Digging into 1,3,5-Trinitrobenzene: More Than Just a Chemical Compound
Tracing Roots: A Chemical Legacy with Explosive Twists
Long before glossy publications and laptops filled with chemical equations, folks in dimly lit labs discovered how powerful certain molecules could be. 1,3,5-Trinitrobenzene, or TNB to chemists, entered the scene in the late 19th century, drawing attention from the scientific community. Back then, curiosity drove early researchers to explore this molecule, eager to squeeze every bit of insight out of its unusual properties. Its origin story is tied to the push for stronger explosives and more reliable detection reagents. History associates TNB with an era where military demands and analytical chemistry walked hand-in-hand. As the world industrialized, a demand for controlled power arose, and trinitro compounds rode that wave. Over time, lab notebooks became digital databases, but the hunt for better explosive ingredients and precise standards kept TNB in the conversation, serving as both a high-energy chemical and a yardstick in analytical work.
What Makes It Special: TNB from a Hands-On Perspective
Chemists often cite TNB for its yellowish crystalline nature—a clue that it doesn’t blend into the crowd. Scoop a bit out of a dish and notice the powder’s distinct feel and muted shimmer. Take a whiff and the sharp chemical odor reminds you why protective gear matters. TNB’s real value comes out when pushing it through a reaction or prepping it for testing. Its solid state and low volatility mean it doesn’t vanish from sight during work, a quality appreciated in busy labs. The compound doesn’t absorb much water from the air, so flasks with TNB don’t get sticky or clumpy. TNB’s role in labs echoes in the way it's chosen as a standard for calibrating devices, proof that consistent physical traits trump theoretical properties in real lab days. Its solubility features—dissolving better in acetone or ethanol than water—set the stage for efficient extraction, separation, and applications in testing explosives or environmental residues.
Technical Details That Actually Matter
Plain numbers like melting point or density mean more when you’ve seen TNB refuse to melt until over 120°C or when a spill runs like a fine sandpile instead of a sticky mess. The trinitro groups on its ring structure do a lot of heavy lifting—turning up electron-withdrawing tendencies, pushing the chemistry toward energetic reactions, and explaining why TNB is stable enough to handle, yet reactive enough for military and detection uses. These three nitro attachments don’t just tweak physical traits; they shape all the technical decisions behind storage, handling, and end-uses. In real work, the reliability of melting, boiling, and combustibility parameters means fewer surprises and smoother synthesis runs. People in the field respect those numbers not as trivia, but as signposts to safer work and dependable results.
Crafting TNB: Making Safe Choices in Preparation
Anyone who’s spent time in synthetic organic chemistry remembers tedious prep work. TNB follows a direct line—take a benzene ring and introduce three nitro groups in a controlled way. This process, industrial or lab-scale, always calls for strong acids like nitric and sulfuric. Safety protocols get drummed in early for a reason: ignore ventilation or fumble with acid mixtures and the day ends badly. A solid workflow avoids temperature spikes and uncontrolled reactions, keeping the trinitro formation in check. It’s a balancing act: too aggressive, and you get runaway heat; too gentle, and yields take a hit. The method rewards patience, precision, and knowledge of explosive hazards. Clean, staged additions and diligent washing at the end of synthesis leave you with a crystalline yellow solid ready for further tests or reactions.
Getting Reactive: What TNB Does Beyond the Basics
Once in hand, TNB’s chemical utility takes center stage. Those three nitro groups make it electron-poor, pushing TNB toward reactions as a solid electrophilic agent. In research, the compound acts in reduction reactions, forming aminobenzene derivatives if treated gently. Some labs turn TNB into sensors, leveraging color changes in response to metal ions or environmental samples. This versatility comes from the nitro group’s willingness to participate—swapping, being reduced, or adding more complexity. People see TNB as a robust backbone in creating standards for trace detection, especially in environmental and forensic science. In explosives chemistry, researchers value its role as a precursor or component, sometimes blending it with other energetic molecules to test detonation strength and stability.
A String of Names: Navigating Synonyms and Aliases
Walk into a library or a modern research database and TNB hides under several names. You might spot it as trinitro-1,3,5-benzene or sym-trinitrobenzene. The “sym” points to the symmetry of the molecule—a shortcut for chemists who care about positions on the benzene ring. Old textbooks sometimes slip in alternative notations or local trade names, a leftover from region-specific manufacturing. Knowing these aliases isn’t trivia work—it helps track patents, unearth older research, and avoid mix-ups in purchases. For researchers toggling between languages or regulatory databases, that string of synonyms links together decades of experiments and industrial practice.
Staying Safe: Real-World Practices, Not Just Labels
Lab safety doesn’t mean tiptoeing around molecules—it’s about forming habits that anticipate trouble. TNB earns respect for its stability, but nobody lets their guard down, especially around heat, sparks, or static. Storing TNB means dry, cool spaces, away from strong bases or amines that could trigger unwanted reactions. Shields, gloves, and eye protection become second-nature after a few close calls or seeing what high-energy compounds can do. Safety data sheets get scanned regularly, but personal experience hammers home the importance of routine checks, proper labeling, and avoiding shortcuts. Real safety culture grows out of lived experience, reinforced by every researcher who’s seen a careless moment turn into an emergency.
Where TNB Gets Used: Explosives, Sensors, and Science
TNB’s claim to fame lives in its role as an explosive ingredient, especially in military formulas and specialized demolition work. It doesn’t grab headlines like TNT, but professionals pick TNB for specific energy requirements and detonation profiles. Analytical chemists put TNB to work as a calibration standard, its structure helping track and quantify nitro aromatic compounds in soil, water, and forensic samples. Environmental science circles value TNB for its consistent signal in residue detection, supporting cleanup efforts near munitions sites. People experimenting with new sensors use TNB as a test case, checking how well devices can pick up trace explosives, teaching machines to spot threats more reliably in crowded airports or at border checks.
Pushing Boundaries: Research and Development Today
Innovation in chemistry happens when staples like TNB spark fresh questions. Lab teams now probe TNB’s behavior under extreme conditions, searching for materials with higher energy yet stable storage and safer handling. Some work focuses on modifying the molecule, swapping out nitro groups for other substituents, testing whether the stability or reactivity tips more favorably. Advanced analytics track every byproduct, pushing the envelope on detection and quantification. Computational chemistry helps map out reaction paths, explaining why TNB resists some processes and leaps into others. By building on TNB’s reliable core, researchers edge closer to cleaner, safer, and more effective explosives or detection agents.
Toxicity: The Hard Lessons from Real Exposure
No credible chemist takes TNB toxicity lightly—its nitro groups mean trouble if inhaled, ingested, or forgotten on open bench tops. Animal testing and accidental exposures have shown real risks: blood disorders, organ stress, or lingering residues in water systems near production sites. Long-term exposure links to chronic health effects, pushing workplace standards to limit airborne dust and calling for robust cleanup routines. Environmental monitoring tracks TNB in soil and water, especially near older military sites, measuring its movement and persistence. Lessons learned from these studies push for better protective gear, improved filtration, and stronger containment during every stage from synthesis to disposal.
Peering Ahead: Opportunities and Challenges on the Horizon
Looking toward the future, TNB’s role evolves with technology and policy. Efforts ramp up to produce safer energetic compounds, with TNB providing a benchmark for comparison. Engineers design sensors that spot TNB traces faster and with less human oversight, expanding security in crowded public spaces. Environmental science urges better methods to track and clean up TNB where it was used for decades, turning it from a pollutant into a case study for remediation. Regulatory circles push for stricter limits and cleaner disposal, responding to what’s learned about its health impacts and persistence. Scientific curiosity drives new combinations, modifications, and detection tricks, all grounded in experience with TNB’s strengths and weaknesses. While research continues, the story grows—linking lab benches, battlefield history, cleanup teams, and future breakthroughs in ways that underline the chemical’s ongoing importance far beyond textbooks.
What is 1,3,5-Trinitrobenzene used for?
Beyond Explosives—Where Does 1,3,5-Trinitrobenzene Fit?
1,3,5-Trinitrobenzene doesn’t show up in most people’s daily conversations. It’s easy to assume this compound just falls into the “chemicals-to-avoid” category, but digging deeper changes the narrative. With a molecular structure that packs three nitro groups onto a benzene ring, its chemistry takes on significance for more than just those in a lab coat.
1,3,5-Trinitrobenzene, often abbreviated TNB, is best known around military and defense circles because it plays a role in explosive formulations. TNB brings stability and a high detonation velocity, which means scientists don’t just throw it in for fun; its properties make a difference in creating powerful and predictable explosives. Its detonation velocity sits around 7,350 meters per second—no small number when precise military engineering matters.
Niche But Crucial: Specialty Explosive Formulations
While TNT grabs most of the headlines, TNB carves out its own space in explosive research. People think about explosives and imagine weapons, but the story’s bigger. Some explosives use TNB to guarantee performance in settings where other materials fall short. For instance, TNB appears in compositions for detonators, where reliable ignition cannot be left to chance.
The reason for TNB’s popularity among chemists and military researchers rests on its insensitivity to impact and friction compared to some alternatives. Handling safety matters. A detonation in the wrong hands is more than a science problem; it’s about keeping people alive.
TNB’s Reach Into the Laboratory
It is not just about blowing things up. TNB stands out as a reagent and analytical standard in laboratories. I became familiar with it during a project that needed a reliable chemical for calibrating detection equipment. If you try to check environmental or forensic samples for contamination, standards like TNB give you a test material with a well-known composition.
This chemical also acts as a strong oxidizing agent, meaning research labs sometimes reach for TNB when synthesizing new molecules that demand precise control over redox reactions. It’s these behind-the-scenes uses that keep science moving forward.
Issues Surrounding Use and Disposal
With any chemical that carries explosive potential, safety takes priority. TNB offers less risk of unplanned detonation than some alternatives, but that never excuses relaxed handling. Factory workers, scientists, and soldiers follow protection protocols because even rare accidents have serious consequences. Long-term storage and disposal present other hurdles. Environmental impact isn’t a small thing—improper dumping of explosives like TNB pollutes soil and groundwater, so specialized incineration or chemical breakdown methods remain necessary. Researchers keep looking for greener alternatives or ways to neutralize waste at the source, but this battle continues.
Moving Toward Safer Practices
People sometimes overlook the human side of chemical handling. In every industrial or research setting where TNB appears, thorough training can make all the difference. Regulatory oversight handles part of the risk but real safety depends on workers understanding the material and not cutting corners. Regular audits, investment in safer containers, and better emergency plans all keep risk low.
Explosive chemistry isn’t going anywhere, but everyone can agree safety for workers, the public, and the environment matters just as much as any chemical’s performance. TNB reminds us every risky compound should be balanced by informed, responsible use.
What are the safety precautions when handling 1,3,5-Trinitrobenzene?
Why It Matters
Having worked plenty of years in research labs, I’ve seen the good and bad sides of chemical handling. 1,3,5-Trinitrobenzene isn’t as well-known as TNT, yet it shares the dangers of unstable nitrated compounds. Ignoring its risks can lead to serious accidents, and in some workplaces, one mistake changes lives. Respect goes further than trusting your PPE—respect builds good habits. The goal isn’t to just follow the rules, it’s to come home healthy at the end of the day.
Handling Makes All the Difference
Every step with 1,3,5-Trinitrobenzene, even if you think it’s routine, deserves careful attention. This compound, used in explosives and as a chemical intermediate, reacts quickly if mistreated. It crumbles easily; friction, static, or a slight bump in the wrong spot can trigger a violent event. Each move with it should feel deliberate. I’ve seen younger lab workers rush, ignoring their nerves. Patience and consistency build confidence, not bravado.
Personal Protection: Not a Costume
Every lab coat and pair of gloves matter. The usual PPE—thick nitrile or neoprene gloves, full face shields, flame-resistant coats, and goggles—can feel bulky, awkward. They save skin when splashes or stray powder appear. Don’t just suit up when someone’s watching. Wearing your gear comes from knowing that protecting yourself is more important than looking tough or getting done fast.
Respirators with appropriate cartridges protect against inhaling toxic dust. When measuring or moving powder, always work in well-ventilated hoods. People skip this step in warm weather, but the risk never drops just because you’re sweating.
Storage and Inventory: No Shortcuts
Shortcuts around storage nearly always go wrong. 1,3,5-Trinitrobenzene needs a cool, dry place away from heat, sunlight, and impact sources. Static build-up and open flames push risk through the roof—always ground equipment and use non-sparking tools. Each container sits lined with compatible packing material. Labels show not just the name, but the hazards, inventory date, and owner. Old material rarely gets safer with age—rotating stock and disposing of expired batches reduces unknown risks for the next shift.
Small Details, Strong Habits
Spill control kits must be visible and ready, not hidden in dusty cabinets. Everyone working with or near these substances should know exactly what to do in case of exposure: get to a vented area, remove contaminated clothes, and call for medical help right away. Simple, practiced drills go a long way. Labs and manufacturers could prevent most incidents by running regular training, not just handing out safety data sheets. Encouraging questions and hard conversations about mistakes sets strong safety cultures.
Stepping Beyond the Basics
Improvement doesn't come from luck or ignorance—each step starts with honest check-ins and a willingness to report near-misses, no matter how minor. Equipment maintenance, keeping walkways clear, investing in anti-static flooring, and clear signage all cut down on snap decisions that lead to harm. Trust between team members gets built in those slow moments—checking each other’s PPE, reminding each other of protocols, and sharing corrections without blame. Facts show that strong teamwork and open communication lower rates of lab accidents and workplace injuries, especially among those handling sensitive compounds like 1,3,5-Trinitrobenzene.
Is 1,3,5-Trinitrobenzene explosive or hazardous?
Looking at a Chemical with a Reputation
Anyone digging into the world of nitrated organic compounds will bump into 1,3,5-trinitrobenzene sooner or later. Its name alone raises eyebrows, hinting at something potentially dangerous. Chemists know this family of molecules for their links to both explosives and specialty chemicals. A quick search will deliver plenty of technical write-ups, but it’s worth unpacking the real concerns and what sets 1,3,5-trinitrobenzene apart in the safety conversation.
Understanding the Properties
People might remember nitrobenzene from high school chemistry, but stacking on three nitro groups changes the story entirely. 1,3,5-Trinitrobenzene looks like a yellow powder. At room temperature, it won’t leap to react without effort, but its structure fuels a higher energy content. The nitro groups push the molecule close to instability, making it much more energetic than basic aromatic compounds.
Manufacturers and researchers do not handle 1,3,5-trinitrobenzene with bare hands or open jars. Specialized gloves, eye protection, and environment controls form a real part of the routine for anyone working with the substance. This doesn’t spring from paranoia; it comes from hard-earned experience and chemical accident records. Inhaling dust or getting it onto bare skin causes irritation. Swallowing it? That sends you to the poison control center. Even small spills call for proper ventilation and cleanup protocols.
Danger in Explosivity
People naturally ask about explosivity when they see “trinitro” in a chemical’s name. 1,3,5-Trinitrobenzene does show explosive properties under the right conditions. It’s not as touchy as pure nitroglycerin or TNT, but triggering a detonation requires only the right spark or impact, especially in larger quantities or finely divided forms. Once, a coworker told me about a lab mishap from decades before, where vibrations from nearby construction were enough to worry the old-timers handling trinitrobenzene samples. Nobody got hurt, but the lesson stuck with our team: respect the chemical, always.
The numbers back up these concerns. Researchers place its sensitivity between less dangerous nitroaromatics and the notoriously risky explosives, like PETN. That means it won’t explode at every bump, but storing or transporting it like any other lab supply invites real risk. Substance bans and stockroom limits exist for good reason.
Regulatory and Environmental Realities
Most countries have flagged 1,3,5-trinitrobenzene as hazardous, not just for explosive risk but also for environmental toxicity. Once released, it contaminates water and soil, threatening wildlife and possibly entering the wider food chain. Even at a local level, the cleanup requires professional handling and disposal to prevent long-term damage. Safety data sheets run several pages detailing disposal, transport, and storage.
What Safety Looks Like
Having seen both strict and lax cultures around hazardous materials, you notice the difference in workplace accidents. Labs and plants with real training, controlled storage, routine inventory checks, and strong PPE culture rarely face disasters. Some companies still cut corners, especially with less well-known chemicals like trinitrobenzene. Turning a blind eye saves money short term, but one mishap carries costs that ripple for years. Continuous education, re-certification for anyone working with explosives, and on-site emergency response plans make a direct difference.
Factoring in Alternatives and Future Research
Researchers constantly look for safer replacements in explosives and specialty chemical synthesis. Some materials with lower toxicity or explosivity can do the same job as trinitrobenzene, albeit with higher costs or less efficiency. Industry needs to balance safety and performance, but slow progress keeps everyone safer in the long run. Until better options exist, transparent handling and education around 1,3,5-trinitrobenzene remains the best tool anyone has to avoid tragedy.
How should 1,3,5-Trinitrobenzene be stored?
Why Safe Storage Even Matters
Handling chemicals always draws out the question of responsibility. With 1,3,5-Trinitrobenzene, it goes beyond ticking off a checklist. This yellow crystal packs a punch in explosives, thanks to its stability and density. Even though it doesn’t catch headlines like TNT, it sits in labs and military storerooms, waiting. Any misstep—like heat, friction, or accidental mixing—can turn a quiet day upside down. Safe storage shows respect for your coworkers, family, and anyone downwind of your facility.
Smart Storage Starts With the Right Spot
Temperature swings do no favors for explosives. Keep it cool and steady, between 2–8°C, to dodge nasty surprises. Forget that dusty shelf in the garage or a cabinet near a heat source. Humid basements or rooms with big windows push their own risks: water seeps in, sunbeams sneak through, and safety drifts away. Stick to spaces with climate control. Use insulated, ventilated rooms, with temperature alarms ready to call out trouble. Don’t crowd containers together; air should travel freely so that overheating isn’t hiding in the shadows.
Containers: Not Just a Box
Glass or tightly sealed metal cans keep moisture out and material in check. Label every jar like you’ll forget what’s inside tomorrow. It’s not just about ticking a regulation—think of the next shift worker, or your future self in a busy week, reaching for the right thing. Always level up security with lockable cabinets. If theft or tampering ever enters the picture, you want hurdles, not invitations.
Keep It Separated
Don’t put 1,3,5-Trinitrobenzene next to acids, alkalis, or other explosives. Mixing these chemicals could change the outcome from routine day to disaster. In my own experience volunteering at a local university lab, I saw how one shelf label out of place led to near misses. Only after strict color-coded bins and separation rules did everyone’s stress drop and mistakes fade away. Just because you’re careful doesn’t mean the next person won’t grab the wrong thing during a power cut or emergency.
Regular Checks Matter
You’d be surprised how quickly a secured setup turns into chaos if no one checks in. Set up a logbook: record storage dates, temperature checks, and inspections for leaks or damage. Even a small crack in a jar, or a sticky lid, spells trouble. Don’t wait until you smell something odd or see a stain. Regular inspections help catch problems early, before they spiral.
Emergency Gear and Planning
Keep protective gear nearby—goggles, gloves, chemical suits—but not buried under piles. Fire extinguishers designed for chemical fires need to be within reach, clearly marked. Post emergency numbers prominently. Practice the drills, don’t just file them away. In real emergencies, panic takes over; muscle memory from rehearsals pulls people out safely. I remember our fire marshal leading unannounced drills. The first time, chaos. The third, second nature.
Legal Oversight and Transparency
Chemical storage isn’t just about being a good neighbor; laws exist for a reason. OSHA and EPA guidelines shine here, not simply as red tape, but as the result of hard-learned lessons. Audits may annoy, but skipping rules costs more in the end. Store detailed records for authorities and your own peace of mind. Transparency beats secrecy every time, for both safety and trust. If you handle 1,3,5-Trinitrobenzene as part of your work, let the safety protocols become routine, not a burden.
What is the chemical structure of 1,3,5-Trinitrobenzene?
Looking Closer at the Molecular Layout
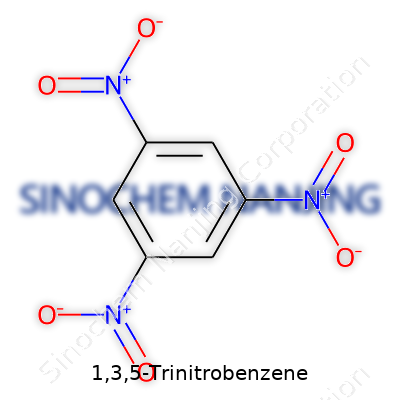
Pulling apart the chemistry behind 1,3,5-Trinitrobenzene, I picture a benzene ring, as familiar to me as that hexagonal honeycomb pattern invented by bees. On this firm foundation, three nitro groups anchor themselves at the 1, 3, and 5 positions on the ring. Each nitro group stacks a nitrogen atom, bonded to two oxygen atoms — one attached with a double bond and the other clinging through a single bond, bringing a negative charge that grabs your attention. Chemical formulas love their shorthand: C6H3N3O6 describes the whole affair. Try to sketch it, and the alternation between carbon-carbon and carbon-nitrogen connections almost feels like a carefully plotted dance.
Why Chemists and Engineers Pay Attention
I’ve seen labs where the smallest shifts in a molecular layout can decide the fate of an entire project. With 1,3,5-Trinitrobenzene, those three nitro groups take a stable ring and fill it with high energy. This property turns the compound into a crucial player for explosives and propellant research. Engineers always aim for materials that can deliver a predictable result, and the symmetric spacing of the nitro groups takes care of that. Compared to 2,4,6-TNT, also known as trinitrotoluene, 1,3,5-Trinitrobenzene often ends up chosen when higher detonation velocity or thermal stability matter most.
Environmental and Safety Considerations
Over the years, handling nitroaromatic compounds left its mark on both workers and ecosystems. Exposure, even at small doses, leads to serious health issues, like headaches or more lasting nervous system problems. Waste from manufacturing processes can seep into groundwater, spelling trouble for nearby communities. I’ve read studies documenting rising nitrobenzene levels in waterways near old production sites, and restoring those waterways stretches far into the future. Rigorous containment and monitoring limit the risk, but the challenge never really disappears.
Possible Pathways Toward Safer Use
Reducing the dangers of 1,3,5-Trinitrobenzene calls not just for containment but for finding alternatives where possible. Researchers constantly scan for new energetic materials that break down into safer byproducts or demand less severe manufacturing conditions. Some groups have turned to green chemistry principles, designing synthesis routes that reduce waste, recycle solvents, or use less hazardous reagents. In my own experience, projects that build disposal and neutralization into their earliest blueprints end up causing fewer headaches and lawsuits years down the road.
Investing in robust training for workers remains essential, too. In my own workplace, routine drills, easy-to-understand safety sheets, and real-time exposure monitoring help people avoid danger. Smart design also limits pathways that let these compounds escape into the environment. Closed handling systems, well-maintained equipment, and transparent reporting keep the risks visible.
Learning from the Chemistry and the Past
Chemicals like 1,3,5-Trinitrobenzene remind everyone in science and industry that molecular structure links directly to real-world impact. Whether that impact lies in safe demolition, efficient manufacturing, or a clean river comes down to choices made not just in a lab but in daily plant operation and regulation. Facts on paper translate directly to safety glasses and water filters in the real world, and the lessons from using compounds like this shape not just policy but chemistry’s future direction.
| Names | |
| Preferred IUPAC name | 1,3,5-Trinitrobenzene |
| Other names |
Trinitrobenzene TNB 1,3,5-TNB |
| Pronunciation | /ˌtraɪˌnaɪtroʊˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 99-35-4 |
| Beilstein Reference | **90616** |
| ChEBI | CHEBI:27870 |
| ChEMBL | CHEMBL15877 |
| ChemSpider | 54642 |
| DrugBank | DB07765 |
| ECHA InfoCard | 100.002.961 |
| EC Number | 208-769-4 |
| Gmelin Reference | 128051 |
| KEGG | C06504 |
| MeSH | D014275 |
| PubChem CID | 982 |
| RTECS number | SY8580000 |
| UNII | 1JXS2Q6U25 |
| UN number | UN1354 |
| CompTox Dashboard (EPA) | DTXSID3039240 |
| Properties | |
| Chemical formula | C6H3N3O6 |
| Molar mass | 213.11 g/mol |
| Appearance | Pale yellow crystalline solid |
| Odor | Odorless |
| Density | 1.76 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 1.8 |
| Vapor pressure | 0.00016 mmHg at 25 °C |
| Acidity (pKa) | -0.07 |
| Basicity (pKb) | -3.02 |
| Magnetic susceptibility (χ) | -89.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.620 |
| Viscosity | 2.04 mPa·s (20 °C) |
| Dipole moment | 3.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 86.8 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1397.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX06 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331: Toxic if swallowed, in contact with skin or if inhaled. H370: Causes damage to organs. |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 2-3-2-Explosive |
| Flash point | 199 °C (390 °F; 472 K) |
| Autoignition temperature | 534 °C (993 °F; 807 K) |
| Explosive limits | 1.3–7% |
| Lethal dose or concentration | LD50 oral (rat) 640 mg/kg |
| LD50 (median dose) | 660 mg/kg (rat, oral) |
| NIOSH | TT2975000 |
| PEL (Permissible) | PEL: 0.5 mg/m3 |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | 40 mg/m3 |
| Related compounds | |
| Related compounds |
Trinitrotoluene Trinitroresorcinol Trinitroaniline Trinitrophenol Nitrobenzene |

