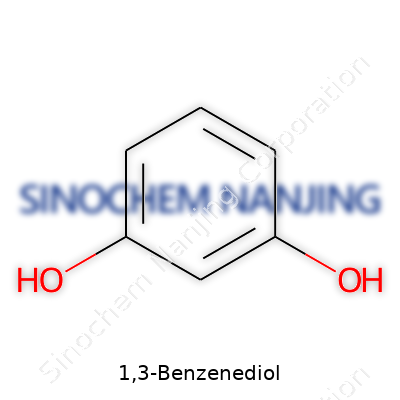
1,3-Benzenediol: A Closer Look at Its Past, Present, and Future
Historical Development
Chemical history always fascinates because it shows just how much effort goes into basic molecules that form the backbone of industries. 1,3-Benzenediol, more commonly called resorcinol, caught the attention of chemists in the late nineteenth century. Pioneering researchers discovered it while breaking down plant resins and exploring new synthetic dyes. Back then, getting pure resorcinol took hours of distillation and hand-crafted benches. Today, refineries churn out this compound in tonnage. Its discovery wasn't just a moment in a quiet laboratory — it marked an early step in the rise of aromatic chemistry and led to a flood of research that spilled into medicine, rubber production, and dyes. It's remarkable to think about the way simple procedures in cramped old labs can lead to global industries decades down the road.
Product Overview
1,3-Benzenediol holds a distinctive place in both industrial processes and daily consumer products. Whether it shows up in topical medications as an antiseptic or in adhesive formulations, the molecule travels far beyond laboratories. It sits in antiseptics, hair dyes, UV stabilizers, and other specialty chemicals. Chemists appreciate its versatility. Its reactivity comes from those two hydroxyl groups linked to the benzene ring, opening countless possibilities for further transformations. Some may walk right past it in a hardware or pharmacy aisle, not realizing they've just glanced at a molecule supporting whole manufacturing sectors.
Physical & Chemical Properties
The physical offering sets 1,3-Benzenediol apart. It appears as a white, needle-like crystalline powder, with a sweetish odor faintly hinting at its origins in natural resins. Its melting point lands around 110 degrees Celsius, which means it rides a fine balance between stability and processibility. The molecule dissolves easily in hot water and alcohol but resists mixing with nonpolar solvents. Chemically, the two hydroxyl groups on alternating carbons allow for reactions that form links, dyes, and polymers. These properties enable broad applications — few molecules of its size pull this kind of industrial weight.
Technical Specifications & Labeling
Talk about technical details, and the discussion quickly turns to purity, granulation, and safety labels. High-purity resorcinol features in pharmaceutical-grade or electronic materials, where even tiny contaminants change results. Industrial batches lean more towards robust grades, as rubber or adhesive manufacturers may put different demands on purity. Safety adherence matters, not just for the labs but anywhere it is handled. Proper labeling with hazard information, storage instructions, and usage protocols ensures that chemists and plant workers have the information to work safely and productively. Too many ignore these steps until something goes wrong — a lesson any experienced hand has learned at some point.
Preparation Method
Production began with extraction from plants, but industrial needs forced a switch long ago to synthetic routes. Today’s methods focus on fusing benzene derivatives with alkali under high temperature, breaking the aromatic ring in tightly controlled conditions. There’s a nod to green chemistry bubbling up, with research looking at enzymatic pathways and alternative feedstocks, but nothing in widespread use yet. Sourcing and energy cost balances affect how big players approach production. Tight oversight and innovation in these synthetic routes could produce more with fewer emissions, something the chemical industry debates every year.
Chemical Reactions & Modifications
Discussing the chemistry of 1,3-Benzenediol brings up classic organic techniques. The two hydroxyl groups lend themselves to further substitutions, such as alkylation, acylation, or forming ethers and esters. In polymer chemistry, these reactive centers drive cross-linking needed for making adhesives and resins. On the pharmaceutical front, ring modifications introduce bioactivity, creating new molecules for topical or systemic drugs. Those familiar with the inner workings of an R&D lab know that modifying a single hydroxyl group can make the difference between a household ink and a promising medicine.
Synonyms & Product Names
Names for this molecule appear all over chemical and product documentation: resorcinol, m-dihydroxybenzene, 1,3-dihydroxybenzene. These synonyms aren't just a matter of semantics; they signal context, origin, and often regulatory registration. Anyone who has scanned regulatory documentation knows the headache poorly managed synonyms can cause. A compound under one name may dodge recognized safety assessments under another. Consistent and transparent naming matters, especially for safety, sourcing, and environmental reporting.
Safety & Operational Standards
Anyone who’s worked with 1,3-Benzenediol recognizes the careful steps necessary to keep workers safe. Skin contact poses risks, and inhaled dust can cause irritation or allergic reactions. Facilities install fume hoods, train employees on appropriate handling, and keep rigorous documentation for audits. Regulatory agencies — from local governments up to global watchdogs — issue guidelines covering storage, personal protective equipment (PPE), spill management, and disposal. In my experience overseeing operations, those regulations only protect when treated as routine, not as paperwork hurdles. Failures often follow lapses in culture, not protocol, so building a culture of safety is critical.
Application Area
Resorcinol finds everyday application from the unseen glue in plywood to the reflective coatings inside sunglasses. Its antiseptic nature brings it into acne and eczema creams on pharmacy shelves. Some of its biggest impacts happen off-stage: strengthening the bonds in car tires, acting as a chemical intermediate that helps make dyes more vivid, and stabilizing plastics from ultraviolet light. With each new use, chemistry and consumer safety demands evolve, driving both new formulations and updated safety testing.
Research & Development
R&D pushes 1,3-Benzenediol into fresh territory year after year. Researchers seek alternatives to phenol or formaldehyde, hoping this molecule can bridge gaps with safer or greener processes. Polymer scientists create advanced adhesives, exploring how changes to benzenediol's structure affect performance under stress or heat. Pharmaceutical chemists revisit its old antiseptic routes, searching for new ways to minimize irritation or boost efficacy. Even after more than a century, labs still chase the edges of what resorcinol can do, proving no molecule stays static in a lively research landscape.
Toxicity Research
Any commentary on 1,3-Benzenediol must address toxicity with honesty and care. Studies document skin irritation, allergic responses, and concerns about long-term exposure, especially in industries with daily contact. Governments set strict workplace limits, and consumer health agencies restrict concentrations in beauty products. Recent laboratory research examines metabolism and possible environmental persistence, recognizing the need for updated risk assessments. My experience has shown that early warnings, routine monitoring, and prompt medical intervention can make all the difference in workplaces. Transparency from companies and rigorous public health reporting turn toxicity management into a matter of shared responsibility — not just box-ticking.
Future Prospects
Looking ahead, 1,3-Benzenediol balances between dependable industry mainstay and a candidate for cleaner, safer manufacturing. Advances in green synthesis methods could one day curb environmental impact if chemistry delivers on its promise. Consumer expectations are changing, with calls for transparent sourcing, responsible supply chains, and ongoing toxicology reassessment. Each year, researchers discover new molecular tweaks that yield fresh uses in medicine, construction, and electronics. In the decades to come, progress for this unassuming aromatic ring may come less from radical discovery and more from steady, thoughtful reengineering to meet new societal and environmental standards. Those of us following chemical industry trends see both the complexity and promise embedded in this molecule's story.
What is 1,3-Benzenediol used for?
Everyday Applications Down to Chemistry
The chemical 1,3-Benzenediol, often called resorcinol, rarely grabs headlines. Yet, it quietly shapes everyday products and industrial practices. It’s a colorless compound with practical value for chemists, manufacturers, and in some cases, doctors. My experience working near labs taught me how chemicals like this go unnoticed until someone’s focus lands on what makes shampoo foam, glue durable, or even hair dye last.
Hair Dyes, Adhesives, and Rubber Goods
The hair salon on my block lists dozens of dye shades. Many of those colors rely on resorcinol. It works as a building block in hair color formulations due to its reactivity with other ingredients, delivering stable, long-lasting color. According to the European Commission’s Scientific Committee on Consumer Safety, proper regulation ensures doses stay safe for repeated use. Some folks think only the pigment matters, but the underlying chemistry makes those colors stick and keeps them safe for skin contact.
Resorcinol appears in adhesives—and not only the stuff in school lockers. It’s a key component for bonding wood in outdoor construction, like bridges and boats, where moisture breaks down ordinary glues. The durable bonds come from the way resorcinol reacts, forming cross-links that resist heat and water. Manufacturers keep turning to it for projects that require trust in every seam.
The automotive industry relies on resorcinol-derived chemicals to get tires gripping the road. The substance helps bond rubber to reinforcing textile cords, ensuring that tires last through years of potholes and hot pavement. Many tire failures trace back to poor bonding, so any improvement becomes both a safety benefit and an economic relief.
Pharmaceutical and Medical Impact
Doctors sometimes reach for formulas containing resorcinol. In dermatology, creams with small concentrations help peel rough skin or treat acne. The substance acts as a mild antiseptic and keratolytic agent, thinning thick skin patches and easing symptoms. Medical guidance matters here because too much can irritate or damage tissue, but in controlled doses, it has helped patients feel more comfortable in their own skin.
Photography, Dyes, and Other Industries
Before the digital camera, developing film captured memories for generations. Resorcinol enters the picture in photographic developing agents. Even today, niche photographers and researchers using traditional methods depend on stable results made possible by chemical agents like this.
On a broader level, industries use resorcinol to make dyes for textiles and plastics. Everyday products like blue jeans, toys, and food packaging might carry colors made more stable by resorcinol chemistry. Every year, thousands of tons get shipped for such uses, fulfilling demand from factories across the globe.
Risks and Responsible Handling
Not every application comes without concerns. High amounts of resorcinol can irritate eyes, skin, or even disrupt thyroid function if mishandled. Agencies like the US Environmental Protection Agency monitor production and disposal, aiming to keep workplace air safe and water supplies clean. Training workers in safety protocols, regular air testing, and strict waste treatment help minimize health risks from this substance. My uncle, who spent years in a chemical plant, emphasized the importance of protective gear and following routines that protect everyone’s well-being.
Looking Ahead
The search for greener chemistry continues. Some manufacturers seek alternatives, yet resorcinol’s properties haven’t been matched in every case. Improved recycling, lower exposures, and tighter regulations lead the way to safer workplaces. The drive for safety in both product and process shapes how this overlooked chemical fits into daily life, from the bottle of lotion to the tires on our cars.
Is 1,3-Benzenediol safe to handle?
Looking at the Facts
In laboratories and some industrial settings, 1,3-Benzenediol turns up far more often than most folks might expect. Chemists recognize it by its other name: resorcinol. This white, powdery compound comes from benzene, and it shows up everywhere from hair dye formulas to acne creams, antiseptics, rubber production, and even photography chemicals. Despite its many uses, not everyone handling it realizes the risks that come with direct exposure.
Everyday Exposure and Real World Risks
Ask anyone who has worked behind a pharmacy counter or in a chemical plant. Handling resorcinol means opening bags or bottles, sometimes spilling powder, mixing solutions, cleaning up. Skin contact happens, and so do occasional splashes to the eyes. Even hobbyists tinkering at home can underestimate what they’re working with. It’s a strong irritant—touching your skin leaves redness, maybe a rash or peeling. Getting it in your eyes stings and can cause lasting trouble without quick and proper action.
Breathing in dust becomes a real concern with resorcinol. Some years back, I worked in a shop handling powdered chemicals, including resorcinol. People new to the work often underestimated the importance of a tight-fitting dust mask. One bad bout of coughing from inhaling chemical dust was enough to drive the point home. Dizziness, headache, and nausea can follow significant inhalation. Studies have shown that, in rare cases, larger doses can affect heart function and cause thyroid problems if exposure goes unchecked.
Resorcinol: Household Staples, Unseen Hazards
Many over-the-counter skin treatments, especially ones designed for acne or warts, list resorcinol on the label. Most of these creams rely on very small concentrations, making them safe for typical use as the skin limits how much is absorbed. The U.S. Food and Drug Administration imposes clear limits on allowable amounts. Safety testing covers short-term and long-term use, especially when products get sold for use on broken or inflamed skin.
Issues start cropping up with people who use these products beyond the recommended amounts. Mixing similar compounds, layering multiple skin treatments, or making homemade remedies with bulk resorcinol pushes the boundaries of safety. Ingesting resorcinol brings a whole new level of danger, with symptoms like vomiting, rapid heartbeat, and even, in severe poisonings, convulsions.
Safer Handling: Lessons from Experience
After years handling both common and specialty chemicals, I found the simplest habits worked best for staying safe. Gloves keep skin safe, goggles shield the eyes, and a dust mask keeps powder out of your lungs. Washing up after handling and working in a space with good airflow keep a lot of problems at bay. Never storing food or drinks near the work area makes accidental ingestion nearly impossible.
Factories and labs can go a step further: training every worker, from entry-level to supervisors. Safety data sheets should never gather dust; I learned most mistakes happen when people skim those details. Encouraging questions and regular safety reviews stop misuse before it starts.
Solutions for Safer Workplaces and Homes
Companies can lower risk by using ready-to-use, pre-measured packages. Home users should only ever buy the amount intended for a specific project. Clear labeling stands out as the simplest fix: bold instructions and warnings keep everyone on track.
People take extra precautions for things like bleach or oven cleaner—resorcinol deserves at least that much respect. Just a bit more awareness, some teamwork, and practical handling habits can turn a risky substance into a manageable part of everyday work and home life.
What is the chemical formula of 1,3-Benzenediol?
The Formula That Connects Science and Everyday Life
You don’t need an advanced chemistry degree to run into molecules every single day. 1,3-Benzenediol, known in some circles as resorcinol, is one compound playing a bigger part in daily life than most folks realize. The straightforward chemical formula for 1,3-Benzenediol is C6H6O2. That’s six carbon atoms, six hydrogens, and two oxygen atoms woven together in a specific dance—a benzene ring with two hydroxyl groups set at the 1 and 3 positions.
Grasping the Structure: More Than Letters and Numbers
This is not just memorizing a formula for a test or rattling off trivia at a pub quiz. The numeric layout tells a bigger story. The structure behind those letters brings out 1,3-Benzenediol’s unique personality. The arrangement of two –OH groups at positions 1 and 3 opens up a host of chemical possibilities. For example, as someone who’s dabbled in pharmaceutical chemistry, I’ve seen resorcinol show up in acne treatments, hair dyes, and even as an intermediate in making other medicines. The ability for this compound to bond, react, and transform makes it important beyond the lab.
Real-World Impact and Safety
The chemical makeup of 1,3-Benzenediol shapes its physical properties: it looks like white crystals and dissolves well in water and alcohols. This solubility allows it to work effectively in topical creams and solutions for skin conditions. But what matters just as much is recognizing the risks. Chemical exposure is not a distant worry; it shows up in real workplaces and sometimes even at home. Inhaling dust or letting it sit on the skin too long can cause irritation. The World Health Organization and the US National Institute for Occupational Safety and Health both recommend using gloves and goggles, making sure any storage area stays well ventilated.
Building Trust Through Transparency
The digital age makes information about chemicals much more accessible. Websites like the US National Library of Medicine’s PubChem database lay out everything from chemical structure to environmental hazards and safe handling directions. For someone working with industrial chemicals or even parents looking into ingredients in skin creams, transparency matters. Studies and health guidelines don’t always use plain words. There's a need for organizations and experts to translate science into clear, honest communication. That means better consumer choices and fewer workplace accidents.
Seeking Balance: Benefits and Precautions
1,3-Benzenediol isn’t just another chemical name on a bottle. Its formula, C6H6O2, shapes a tool that handles tough skin conditions, helps formulate adhesives, and moves research forward. While scientists focus on synthesizing purer forms or seeking even safer alternatives, the current challenge is education. Everyone from researchers and manufacturers to everyday consumers plays a part. Reading labels, asking questions, and following safety protocols keeps people in charge of their own well-being.
Practical Solutions for the Future
Investing in safety training makes sense on the shop floor. Research teams investing in less toxic alternatives to 1,3-Benzenediol keep the benefits without as much risk. Wider adoption of clearer labeling and data sharing can help bridge the gap between insider knowledge and public trust. C6H6O2 may look like just another formula, but it shows what’s possible when scientific know-how connects with responsible choices.
How should 1,3-Benzenediol be stored?
Looking at Storage With Common Sense
Plenty of folks who work in labs know their shelves can get cluttered fast. 1,3-Benzenediol, also called resorcinol, is one bottle that deserves a little respect. This white crystalline solid isn’t somebody’s backyard chemistry set—it often shows up in antiseptics, dyes, and some high-strength adhesives. Storing it right keeps everyone safe, saves money, and avoids waste.
A Warm Place Spells Trouble
Temperatures behind locked doors in storerooms creep up easily, especially in summer. If you leave resorcinol in the sun or next to heat sources, it cake ups, gets more volatile, and may even start to break down. Those changes invite risk. Keeping this chemical in a cool, well-ventilated space—think below 25°C (77°F)—means it’ll stay stable and won’t react before its time. That space should protect from direct sunlight, which can not only accelerate decomposition but can also mess with purity. A steel cabinet away from any furnace works better than any fancy equipment for most labs.
Moisture Isn’t Just an Annoyance
Humidity isn’t your friend. It dissolves these crystals, turning them sticky and miserable. This compromises both shelf life and safety. If moisture gets in, resorcinol can react with metals and even with some packaging. I’ve seen people try to save space by using makeshift lids, coffee cans, or plastic wraps—that shortcut often leads to disaster. Resealing with the factory cap, using a desiccant, and checking that there’s zero water nearby helps keep it from lumping or turning brownish. Even standard air exposure over a weekend can ruin an open jar. Simple choices—like using tight lids and storing away from sinks—keep your supply out of trouble.
Why Not All Shelves Are Equal
It can be tempting to group bottles together on top shelves. If you do that with resorcinol, there’s a chance of cross-contamination with acids or strong oxidizers. This is more than a theoretical risk—any spilled acid or chlorine bleach can start a dangerous reaction. Standard protocols recommend a designated chemical storage area, not a shared shelf in a cleaning closet. Color-coded bins and clear labeling save confusion in a pinch. Staff in larger labs train for these habits, but smaller shops sometimes skip them and pay the price in ruined product—or worse, workplace accidents.
Thinking About Regulations and Health
OSHA and the CDC both warn about the health risks of handling resorcinol wrong. Inhalation or skin contact causes problems like irritation or even chemical burns. You benefit from storing the solid in containers built for chemicals—glass with tight-sealing lids takes the crown. Avoid mason jars or food storage. If a spill happens, you want proper absorbents close by, and a well-maintained eyewash station within a few steps. You don’t have to work in a big pharma plant for these rules to matter. Even a small research shop must keep up with Safety Data Sheet guidelines and train every technician before they start touching chemicals.
Improvement Starts With Good Habits
Safe storage turns into a habit when people see the payoff. Nobody enjoys the paperwork or costs after a shelf collapses or a sample spoils. Investing in a sturdy cabinet, running regular checks for leaks, and posting friendly reminders about chemical compatibility may seem basic. Over time, these moves build a workplace where the risks stay low and folks get to focus on research, not cleaning up accidents. In the long run, storing 1,3-Benzenediol right mirrors how seriously you take your own health and the well-being of the people who work alongside you.
What are the hazards associated with 1,3-Benzenediol?
What 1,3-Benzenediol Means for Workers and the Public
1,3-Benzenediol, often called resorcinol, shows up in plenty of places. You might run across it if you work in a chemical plant, a hair dye factory, or even down the supply line for pharmaceuticals. This stuff packs more risk than people often realize. Skin exposure, inhalation, or accidental swallowing all bring up real concerns for anyone handling it.
Health Risks Right Up Close
Anyone working with this chemical notices the skin irritation first. Resorcinol can burn and cause redness on contact. Some of the folks I’ve talked to on production floors say they learned the hard way how gloves are not optional. Just a day without good protection, and they wound up with rashes that stuck around far longer than expected. The risk climbs if the chemical gets on broken skin, opening the door for it to be absorbed more quickly into the body.
Breathing in dust or vapors also matters. Shortness of breath, coughing, and a sore throat often come up right after exposure. In closed environments, fumes build up, and workers sometimes face dizzy spells or headaches. Over the years, studies have pointed out repeated exposure might lead to problems such as thyroid disruption and even changes in blood counts. The high water solubility makes it easier for the chemical to move around, increasing the risk indoors, in manufacturing lines, and even during transportation accidents.
Environmental Trouble Spots
Spills and leaks create bigger emergencies. Resorcinol dissolves quickly in water, threatening ecosystems if it gets into rivers or groundwater. Once it’s out, fish and microorganisms struggle. High concentrations kill off organisms or at least slow down natural decomposition in water bodies. These aren’t just lab findings — some small chemical accidents in rural facilities have forced local authorities to warn people off water sources for weeks. Clean-up, from all I’ve seen, never happens fast and doesn’t always undo all the damage.
Handling Risk in Real Life
Many safety consultants I’ve worked with stress training and ongoing reminders. Signage, personal protective equipment, and strong ventilation take real effort and money, but skimping leads to the kind of hospital visits that bring lawsuits. OSHA’s frequent checks in chemical plants push for transparency, but company cultures make a difference too. Where leadership values fast, safe response to spills, people trust each other to report near-misses and correct problems before they spread.
Emergency response teams need more than a plan on paper. Regular drills, chemical spill kits, and quick access to first aid should be daily realities. For the public, better labeling and community information stop panic during transport accidents. Sometimes, simple changes like switching to closed systems for mixing or installing better leak detectors have cut down accident rates at sites I’ve visited by huge margins.
Looking for Safer Alternatives
Some companies switch to greener materials, though the process takes time. Research keeps pushing into less-toxic substitutes for resorcinol, and some labs already test new adhesives. Change moves slowly, but regulatory pressure and real-life stories keep the industry shifting toward safer ground. Until these safer options become the norm, taking hazards seriously with solid, everyday precautions keeps workers, communities, and the environment healthier.
| Names | |
| Preferred IUPAC name | benzene-1,3-diol |
| Other names |
Resorcinol m-Dihydroxybenzene 1,3-Dihydroxybenzene m-Benzenediol |
| Pronunciation | /waɪˈdiːn.dɪˌɒl/ |
| Identifiers | |
| CAS Number | 108-46-3 |
| Beilstein Reference | 136173 |
| ChEBI | CHEBI:17606 |
| ChEMBL | CHEMBL1407 |
| ChemSpider | 682 |
| DrugBank | DB01936 |
| ECHA InfoCard | 00c38b3a-8a9b-4e9b-950a-f84571bf918d |
| EC Number | 1.10.3.2 |
| Gmelin Reference | 82262 |
| KEGG | C00137 |
| MeSH | D002907 |
| PubChem CID | 136 |
| RTECS number | DC3325000 |
| UNII | 203D347Q3R |
| UN number | UN2876 |
| CompTox Dashboard (EPA) | DTXSID2021110 |
| Properties | |
| Chemical formula | C6H6O2 |
| Molar mass | 110.11 g/mol |
| Appearance | White solid |
| Odor | odorless |
| Density | 1.3 g/cm³ |
| Solubility in water | soluble |
| log P | 1.3 |
| Vapor pressure | 0.001 mmHg (25°C) |
| Acidity (pKa) | 9.85 |
| Basicity (pKb) | 10.58 |
| Magnetic susceptibility (χ) | -46.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.569 |
| Viscosity | 7.4 cP (25°C) |
| Dipole moment | 2.34 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -242.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –3240 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 138 °C |
| Autoignition temperature | 548 °C |
| Explosive limits | 1.8–9.2% |
| Lethal dose or concentration | LD50 oral rat 2830 mg/kg |
| LD50 (median dose) | LD50 (median dose): 330 mg/kg (oral, rat) |
| NIOSH | KW2975000 |
| PEL (Permissible) | No PEL established |
| REL (Recommended) | 0.02 |
| IDLH (Immediate danger) | 100 mg/m3 |

