinated compounds in manufacturing pharmaceuticals and specialty chemicals. The availability of cheap chlorine gas and advances in purification meant production could scale, encouraging further research and giving this ketone a niche behind laboratory doors that it never really lost.
Product Overview
As a colorless liquid with a distinct pungent odor, 1,3-Dichloroacetone stands out for its chemical activity. Folks working around it respect its volatility and razor-sharp reactivity. Anyone familiar with synthetic methods that require strong alkylating agents or highly electrophilic carbonyls has probably seen this compound show up on a shelf or catalog. While it doesn't turn up in household cleaners or fertilizers, it quietly enables other products in the pharmaceutical and agrochemical sectors to take shape.
Physical & Chemical Properties
With a molecular formula of C3H4Cl2O and a molar mass around 126.97 g/mol, 1,3-Dichloroacetone looks like water in the flask but delivers a punch when exposed to skin or the nose. Its boiling point floats around 154°C, and it sports a density of about 1.38 g/cm³. The vapor pressure clocks in on the moderate side, nudging technicians to work with good ventilation. Chlorine atoms at both the first and third positions in the chain turn what would be a standard acetone backbone into something far more reactive. The chlorination boosts electrophilicity, making carbonyl carbon easy prey for nucleophiles. The molecule’s solubility in organic solvents comes in handy during multi-step syntheses, since the operator can pull it out of a reaction mix with little effort.
Technical Specifications & Labeling
Product labeling for 1,3-Dichloroacetone speaks volumes. Labels typically scream flammable and toxic, marked with hazard pictograms that nobody with common sense ignores. Purity levels often run above 98%, demanded by the needs of fine chemical synthesis. Suppliers flag any water content or hydrolyzed derivatives, since these can tank a planned reaction. Volume packaging varies from one liter amber glass bottles to bulk metal drums, but the smaller sizes prevail at research scale. Tech sheets spell out spectroscopic data and fingerprint the substance using GC-MS or NMR, leaving little doubt as to what’s inside each container. Regulatory identification numbers and hazard codes appear front and center to warn even the most distracted handler what they are dealing with.
Preparation Method
Manufacturing 1,3-Dichloroacetone isn’t exactly backyard chemistry. Factories usually rely on either the chlorination of hydroxyacetone or the controlled halogenation of acetone derivatives. Brands prefer batch processes for smaller runs but shift to continuous systems when larger volumes make sense. The reaction management involves tight temperature control and fume extraction, since runaway chlorination generates dangerous byproducts. Purification steps often include fractional distillation under reduced pressure—crucial for removing residual reactants or chloroacetone isomers. Quality control samples routinely head straight for the analytical chemist’s bench, who hunts for minute color or odor shifts as an early warning of decomposition or contamination.
Chemical Reactions & Modifications
This ketone’s appetite for nucleophilic attack makes it a favorite in organic synthesis. The chlorine atoms activate the methylene group, making the compound react eagerly in alkylation or condensation steps. Students see 1,3-Dichloroacetone featured in lab manuals for halohydrin or ether syntheses, and professionals turn to it for crafting heterocycles or intermediates used in antibacterial drugs. Its behavior under Grignard or organolithium reagents brings practical challenges, since side reactions can compete, but also allows for unusual transformation routes. Explorers in medicinal chemistry modify it by swapping out the chlorine for other functional groups or using its carbonyl as a handle to build new scaffolds, keeping the molecule relevant after more than a hundred years.
Synonyms & Product Names
I’ve seen it marketed under names such as 1,3-Dichloropropan-2-one and 1,3-Dichloro-2-propanone. For folks flipping through older textbooks or international supply catalogs, dichloroacetone or sym-dichloroacetone often appears. CAS number 534-07-6 identifies it for regulatory and procurement clarity. No matter the moniker, the chemistry never changes, and all these aliases end up describing the same reactive, eye-watering liquid.
Safety & Operational Standards
Ignorance toward this chemical ends badly. In my own work with halogenated ketones, I never skip goggles, gloves, or a fume hood. 1,3-Dichloroacetone evaporates fast, and its vapors attack mucous membranes, producing burning in the eyes and lungs. Spill kits and vapor monitoring belong in any lab where this bottle appears. Safety sheets recommend safe handling far away from open flames or sources of static electricity, and you can’t overstate the case for double-checking all connections and seals. Disposal routes avoid drains and rely on incineration at hazardous waste plants, not the municipal bin. Training staff and enforcing clear standard operating procedures keep both people and property out of trouble.
Application Area
You probably won’t find this compound featured in consumer headlines, but its fingerprints show up in surprising niches. Pharmaceutical chemists reach for 1,3-Dichloroacetone as a building block for new drug candidates, especially when exploring compounds targeting bacterial infections or metabolic pathways. Some companies rely on it to make specialty polymers where reactivity trumps material cost. It helps in crafting certain pesticides and fungicides, giving growers more control over crop diseases—though environmental groups watch these uses closely for chlorinated residue concerns. In academic labs, 1,3-Dichloroacetone helps teach students about carbonyl reactivity and synthetic planning, often serving as a case study in practical hazards and risk management.
Research & Development
Research groups lean on 1,3-Dichloroacetone for inspiration. Its versatility sparks new synthetic schemes for preparing advanced materials or exploring uncharted chemical reactions. In my experience, chemists enjoy the challenge of taming this reactive molecule, seeking gentler routes to modify or harness it. Computational chemists use it to probe reaction mechanisms, mapping out electron flows during halogen exchanges or nucleophilic attacks. The pharmaceutical pipeline regularly tests derivatives for activity against antibiotic-resistant pathogens. A handful of teams examine greener chlorination techniques, hoping to cut out toxic byproducts without sacrificing yield or purity.
Toxicity Research
Toxicologists worry about chloro-ketones for good reason. Rat studies report eye, skin, and respiratory tract irritation at low concentrations. The molecule’s high reactivity means it modifies proteins and enzymes on contact, accounting for acute symptoms in laboratory accidents. Environmentally, it persists in water unless broken down under harsh conditions, raising concerns among municipal water testers. Chronic exposure data remains limited, but as a rule of thumb, even brief contact or inhalation needs prompt and thorough first aid. Embedding a safety-first mindset in every operation involving this chemical—whether academic or industrial—helps cut down on incidents and longer-term problems.
Future Prospects
Looking ahead, chemists remain interested in new uses for 1,3-Dichloroacetone, mainly because of the demand for ever more complex drug molecules and specialty materials. Efforts continue to design catalytic protocols that reduce waste and energy use in its manufacture, responding to pressure for more sustainable processes. Regulators may clamp down harder on discharges and workplace exposure, pushing producers to develop safer packaging or in situ generation systems that cut down bulk storage and transport risks. Computer-aided design tools now help researchers predict what new molecules can be built using this ketone as a springboard, expanding its appeal beyond traditional uses. All signs point to 1,3-Dichloroacetone keeping its place as a linchpin in chemical synthesis, as long as handlers treat it with the seriousness it deserves.
What is 1,3-Dichloroacetone used for?
Chemical Curiosity Meets Practical Value
People don’t often talk about 1,3-Dichloroacetone, unless they’re up to their elbows in a chemistry lab or somewhere deep in a manufacturing facility. This isn’t a substance sitting around in cleaning cabinets or medical kits. It has a sharper purpose, drawn from its unique structure as a chlorinated ketone. As someone who’s kept track of chemical safety for work, the uses and risks attached to such chemicals always get my attention.
Why Industries Care About 1,3-Dichloroacetone
This chemical finds its place in research labs and development pipelines, mostly because of its reactive nature. Chemists use it as a building block, working its way into bigger, more complicated molecules. Pharmaceutical research often explores many obscure reagents, and 1,3-Dichloroacetone attracts chemists with its two chlorine atoms flanking the acetone backbone. This structure lets researchers branch out into new territory when building medicines or exploring metabolic pathways.
In the realm of organic synthesis, 1,3-Dichloroacetone behaves as a reliable intermediate. Researchers use it to introduce specific groups onto molecules, shaping everything from experimental drugs to new agricultural agents. Its place isn’t permanent; it often kicks off a reaction and then quietly disappears as the work gets more focused and specialized. Over the years, direct experience with chemical inventories has taught me to appreciate how these small molecular tweaks can unlock new solutions or, sometimes, unknowingly invite fresh risks.
Risks Demand Respect, Not Just Rules
Having handled and tracked hazardous substances, I know safety isn’t just about regulations—it’s about attitude. 1,3-Dichloroacetone carries warning labels for good reason. It can irritate skin and eyes, and inhaling its vapors proves dangerous. Researchers must stay up to date with safety data sheets, keep ventilation systems in good working order, and never skip on gloves or goggles. Over the past decade, I’ve seen enough lab mishaps to believe that careful handling and strong training do more for safety than signs on a wall.
Hidden in the Background—and for a Reason
You won’t find many products listing 1,3-Dichloroacetone as an ingredient. Its purpose lives behind the scenes, helping guide molecular building blocks into new forms. Think of it as scaffolding—essential for construction, removed before the building stands tall. Its chemistry supports many things, but leaves little evidence when all is done.
Research into new medicines and materials keeps this chemical in occasional demand. With the constant need for better treatments and resilient crops, these foundational substances don’t lose their relevance. At the same time, proper tracking, storage, and disposal remain essential to protect workers, communities, and the environment.
Looking Forward: Balancing Progress and Responsibility
As innovation pushes boundaries in synthetic chemistry, safer alternatives and greener methods should stay on the priority list. Labs can audit their protocols, look for less hazardous reagents, and advocate for protective gear that really gets used, not just stored on a checklist. Having seen the tension between productivity and safety, it’s clear that real progress links new discovery with genuine respect for every person who handles these chemicals.
So even if 1,3-Dichloroacetone rarely gets headlines, anyone working with it deserves up-to-date information, access to strong training, and a culture that looks out for more than just profit or output. That commitment keeps science moving ahead—without leaving health and safety behind.
What are the hazards and safety precautions for handling 1,3-Dichloroacetone?
A Closer Look at 1,3-Dichloroacetone
Few chemicals grab attention in the lab quite like 1,3-Dichloroacetone. The smell alone—the sharp, acrid edge—sends a message: this is not something casual. Used as a building block for pharmaceuticals and organic synthesis, its reactivity comes paired with real risk. Not many folks outside chemistry circles have handled it, though anyone who has will tell you, mistakes stick with you for years.
Hazards That Can’t Be Ignored
1,3-Dichloroacetone belongs to a family of halogenated ketones known for toxicity, volatility, and corrosiveness. Each of those qualities brings its own baggage.
Contact with skin or eyes causes severe burns. Vapors irritate the respiratory tract almost immediately. Anyone with asthma or preexisting conditions feels it most. Inhalation exposure can spark headaches, nausea, and even shortness of breath. I’ve seen a colleague cough for hours after a single unplanned whiff. Even in small amounts, this chemical can overwhelm basic lab ventilation.
There’s also flammability. 1,3-Dichloroacetone catches fire easily at typical room temperatures and burns with an invisible flame. Fire means poisonous gases: hydrogen chloride, phosgene, carbon monoxide, all much more dangerous than smoke alone. Once, an improperly discarded towel started to smolder; only fast action stopped a much bigger disaster.
Longer term, this compound may harm organs after sustained or repeated exposures. Unlike some solvents that evaporate before causing much harm, 1,3-Dichloroacetone lingers on surfaces and gloves.
Practical Safety Steps Every Handler Should Know
There’s no shortcut with chemicals like this. Anyone handling 1,3-Dichloroacetone should plan each session in advance. Proper engineering controls help, but I’ve noticed personal habits matter every bit as much.
A certified fume hood is the starting point. Not just a bench shield or desktop vent—a true hood with airflow checked that day. Goggles and face shields come next, no exceptions. Standard safety glasses leave gaps where a splash loves to land. I’ve picked up the habit of checking for damaged spots on gloves before and after every use. Double gloving makes sense for anything this aggressive.
Clothing protects more than people realize. Lab coats made from flame-resistant material catch drips and can be whipped off in an emergency. Disposable sleeves and shoe covers make decontamination easier and remind you of the seriousness each time you suit up.
Anyone handling waste knows mixing with acids or bases makes things worse fast. Labeling and segregating waste cuts down on surprises. If a spill occurs, most dry absorbent pads just spread vapors, so I prefer neutralizing agents matched to halogenated ketones. Cleanup kits belong close at hand, not across the lab.
Training grows more important than gear. My most cautious mentors drilled routines into us: never work alone, keep antidote stations restocked, remind others to check emergency eyewash, and rehearse what to say if you call for help.
Building a Safer Practice
Hazards from 1,3-Dichloroacetone draw sharp lines in the sand: smart habits, clear training, and respect for what this chemical can do. Nobody forgets the experience of dealing with a spill or a near miss. Shortcuts don’t save time; they raise risk. I’ve learned careful planning and teamwork do more to create safety than any single piece of equipment. Each time I see a bottle on the bench, I remember the lessons I picked up—the hard way—and pass them on to the next chemist I meet.
What is the molecular formula and structure of 1,3-Dichloroacetone?
Molecular Formula of 1,3-Dichloroacetone
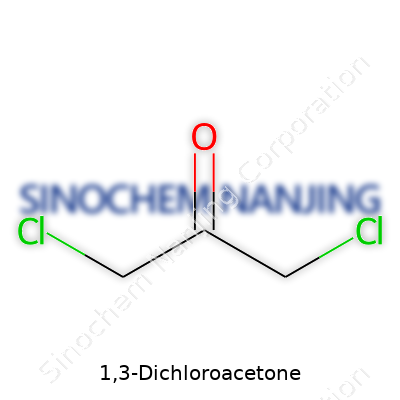
1,3-Dichloroacetone carries the formula C3H4Cl2O. This combination tells you it's a three-carbon molecule, trimmed with two chlorine atoms and capped by a single oxygen atom. The backbone follows the template of acetone (propanone), except the usual hydrogen atoms at the first and third carbon positions have been swapped out for chlorine. This shift sets up a reactive and polar compound, one that demands respect in any lab setting. I’ve encountered a handful of chlorinated ketones in the lab, and they tend to command both scientific curiosity and a bit of caution from the crews who work with them.
Chemical Structure and Functional Groups
The structure stands as CH2Cl–CO–CH2Cl. Layman's terms: take propanone, swap out the hydrogens at each outer carbon for chlorines, and you’re left with a symmetrical molecule. The middle carbon still forms the core ketone group (C=O). Chlorine at the first and third carbons draws electrons, changing the reactivity that’d show up in plain old acetone. This electron-withdrawing effect can push the molecule to behave as a more potent electrophile.
Picture this: the molecule sits with a central carbonyl and two identical arms, each tipped with a chlorine. The structure isn’t just a textbook example; it’s the kind of feature that hints at how a small tweak in atoms changes everything about a material’s behavior, right down to its boiling point and how it interacts in organic synthesis.
Why This Matters in Chemical Research
1,3-Dichloroacetone opens the door to several synthetic routes in organic chemistry. In my own research, compounds like this ended up on the reaction bench as intermediates, not final products. Their use shows up in designing new pharmaceuticals, creating advanced polymers, or engineering specialty reagents. The two chlorines add a handle for nucleophilic substitution — a tactic that helps chemists build bigger, more complex molecules from smaller pieces.
This goes beyond just curiosity, as chlorinated ketones hydrolize easily and can form hazardous byproducts. For someone in the lab, that means thinking twice about safety shields and ventilation. The risk isn’t theoretical—exposure can cause respiratory or skin irritation and, in poorly managed circumstances, more severe toxic effects. It’s impossible to overlook the environmental baggage attached to halogenated organics. Poor disposal contaminates soils and water and even interferes with some treatment facilities. Awareness here isn’t overkill; it’s required for any responsible operation.
Building Responsibility Around Use and Disposal
Handling chemicals like 1,3-dichloroacetone responsibly means aligning daily practices with environmental and safety protocols. In my experience, double-checking waste labeling and setting up a secondary containment system reduces risk significantly. Simple steps like tracking inventory and updating safety data sheets help keep folks in the loop about what’s present in storage and what risks each chemical carries. Industry and research teams can invest in closed-loop waste systems and greener alternatives to minimize impact. Modern chemical education pushes for greener solvents and reduced chlorinated compound use, but sometimes scientific demands require employing these tools anyway.
Solid knowledge of the real structure and properties of 1,3-dichloroacetone forms a backbone for progress, not just in the lab but in the policies and choices that shape chemical safety every day.
How should 1,3-Dichloroacetone be stored and disposed of?
Why Safety with 1,3-Dichloroacetone Matters
A bottle of 1,3-dichloroacetone sitting in a lab isn’t just another chemical—it holds risks that demand respect. Having worked alongside seasoned chemists, I learned quickly that carelessness with reactive or toxic chemicals often leads to near-misses. 1,3-Dichloroacetone isn’t just harsh-smelling; inhaling its vapors, letting it splash on skin, or mishandling a leaking container opens the door for real harm. Poisoning, burns, or even fire breaks out in places where shortcuts trump care.
Facts matter here. 1,3-Dichloroacetone irritates eyes, skin, and lungs. Vapor can accumulate in poorly-ventilated storage rooms. In my experience, accidents usually come from simple errors: not labeling a bottle clearly after pouring off a portion, or leaving containers near heat or direct sunlight. All it takes is a cracked cap or broken bottle and suddenly everyone nearby deals with fumes and potential fires.
Practical Storage Tips from the Field
Every seasoned chemist knows: store volatile or unstable chemicals away from common traffic. 1,3-Dichloroacetone fits this rule. Use tightly sealed, chemically resistant containers—polyethylene, glass with PTFE-lined caps, or Teflon serve best. Never decant this chemical into an unmarked or food-use container.
Temperature swings break seals and stress bottles. Store this solvent in cool, dry, and shaded spots. Direct sunlight doesn’t just degrade it; it can also ramp up pressure inside the bottle. Fume hoods or ventilated chemical cabinets beat regular shelves every time.
In my own lab days, storing all organochlorines together helped contain risks. Separating oxidizing agents, acids, and bases lowered the odds of messy, sometimes explosive reactions. Anyone who’s ever had to mop up after a spill knows chemical segregation pays off.
Thinking Ahead: Disposal Done Right
Waste from 1,3-dichloroacetone doesn’t belong in the drain, dumpster, or regular trash. That’s not just policy—improper disposal damages plumbing, harms water tables, and violates the law. Watching a colleague call emergency response after a drain pour gone wrong drilled home: rules exist for a reason.
Diluting or neutralizing this chemical yourself makes trouble if you lack the right training and protective gear. Instead, collect all residues, spills, and contaminated items—gloves, pipette tips, or paper towels—in sealed, chemical-resistant waste containers. Label every container with its contents, concentration, and date. Hazardous waste firms handle pickup, incineration, or chemical treatment, far better than any DIY approach.
Education and Vigilance—Every Day
Rules around 1,3-dichloroacetone protect more than individual workers or students. A well-run lab posts emergency plans, makes spill kits available, and gives regular hazardous material refresher courses. Staff who feel comfortable asking for help spot errors before they snowball.
Most chemical safety boils down to discipline and routine: check seals, log inventory, keep storage lists updated, and avoid letting curiosity tempt you to throw waste in the sink. A single careless moment ruins reputations, damages health, and sometimes costs lives. Real accountability lifts everyone’s safety standards.
Making Safety Stick
As science marches on, chemical management keeps getting more complicated. 1,3-Dichloroacetone demands attention at both the lab bench and during disposal. Staying on top of the rules, sharing stories about mistakes, and holding each other to account keeps accidents rare and staff healthy. Every chemical—from the most benign to the most unpredictable—deserves the same tough respect.
What are the physical and chemical properties of 1,3-Dichloroacetone?
1,3-Dichloroacetone: Not Your Everyday Chemical
Imagine opening a bottle filled with a sharp, pungent smell—1,3-Dichloroacetone delivers just that. This compound keeps a low profile, mostly hanging out in labs. It makes eyes water and throats itch even with a quick whiff. While some see chemicals as harmless at a glance, this one asks for respect. 1,3-Dichloroacetone tends to feel oily upon contact, and drips as a colorless or very pale yellow liquid. Clarity matters here, since a chemical that looks harmless might push safety boundaries without too much warning.
Physical Properties: Handle with Care
Safety goggles mean a lot around this stuff. Its boiling point sits close to 150°C, and that’s higher than plain water but lower than many bulk chemicals. In the hands, 1,3-Dichloroacetone’s volatility stands out. Left open on a warm day, it can vaporize pretty quickly, spreading irritation in the air. This vapor can linger, so working under a fume hood isn’t just for show. It also tends to absorb moisture from the atmosphere, leaving traces of humidity wherever it’s stored without proper sealing.
The molecule doesn’t punch through plastics, since it isn’t especially corrosive. Still, skin doesn’t take kindly to even brief exposure. Folks on the front lines in chemistry have seen red, inflamed skin and sore hands—even from accidental splashes. Gloves matter. There’s no fancy transformation when it freezes; 1,3-Dichloroacetone drops solid at roughly -25°C. On the shelf, it looks simple. Up close, it means business.
Chemical Behavior: A Reactive Actor
What sits at the heart of this molecule are two chlorine atoms flanking a central ketone. This layout draws reactions like moths to a flame. That carbonyl group, basically the “business end” of the molecule, pulls in nucleophiles from all directions. Organic chemists rely on it when they want to set off a chain reaction. The chlorines on the ends make it even more reactive—start mixing, and substitutions or eliminations come easy, even in cold beakers.
Out in the wild world, 1,3-Dichloroacetone doesn’t last. It breaks down quickly in sunlight or under basic conditions. Storing it long-term outside a dark, cool spot doesn’t work. Light will split it into smaller, sometimes more dangerous fragments. In fact, incidents in poorly ventilated storerooms show that neglecting this detail fills the place with fumes—resulting in costly evacuations and headaches that nobody wants.
Big Picture: Handling and Hazards
Stories stick with those who’ve been rushed out of a building after a chemical spill. 1,3-Dichloroacetone doesn’t need gallons to cause trouble. A broken flask can spark evacuations if nobody pays attention to ventilation. Eye injuries or burn marks from handling slips through poor gloves remind researchers to treat simple-seeming liquids seriously.
Labeling, sealed glass, and clear protocols matter. Staff need quick access to eye washes and fresh air. Companies must teach both rookies and pros alike—not just about the hazards, but about what proper respect for these tools looks like. Secure storage away from acids or bases, clear PPE, and working under a modern hood keep research safe and productive.
Looking Ahead: Learning and Adapting
My own experience in research labs taught plenty about letting shortcuts slide. Chemicals with seemingly straightforward structures have surprising teeth. Staying safe means acknowledging that each property—boiling point, volatility, or chemical reactivity—carries real-world implications. With 1,3-Dichloroacetone, it pays to remember: just because it’s obscure doesn’t mean it won’t push limits. Safer labs don’t happen by luck. They start with respect, understanding, and hands-on training that brings the textbook to life.
| Names | |
| Preferred IUPAC name | 1,3-dichloropropan-2-one |
| Pronunciation | /ˌwʌn,θriː-daɪˌklɔːr.oʊ.əˈsiː.toʊn/ |
| Identifiers | |
| CAS Number | [534-07-6] |
| Beilstein Reference | 1209288 |
| ChEBI | CHEBI:82235 |
| ChEMBL | CHEMBL46308 |
| ChemSpider | 12630 |
| DrugBank | DB02108 |
| ECHA InfoCard | 03fc86d5-eda7-4047-bec4-3d2ac409385b |
| EC Number | 203-900-3 |
| Gmelin Reference | 1711325 |
| KEGG | C14616 |
| MeSH | D000197 |
| PubChem CID | 12311 |
| RTECS number | UC5950000 |
| UNII | 37MTC6NH1J |
| UN number | UN2511 |
| Properties | |
| Chemical formula | C3H4Cl2O |
| Molar mass | 137.00 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | penetrating |
| Density | 1.48 g/cm3 |
| Solubility in water | Soluble |
| log P | 0.67 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | 14.71 |
| Basicity (pKb) | -6.2 |
| Magnetic susceptibility (χ) | -48.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4660 |
| Viscosity | 2.542 cP (20°C) |
| Dipole moment | 2.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -204.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -725.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H302 + H315 + H319 + H332 + H335 |
| Precautionary statements | Precautionary statements of 1,3-Dichloroacetone: "P210, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Flash point | 88 °C |
| Autoignition temperature | 210 °C |
| Explosive limits | Explosive limits: 6–14% |
| Lethal dose or concentration | LD50 oral rat 205 mg/kg |
| LD50 (median dose) | LD50 (median dose): 270 mg/kg (rat, oral) |
| NIOSH | SN3625000 |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
1,3-Dibromoacetone 1,3-Diiodoacetone Chloroacetone Hexachloroacetone |

