1,3-Dichloro-2-Butene: Unpacking a Cornerstone of Organic Chemistry and Its Lasting Impact
Historical Development
1,3-Dichloro-2-butene has seen its reputation shaped by the story of chemical innovation across the last century. Chemists searching for efficient halogenated building blocks landed on this compound early as efforts ramped up to produce more complex chlorinated intermediates for synthetic rubber and specialty materials. Back in the era when large-scale chemical synthesis powered everything from materials science to crop protection, this compound appeared as one of many dialkenes featuring strategic halogen substitutions. As I look at how industry handled molecules like 1,3-dichloro-2-butene, it strikes me that each step—from bench-scale trials to pilot runs in the mid-20th century—drew on trial-and-error and the hard-won knowledge of process engineers battling unforeseen side reactions. The compound rode the wave of chemical manufacturing’s golden age, shaping processes that still underpin sectors like plastics and pharmaceuticals.
Product Overview
Anyone who has worked with halogenated olefins knows how quickly a lab can fill with their distinctive odors and the challenge of handling their reactive double bonds. 1,3-Dichloro-2-butene has carved out a place as a handy alkylating agent and a precursor to various chloro-organics. What stands out is how its structure—a four-carbon backbone, with chlorines marking each end of the diene—makes it a cornerstone for further modification. There’s a practical reason chemists keep turning back to it: the balance of stability and reactivity forms a sweet spot, whether you’re synthesizing intermediates for fine chemicals or exploring new polymerization reactions.
Physical & Chemical Properties
1,3-Dichloro-2-butene stands apart from many unsaturated hydrocarbons with its moderate volatility and clearly defined boiling point—usually falling in the low hundreds of degrees Celsius range. Its flammability and a boiling point shy of 160°C mean it doesn’t demand exotic apparatus to manipulate, but it will test your respect for lab safety. Chemically, the molecule’s alkene makes it a keen partner for electrophilic addition, yet those chlorines on the chain boost its resistance against random oxidation, giving it a modest edge over plain butenes. Experience shows that the pungent odor lingers if ventilation runs short, reinforcing the need to handle the compound inside a well-maintained fume hood. The density and refractive index get logged for regulatory and analytical work, though most chemists quickly learn the more tactile lessons: it forms a clear liquid that clings to glassware and splits cleanly from water but won’t play nicely with strong bases.
Technical Specifications & Labeling
Regulations have made technical labeling a non-optional detail for all chlorinated solvents like 1,3-dichloro-2-butene. Over time, product containers gathered color-coded symbols for toxicity, flammability, and environmental risk, partly in response to stricter workplace standards. Chemical purity sits at the center of many debates: trace contaminants, moisture sensitivity, and batch-to-batch consistency complicate quality control, whether for commercial batches or lab-sized bottles. I’ve seen how a mid-quality lot—loaded with side-product isomers or polymerized junk—can derail entire reaction schemes, forcing researchers to demand higher purity and more detailed specification sheets. These practical headaches have driven a steady improvement in labeling methods and traceability, keeping both regulatory teams and end-users happy.
Preparation Method
Ask around industrial chemistry circles and you’ll hear a few classic routes to 1,3-dichloro-2-butene, each with its devotees. Most often, production tracks through a stepwise addition of chlorine to butadiene, followed by careful distillation. This is no armchair exercise: reaction vessels need tight control over temperature and agitation to steer the process away from di- or poly-chlorinated byproducts. The ingenuity in practical plant design—especially with corrosion-resistant linings and automated dosing equipment—has reduced the risk of runaway chlorinations, but the process still calls for vigilance. Watching seasoned operators balance yields and purity with painstaking tweaks to feed rates always underscores that every large-scale synthesis owes far more to hands-on experience than textbook recipes.
Chemical Reactions & Modifications
In practicing organic chemistry, the allure of 1,3-dichloro-2-butene comes from its dual promise as both a substrate and a reactive intermediate. It cracks open a world of transformations; it undergoes nucleophilic substitutions at the chlorines, making it a flexible platform for building longer carbon chains or latching on heteroatoms. I’ve worked with research teams exploring epoxidation on the double bond—a reaction that delivers useful oxiranes—while allylic rearrangements tempt both the daring and the careful since controlling regioselectivity asks for a delicate touch. In synthesis workshops, more than a few discussions have returned to this molecule as a model compound for teaching alkene chemistry, since it combines manageable hazards with a repertoire of possible transformations.
Synonyms & Product Names
Chemists tend to pile up an arsenal of names for everyday molecules, and 1,3-dichloro-2-butene has worn its fair share of them—ranging from casual shorthand to IUPAC-verified names. You might hear “Dichlorobutene” in process settings or references to “1,3-DCB” on safety documents. Catalog numbers and database conventions shift between organizations, but what remains constant is the need for clarity. Call the compound the wrong name, and you risk confusing suppliers or—and I’ve seen this happen—grab the wrong chlorinated olefin off the shelf, derailing hours or days’ worth of planning.
Safety & Operational Standards
1,3-Dichloro-2-butene’s hazards extend beyond its acute toxicity. Those chlorines spell trouble if the compound escapes containment; it poses a risk to workers’ skin, eyes, and lungs and lingers in runoff as an environmental contaminant. Modern labs back up their protocols with extensive air monitoring, spill containment kits, and full PPE requirements. Over the years, training programs have taken on new urgency after high-profile incidents. Watching safety officers walk new staff through containment drills and emergency decontamination has made clear that organizational culture matters as much as regulatory checklists. I’ve also noticed insurers and compliance inspectors push for closed handling systems to reduce worker exposure, driving innovation in labware and bulk chemical logistics.
Application Area
Diving into the real-world uses, this compound’s track record in industrial chemistry goes deep. Large-scale syntheses for agricultural chemicals, specialty rubbers, and fluoropolymer production still lean on its presence as an intermediate. I’ve seen projects where 1,3-dichloro-2-butene becomes the key link—adding both structure and reactivity to downstream molecules. Laboratories focused on synthetic methodology treat it as a testbed for studying reaction mechanisms, drawing on its functional groups to probe everything from radical halogenations to base-driven rearrangements. Its usefulness as a cross-coupling partner keeps it relevant for those searching for creative paths to complex targets, especially those who need a balance between price and performance.
Research & Development
A closer look at current research trends shows that attention has shifted toward making handling safer and finding greener alternatives. Innovations in catalysis are extending the compound’s reach while sidestepping legacy issues like chlorinated byproduct waste and hazardous emissions. Academic groups tackling sustainable synthesis often circle back to 1,3-dichloro-2-butene as a yardstick: novel catalysts or greener solvents hit their stride when they deliver real gains with well-understood case studies. Industry-funded collaborations keep the momentum alive, investing in process upgrades or substitutions that dampen environmental burdens and slash operational costs. Reflecting on my own time supporting pilot-plant scaleups, I’ve noticed that outcomes depend as much on team coordination and buy-in as they do on the technical merits of new chemistries.
Toxicity Research
Regulatory science flags 1,3-dichloro-2-butene as a compound worth monitoring, given its acute toxicity and potential for chronic harm. Years of rodent studies and toxicological profiling point to skin and mucous membrane irritation and potential impacts on liver function, especially at higher concentrations or following accidental exposure. Real-world incidents—usually traced back to spills or improper disposal—underscore the need for mitigation plans rooted in science, not just compliance. The tension between practical chemical utility and the precautionary approach shows up in discussions about permissible exposure levels and waste regulations. I’ve talked with industrial hygienists who push for integrating more real-time monitoring and medical surveillance, hoping to catch subclinical effects early rather than wait for evidence to accumulate through hard experience.
Future Prospects
Looking forward, the conversation around 1,3-dichloro-2-butene isn’t about elimination but adaptation. Regulatory agencies and advocacy groups call for tighter emission limits and greener synthesis routes, opening space for innovation. Teams experimenting with alternative starting materials or enzymatic routes see an opportunity for phased transition—not just because it satisfies regulatory or public pressures, but because rethinking workflow can drive efficiency. In academic circles, research keeps building on foundational knowledge, sharpening techniques for detection, disposal, and remediation. Real progress, based on what I’ve seen, will come from tying technological change to practical incentives: safer plants, reduced waste, and transparent sharing of risk data so that future generations of chemists face fewer trade-offs between progress and precaution.
What is 1,3-Dichloro-2-Butene used for?
Understanding the Role of 1,3-Dichloro-2-Butene
Walk into any chemical plant and the unassuming names of compounds—names like 1,3-dichloro-2-butene—can hide pretty big jobs. In the world of manufacturing and industry, this chemical serves mostly as an intermediate for creating other compounds. It’s not something you’re likely to find in your garage or under the sink, but it does show up in the background of some everyday products.
The Manufacturing Backbone
Companies turn to 1,3-dichloro-2-butene because it can kick-start or speed up the creation of other chemicals. I remember talking to a manufacturer who pointed out that this substance fit right into their process for producing specialized polymers and rubber additives. Polymers built with these sorts of raw ingredients end up in things like hoses, seals, and automotive parts—places where resilience to heat and corrosion matter.
The value of such intermediates shows itself in the variety of jobs they fill. One example: producing fungicides and other protective chemicals for agriculture. Fields might get sprayed to hold off fungus or pest attacks, but the story of those sprays often starts in a drum holding 1,3-dichloro-2-butene hundreds or thousands of miles away. Moving from raw material to finished product involves stages full of chemistry, skill, and experience along the way.
Sourcing and Safety Concerns
Few folks are eager to handle this stuff without proper protective gear. Gloves and goggles are a must. Breathing in the fumes or touching skin can cause trouble. That fact shapes how plants store and move it—it’s packed away from workers, sealed off to limit accidental contact, and handled only in ventilated spaces. After spending a day shadowing a safety inspector at a mid-sized plant, I can vouch for the attention given to leaks and storage temperatures.
There are also big-picture questions to think about. The demand for 1,3-dichloro-2-butene matches the world’s appetite for things like synthetic rubbers and crop protection. As supply chains tighten, the industry faces pressure to cut down on waste and look for greener alternatives. Some researchers guess that shifting to renewable raw materials for industrial chemistry could offer new ways to build the same end products without as much reliance on these chlorinated intermediates.
The Search for Alternatives
Nobody likes a bad spill, and hazardous waste rules around chemicals like this keep getting stricter. Instead of waiting for regulations to force a change, some manufacturers have started pilot programs exploring less hazardous or more sustainable replacements. It isn’t easy to swap out a high-performing chemical, but companies stand to benefit from being early movers. If a new material matches the performance of 1,3-dichloro-2-butene and carries less risk, it could mean fewer accidents and an easier time meeting regulations down the road.
Looking Ahead
Industries that touch on 1,3-dichloro-2-butene—synthetic rubber, agriculture, specialty chemicals—aren’t likely to ditch such a useful tool overnight. The path forward might mean better containment, smarter recycling, or gradual adoption of safer reagents. The chemistry isn’t going away, but the ways the world handles it will keep changing, and that’s something worth watching on the factory floor and beyond.
What are the safety precautions when handling 1,3-Dichloro-2-Butene?
What’s At Stake With 1,3-Dichloro-2-Butene
I’ve spent enough time in labs to know that some chemicals ask for your respect. 1,3-Dichloro-2-butene sits in that category. Clear liquid, faint odor, looks ordinary—yet the risks it carries don’t sit on the surface. Anyone working with this substance faces health hazards that stretch from skin blisters to trouble breathing. NIOSH and the CDC both list it as hazardous; regular exposure can hurt the liver, lungs, and eyes. So it’s not just about compliance—it's about keeping people out of the emergency room.
Personal Protective Equipment: The Real Deal
Thinking gloves are enough gets folks in trouble. I’ve seen new lab techs trust cheap latex, but 1,3-dichloro-2-butene can slip through. Nitrile or heavy rubber gloves stand as the best barrier; even so, I double up for jobs that risk heavy splash. Face shields matter when pouring or transferring—chemical goggles only cover so much real estate. A solid lab coat, sturdy enough to stand up to accidental splashes, and shoes that actually cover the foot add another line of defense. Simple enough, but skipping basics is what lands you in trouble fast.
Fresh Air and Fume Hoods: Not Optional
Breathing in vapors brings the quickest harm. My rule: open containers or do any transfer work only under a chemical fume hood. Airflow needs regular checks—if you smell anything strange, stop the work right there. The risk isn’t just acute poisoning; long-term effects show up later, and nobody wants chronic lung problems because a vent didn’t suck hard enough one afternoon. For folks in bigger facilities, installing continuous air monitoring stations turns into money well spent. Most major incidents I’ve read about link back to poor ventilation rather than single mistakes.
Planning: Storage and Handling
Storing 1,3-dichloro-2-butene in just any cabinet can wreck a workspace. I use dedicated chemical cabinets with tight seals. Heat and sunlight speed up decomposition, and vapors build up explosively in warm rooms. The chemical sits best in cool, dry, out-of-the-way storage. Placing it next to acids, oxidizers, or even regular cleaning solutions risks unexpected reactions. I always keep a clear label on bottles—no exceptions. More than once I’ve seen someone pour waste into the wrong drum because helpful labels got peeled off.
Emergency Prep: Not Just a Box On the Wall
Most chemical safety manuals talk about eyewash stations and showers, but do folks actually check if they work each month? Accidents happen without notice, and the last thing anybody needs is to fumble around or learn too late that a valve jammed. Nearest exits, spill kits stocked with absorbents, and a fire extinguisher fit for chemical fires: these pieces keep panic short if the worst happens. Regular drills help turn scrambling into calm action. I always jot down what went right and what needs fixing after each practice run; it’s not about the rules, but about genuine readiness.
Learning From Mistakes, Mine and Others’
Sifting through incident reports or talking to lab veterans always brings up the same themes: overconfidence, shortcuts, and misplaced trust in subpar gear. Everyone works to get results, but cutting safety corners just doesn’t pay off in the long run. Sharing stories, keeping safety meetings short and focused, and updating protocols with each new lesson keeps folks sharp. Reading up on new guidance from OSHA and similar watchdogs stays just as important as remembering the steps for a synthesis reaction. Safety grows from habit, not from reminders on the wall.
What are the storage requirements for 1,3-Dichloro-2-Butene?
A Chemical that Demands Respect
1,3-Dichloro-2-butene looks simple on paper but its storage calls for some real attention. Not just because of how quickly it reacts with other chemicals, but because ignoring its risks can land people in trouble. Anyone who has spent time working with industrial chemicals knows that safety often comes down to small decisions made in the storage room. That feeling never really leaves, even decades after leaving the lab.
Why Proper Storage Matters
One wrong move with a product like this, workers can get badly hurt or property gets damaged. Let’s be clear: 1,3-Dichloro-2-butene gives off fumes you don’t want to breathe. It irritates skin, eyes, and lungs. The world knows what happened in factories where safe storage of volatile organics gets ignored—contamination, fires, emergency evacuations.
Physical and Chemical Properties That Drive Safe Storage
This chemical is a colorless to pale yellow liquid that boils at just over 108°C. If kept where temperatures fluctuate, containers can swell or rupture, especially in a sunlit warehouse or a poorly vented shed. 1,3-Dichloro-2-butene evaporates really fast, and its vapors are heavier than air, sinking low and spreading out along floors. The risk doesn’t stop at skin contact—fumes gather and light up easily. “Well-ventilated area” isn’t just a phrase on a safety sheet; it’s essential for anyone walking close by.
Workable Storage Solutions
Most experienced chemical managers store this liquid in tightly sealed, clearly labeled drums designed for halogenated solvents. Metal containers with good-quality gaskets work, unless they start to corrode. That’s why checking for leaks and rust often can’t be skipped. Temperature control means a lot here: stable, under 25°C, out of sunlight and far from anything that can ignite fumes. Locking cabinets separate from acids, alkalis, oxidizing agents, and sources of moisture make a big difference. It’s tempting to tuck everything into one storeroom, but one spill or reaction undoes years of safe habits.
Having worked in tool shops and chemistry labs, it is easy to say that every piece of protective equipment—gloves, goggles, aprons—pays off in the end. The real key comes from regular training and walking through spill response steps before anything goes wrong. Keep spill kits nearby and practice using them. Pouring and transferring this chemical warrant secondary containment trays. Fumes can set off alarms if ventilation isn't up to the task.
Preventing Dangers to Health and Environment
Regulations aren’t only there to trip you up. The OSHA and EPA rules, for instance, often stem from what’s gone wrong in the field. More than once, I’ve seen situations where lax storage invites fines—or worse, emergency cleanup. Keeping logs of inspections and knowing the shelf life of chemicals help prevent buildup and surprise reactions.
The Bottom Line
Storing 1,3-Dichloro-2-butene is demanding, but ignoring those demands costs far more than working smart from the outset. Anyone who has ever swept up after an accident knows that the true expense—and the real stress—comes after the fact, not before. Good containers, careful separation, protection from heat and sunlight, and solid ventilation keep people, property, and the business safe.
What is the chemical formula and structure of 1,3-Dichloro-2-Butene?
Understanding the Compound
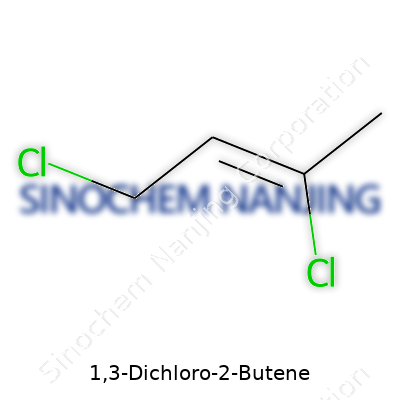
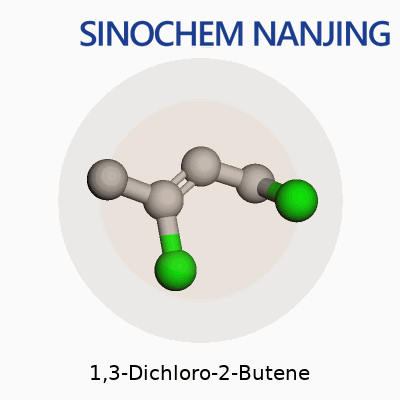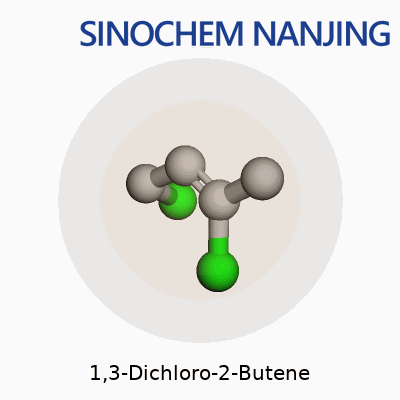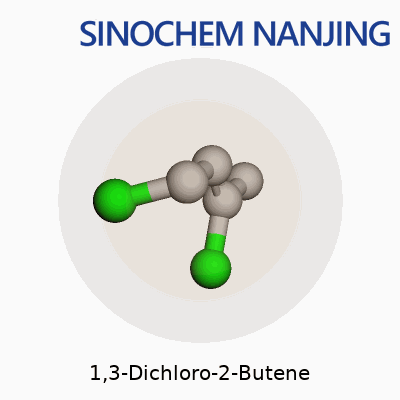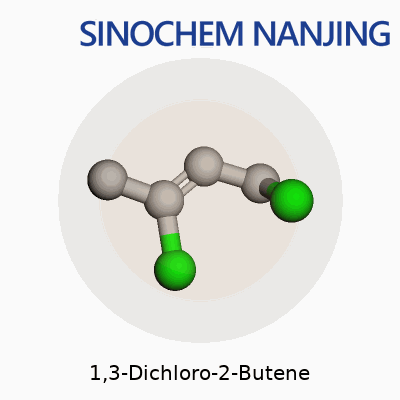
Anyone who has spent time around a university chem lab or even just read a little into industrial chemistry has likely come across simple chlorinated hydrocarbons. 1,3-Dichloro-2-butene stands out among those due to its structure and presence in organic synthesis. The molecular formula is C4H6Cl2. Breaking it down, that means it contains four carbons, six hydrogens, and two chlorine atoms. That stark simplicity gives rise to a molecule with very specific uses and some risks that should not be ignored.
Visualizing the Molecular Structure
In structural terms, 1,3-Dichloro-2-butene builds off the butene backbone format. The chain organizes like this: Cl–CH2–CH=CH–CH2–Cl. The double bond sits between carbons 2 and 3. Chlorine atoms attach to the 1 and 3 positions. Thinking back to crowded lab benches, someone always has to draw the Lewis structure before lunch. The two chlorine atoms, placed at opposite ends, completely change how the molecule reacts, especially under UV light or strong bases. Those halogen atoms drive reactions that turn out new compounds, sometimes with sharp smells that linger for hours on glassware.
The Role and Importance in Chemistry
1,3-Dichloro-2-butene comes up most often in organic synthesis and manufacturing. I’ve seen it show up as an intermediate for more advanced chemicals in the plastics and rubber industry. Its structure, with the double bond and chlorines, opens up quick reactions in both addition and elimination settings. Even in undergraduate labs, its reactivity under heat stands out—chlorines don’t just hang on the edges; they invite substitution, typically with nucleophiles. With chlorinated hydrocarbons, environmental persistence is never far from anyone’s mind, especially when drilling down on regulatory filings or safety data sheets.
Safety and Environmental Impact
Handling solvents and reagents packed with chlorines isn’t a light matter. Exposure risks multiply if there isn’t proper ventilation or if spills go unreported. Chlorinated compounds like this one tend to stick around in soil and water. Chlorination increases longevity, and without careful containment, compounds like 1,3-dichloro-2-butene can travel far beyond the lab. Chemists rely on fume hoods and gloves, but broader responses need to come from tighter waste control policies and real investments in neutralization technologies. Local communities benefit most from initiatives that force industries to reduce spillage and process waste for proper destruction instead of simple dumping or incineration.
Practical Solutions and Responsible Use
From my own time in industry, safer alternatives rarely get adopted unless rules or incentives push their use. Close tracking from the stockroom to disposal makes a huge difference. Regular audits, process upgrades, and even switching to less persistent chemicals can limit exposure. Researchers pushing green chemistry have started to roll out better methods—fewer chlorines means fewer headaches down the line. Investing in catalyst technologies that run reactions at lower energy or without halogens at all takes time, but every scale-up test that works pushes the industry to cleaner production. Keeping an eye on chemical structure helps predict both utility and risk, and understanding both sides of that coin makes for lasting, positive change in lab safety and environmental health.
Is 1,3-Dichloro-2-Butene hazardous to health or the environment?
Understanding 1,3-Dichloro-2-Butene
1,3-Dichloro-2-butene turns up in some chemical manufacturing processes. Simply put, it’s an industrial chemical that doesn’t show up in everyday products you grab off supermarket shelves. Some workers in the chemical sector deal with it more directly than the average person ever will.
Health Hazards: Worker Safety at the Forefront
Anyone breathing near 1,3-Dichloro-2-butene on the job faces some strong risks. The compound can irritate the respiratory system and eyes right away if a spill or accidental leak happens. Inhaling it even at low levels brings headaches, nausea, and dizziness. Lab studies also show it can mess with the liver and kidneys after prolonged exposure.
One thing that sticks out for me is how often workplace safety gets brushed aside until something goes wrong. There are plenty of chemicals out there like this one, but 1,3-dichloro-2-butene has flagged up in animal research as possibly increasing cancer risk. The International Agency for Research on Cancer (IARC) hasn’t made a public statement about this exact chemical, but its relatives have raised concern in toxicology communities for years. That’s enough to suggest thorough handling guidelines and regular checks matter here, even if the public rarely sees it.
Environmental Impact: Where Does It End Up?
If 1,3-dichloro-2-butene escapes into the environment, problems follow quickly. Spills or leaks can contaminate water and soil. Bacteria and sunlight break it down over time, but until nature does its job, fish and other wildlife could get a big hit. I once lived near a small industrial park—folks there worried about runoff into the river, too. Even a minor spill left a strong chemical smell in the air for days. If it can do that above ground, you have to wonder how much plants and aquatic life can tolerate.
The Regulatory Picture and Solutions
The United States Environmental Protection Agency keeps 1,3-dichloro-2-butene on lists for hazardous spills (under CERCLA/SARA regulations). This means reporting requirements kick in if an accident occurs, and cleanups have to follow federal guidance. This makes sense, since unchecked chemical releases can cause long-term damage. Legal limits for workplace air usually land below 1 part per million. That’s a clear red flag for anyone working around it.
Stronger protections can reduce these risks. Facilities can upgrade their ventilation and air monitoring. Workers can use certified respirators and chemical splash gear. Unexpected leaks need quick, well-rehearsed responses. Investing in closed-loop systems also helps prevent chemicals like this from getting out in the first place. I’ve seen smaller shops invest in safer storage as soon as someone close gets ill, but that’s a lesson best learned before trouble starts.
Moving Forward, Eyes Wide Open
Many dangerous chemicals played a role in economic progress. Still, companies using 1,3-dichloro-2-butene have a duty to tip the balance toward safety. It doesn’t belong anywhere near drinking water, playgrounds, or garden beds. Treating chemical hazards with respect—before anyone gets hurt—always costs less than waiting for cleanup bills or health problems down the line. Even if this chemical lives mostly behind factory doors, the consequences of carelessness reach far beyond them.
| Names | |
| Preferred IUPAC name | (E)-1,3-dichlorobut-2-ene |
| Other names |
1,3-Dichloro-2-butene 1,3-Dichloro-2-butylene Butene, 1,3-dichloro-2- 2-Butene, 1,3-dichloro- 1,3-Dichlorobutene-2 1,3-DCB |
| Pronunciation | /ˈwʌn ˌθri daɪˈklɔːroʊ tuː bjuːˈtiːn/ |
| Identifiers | |
| CAS Number | 928-52-3 |
| 3D model (JSmol) | `JSmol.loadInline("data/mol/1_3-dichloro-2-butene.mol")` |
| Beilstein Reference | 1361164 |
| ChEBI | CHEBI:51170 |
| ChEMBL | CHEMBL50459 |
| ChemSpider | 16246 |
| DrugBank | DB14088 |
| ECHA InfoCard | 03baae14-4ad1-4080-a8cb-285582250574 |
| EC Number | 203-445-0 |
| Gmelin Reference | 785 |
| KEGG | C19107 |
| MeSH | D004055 |
| PubChem CID | 7902 |
| RTECS number | EK1610000 |
| UNII | 6X6U1Y4X5Z |
| UN number | UN2322 |
| CompTox Dashboard (EPA) | DTXSID7020219 |
| Properties | |
| Chemical formula | C4H6Cl2 |
| Molar mass | Molar mass: 125.00 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Sweet odor |
| Density | 1.145 g/mL at 25 °C(lit.) |
| Solubility in water | slightly soluble |
| log P | 1.98 |
| Vapor pressure | 29 mmHg (20°C) |
| Acidity (pKa) | 5.46 |
| Basicity (pKb) | pKb = 8.23 |
| Magnetic susceptibility (χ) | -78.6·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.473 |
| Viscosity | 0.696 cP (25°C) |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 323.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -64.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4827.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Warning |
| Hazard statements | H315, H319, H332, H351 |
| Precautionary statements | P210, P261, P280, P301+P310, P304+P340, P305+P351+P338, P308+P313, P330, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-2 |
| Flash point | 50 °C (122 °F) |
| Autoignition temperature | 651°C |
| Explosive limits | Unknown. |
| Lethal dose or concentration | LD50 (oral, rat): 455 mg/kg |
| LD50 (median dose) | LD50 (median dose): 145 mg/kg (oral, rat) |
| NIOSH | NIOSH: KX9625000 |
| PEL (Permissible) | PEL (Permissible): 0.005 ppm |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | IDLH = 50 ppm |
| Related compounds | |
| Related compounds |
cis-1,4-Dichloro-2-butene trans-1,4-Dichloro-2-butene 1,2-Dichloroethene 1,3-Butadiene Vinyl chloride 1,2-Dichloroethane |

