1,2-Propanediamine: Looking at a Forgotten Cornerstone of Chemistry
Historical Development
Chemical history often skips right past backbone molecules like 1,2-Propanediamine, but without these sturdy compounds, today's research and industry would look a lot different. The story traces back to the surge of interest in amines in the early twentieth century, when synthetic chemists began tinkering with diamine structures for new pharmaceuticals, rust inhibitors, and polymer additives. Over time, 1,2-Propanediamine became a quiet staple in lab drawers and industrial tanks. Finding solid documentation about the first batches or key discoveries is tough, but the fact that this molecule frequently appeared in old reaction schemes and early patents suggests it played an underappreciated role as researchers mapped the world beyond ethylenediamine.
Product Overview
Flanked by two amine groups and a central propyl backbone, 1,2-Propanediamine comes across as unassuming. One whiff tells you this is a basic compound, packing an ammonia-like punch. Its structure brings reactivity without excessive fuss. Chemists who work with it often value it for the fine balance between its reactivity and manageability. Unlike bulkier, less volatile diamines, it handles well in day-to-day lab work, and it rarely throws unpredictable curveballs in the middle of synthesis runs.
Physical & Chemical Properties
Colorless or nearly so and wholly liquid at room temperature, 1,2-Propanediamine shines through with a sharp, fishy odor—one that lingers in memory long after the bottles are sealed. With boiling points that hover above 100 degrees Celsius and plenty of solubility in water owing to the amine groups, it finds its way into aqueous solutions easily. Its miscibility in organic solvents doesn't disappoint, making it easy to rinse from glassware or blend into more complex formulations. These are not flashy properties, but I’ve always found its simplicity makes lab prep run smoother.
Technical Specifications & Labeling
Standardization matters with chemicals like this, especially since impurities in amines create a headache for both reactivity and safety. Reagent-grade 1,2-Propanediamine typically hits purity benchmarks above 98%, sometimes higher if required for specialty synthesis. Strong, corrosive tendencies demand that containers are tightly sealed and clearly labeled, and anyone who has ever sorted out a labeling mix-up learns quickly about the importance of proper marking and storage—one false grab, and your nose or skin complains for hours. Labels usually reflect regulatory standards to satisfy both shipping guidelines and hazard communication. Safety-conscious operators look for labeling that includes hazard pictograms, signal words, and precise chemical identifiers.
Preparation Method
Industrial plants most often turn to catalytic hydrogenation and amination routes. In practice, starting with 1,2-propanediol and introducing ammonia or another amination source under controlled conditions leads to the target diamine. Depending on the precious metal or oxide catalyst, yield and byproduct patterns shift, and I've noticed people in the field constantly look for tweaks that cut energy costs or push up selectivity. Batch processes run the risk of off-gassing or byproduct buildup, so continuous-flow reactors sometimes offer better control for larger-scale production. Most synthetic routes prioritize minimizing hazardous waste and optimizing raw material use, mainly for economic reasons, which frankly makes both environmentalists and accountants smile.
Chemical Reactions & Modifications
For those of us grinding through organic synthesis, 1,2-Propanediamine opens up dependable reaction pathways. As a nucleophile, it readily forms Schiff bases with aldehydes and ketones—a workhorse step in both research and industry. Its ability to chelate metal ions gives it a role in coordination chemistry, where it can stabilize useful complexes. With both primary and secondary amine functions, it's not out of place in cyclization chemistry or as a cross-linking agent for epoxies. Over the years, I've watched innovation blossom in pharmaceutical and material science by swapping functional groups or modifying the backbone, all starting from simple diamines like this one.
Synonyms & Product Names
Chemists often toss around names like 1,2-diaminopropane, propylene diamine, or even propylenediamine, each pointing to the same compound but sometimes varying depending on which country or codebook is in play. Commercial suppliers sometimes stick to the trade name "PDAM," but formal chemical registries prefer standardized IUPAC or CAS labeling. Knowing your synonym list pays off when sourcing from global vendors or scanning literature from different traditions. I've lost count of the times an oddball trade name tripped up a new researcher, so folks in the field get into the habit of cross-referencing before placing an order.
Safety & Operational Standards
Handling 1,2-Propanediamine calls for attention, not fear. Its caustic effect on skin, eyes, and airways means anyone working with it needs gloves, goggles, and decent ventilation. Experience in shared labs showed me the value of safety training—not just for legal compliance, but for practical peace of mind. The compound's flammability nudges labs and factories toward robust spill and fire prevention, and chemical hygiene manuals highlight the importance of proper waste collection and thorough cleanup routines. Regulations around workplace exposure, shipping, and storage stem from real-world lessons in chemical safety—nobody wants another headline about avoidable chemical burns or respiratory emergencies.
Application Area
This diamine finds steady work in polymer and resin production, where it acts as a crosslinker or curing agent. Water treatment outfits rely on it as a chelating agent to guard against scaling and heavy metal fouling, key for environmental and municipal water systems. Specialty applications pop up in pharmaceuticals, where structure-based drug design sometimes calls for diamine-modified scaffolds. The agrochemical sphere draws on its reactive backbone for pesticidal formulations, making it a quiet support act to food security. Material scientists also use it when crafting new surfactants and corrosion inhibitors, broadening its reach into everything from oil fields to spray cleaners.
Research & Development
Current innovation homes in on increasing selectivity in synthesis and finding greener pathways, with biotech approaches challenging older, more energy-intensive processes. Researchers push for catalysts that reduce waste, exploiting bio-based feedstocks in search of closing the loop from renewable starting materials to finished product. In the world of advanced materials, new polymers that improve heat resistance or mechanical strength sometimes start by tweaking simple amine backbones, like those found in 1,2-Propanediamine. This constant push for improved outcomes keeps industrial chemists busy, with upstream research in academia and startups driving down costs and hazards.
Toxicity Research
Serious investigation digs into both acute and chronic effects. Animal testing and occupational studies show that repeated exposure harms respiratory systems and can sensitize skin in some people. There’s concern about long-term handling, so tight exposure limits guide factory workers and researchers. Though cases of lethal poisoning are rare, stories of skin burns and upper airway irritation show that having basic protections in place always makes sense. Researchers studying alternatives aim for derivatives or process tweaks that reduce volatility or boost biodegradability without robbing the compound of its useful core features.
Future Prospects
Looking forward, the push for sustainability and circular chemistry shines a light on overlooked building-block molecules like 1,2-Propanediamine. Interest grows around bio-based amine synthesis, with engine-room innovation aiming to meet stricter environmental footprints and regulatory controls. Downsizing plant emissions, recycling process wastes, or swapping hazardous steps for mild, enzyme-driven ones promises a better future for both chemistry and the people touching it. As technology advances, diamines like this one could form the backbone of new drug discovery, coatings, or even green materials—provided research keeps a steady focus on balancing utility with health and planet impacts.
What are the main applications of 1,2-Propanediamine?
Chemical Synthesis and Industrial Production
Looking at 1,2-Propanediamine, the story starts in industrial labs and chemical plants. Manufacturers rely on this diamine as a building block for bigger, more complex molecules. Paints, dyes, pharmaceuticals, and polymers aren't possible without the right ingredients. 1,2-Propanediamine takes on a central role here. Chemists count on its dual amine groups to create chains or ring structures, linking up molecules in a way that gets the chemistry just right for their end product. Many of the lightweight plastics and resins that make up household items or commercial goods get their toughness or flexibility from starting chemicals just like this one.
Personal Care and Household Products
Open up the cabinet under the kitchen sink or look at a bottle of face wash in the bathroom. Surfactants in cleaning products or emulsifiers in skincare haven't come out of nowhere. 1,2-Propanediamine acts as a precursor to these surface-active agents, making it easier for oil and water to mix. Soap makers and cosmetic chemists choose it for its ability to boost performance and safety in their formulations. At home, this means cleaner dishes or smoother lotions—outcomes that might seem simple unless you look behind the label.
Pharmaceuticals and Medicine
Drug developers see real value in 1,2-Propanediamine. Its molecular structure offers up key bonding spots for creating new medications. Some antihistamines, blood pressure drugs, and anti-infectives depend on this type of compound to deliver therapeutic effects. The selection of ingredients follows strict safety rules. It goes beyond just mixing chemicals—scientists must confirm compounds stand up to scrutiny and that patients can trust the outcome. In this sense, people working in drug research look for reliability, backed by well-documented testing and trusted sources.
Corrosion Inhibitors in Industry
Metal pipes and machinery run into moisture and oxygen every day. Over time, rust and corrosion can shut down production lines or water systems. 1,2-Propanediamine steps in as a corrosion inhibitor. Anyone working on a maintenance crew in a factory or water treatment facility values prevention. By halting the chemical reactions that lead to rust, it helps extend the life of equipment, saving money and downtime. Safety matters here too; workers need assurance that what gets added to water systems won't bring unexpected risks.
What Responsible Use Looks Like
Every application of 1,2-Propanediamine connects to real challenges: product quality, cost control, environmental care, and worker health. Experience on the production side highlights the need for careful storage, smart transport, and solid waste handling. Companies can use closed systems, routine monitoring, and training that keeps everyone aware of potential hazards. Following guidelines from regulatory bodies gives buyers and users confidence that what ends up in consumer goods or factory systems meets strict standards.
Seeking New Solutions
Chemists keep improving how they use chemicals like 1,2-Propanediamine. Greener processes and safer derivatives attract more attention, especially as factories push for smaller environmental footprints. Research focuses on better recycling, improved synthesis methods, and smarter applications that use less material to get the same result. Everyone from plant managers to lab staff has a role in making chemical manufacturing safer and more sustainable—and that starts with making informed choices about what ingredients to use, how to handle them, and where to find credible information.
What is the chemical formula and structure of 1,2-Propanediamine?
Understanding 1,2-Propanediamine: Where Chemistry Gets Practical
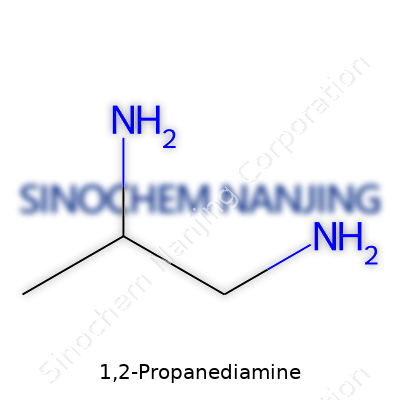
Let’s talk about 1,2-propanediamine, a compound that tends to pop up in labs and industries where people want reliable, straightforward amines that do their job without fuss. The chemical formula is C3H10N2. That's three carbons, ten hydrogens, and two nitrogens, laid out in such a way that you really see how different functional groups can change how a molecule behaves, both on paper and out in the real world.
Molecular Structure: Building from the Basics
The backbone of this molecule looks simple at first glance. There’s a propane chain, which reminds me of those basic hydrocarbon models in chemistry class—three carbon atoms linked together. Add to that, two amino groups (-NH2) hanging off the first and second carbons. Rather than stacking one functional group after another in random places, the arrangement really matters. In 1,2-propanediamine, those amine groups sitting on adjacent carbons actually influence reactivity, solubility, and several other properties that crop up in synthesis or formulations. The structure can be drawn as H2N–CH(CH3)–CH2NH2. Basically, you have a methyl group attached to one carbon, which helps the molecule stand out from its cousin, 1,3-propanediamine, where the amines are at the ends instead.
Significance in Applications
This isn’t just a textbook chemical. 1,2-propanediamine steps into the role of a linker or a building block in pharmaceutical research, specialty polymers, and even in select agricultural products. There’s a reason labs choose this compound for specific syntheses—its positioning of amine groups allows for unique reactivity. For example, it often acts as a chelating agent, binding metals for use in chemical analysis or industrial processes. That ability pops up in everyday work for chemists handling metal ions, especially since separating out or stabilizing metals gets easier with compounds like this.
Having encountered it in a few organic synthesis projects, I learned that its water solubility beats out many heavier amines. This means efficient mixing and extraction, and it reduces headaches in the clean-up stage. Side reactions stay in check, which is a small miracle during delicate steps in multi-stage syntheses. Plus, access to both primary amines on a small molecule comes in handy for making new heterocycles or trying out creative modifications in medicinal chemistry.
Health, Safety, and Environmental Considerations
Handling 1,2-propanediamine reminds me that even simple molecules deserve respect. At room temperature, it's usually a colorless to pale yellow liquid. It has a distinctive amine odor—pungent enough to turn heads if you crack open the bottle in a poorly ventilated room. Its vapor can cause respiratory irritation in the short term, and skin contact brings risk of chemical burns. Safety goggles and gloves stop close encounters with its corrosive tendencies. In my time in the lab, a good fume hood and alertness to spills made the difference between routine experiments and memorable emergencies.
Companies and researchers ought to pay attention to disposal as well. Amines have a tendency to slip into wastewater, and that’s not a scenario anyone wishes to see repeated at scale. Following local and national regulations will protect both the ecosystem and the working environment.
Looking Ahead: Responsible Use and Synthesis
While 1,2-propanediamine does important work, sustainable practices make all the difference. Using the right protective equipment, sticking to best handling practices, and choosing greener synthesis routes help reduce risks for researchers, industry workers, and the broader environment. The more we hold chemistry to these standards, the better the outcome for everyone who relies on the building blocks of science and industry.
What safety precautions should be taken when handling 1,2-Propanediamine?
Understanding 1,2-Propanediamine Risks
1,2-Propanediamine sits on shelves in many labs and industrial plants. Small molecule, strange smell — it’s not out to get you, but it doesn’t play nice if you skip the basics. Touching this chemical with bare skin or breathing in its vapor can sting your eyes, irritate your lungs, and leave you with a nasty headache. That lesson hit home for me in a summer internship, when one careless moment left a co-worker scrambling for the safety shower. The real trouble comes from thinking you’re above basic safety routines.
Personal Protective Equipment: The First Defense
Goggles never seem comfortable in a hot, humid shop, but they’re a must. Splashing this stuff anywhere near your face can cause serious burns. Nitrile gloves do the job—don’t reach for latex, since they break down too quickly. Cover exposed skin, long sleeves and closed-toe shoes. With 1,2-Propanediamine, you can’t cut corners. Sooner or later, that shortcut comes back to bite.
A sturdy lab coat does more than add flair. The fibers help block direct contact if you get sprayed, and they come off easily in an emergency. If you see foggy vapor clouding the air, that’s a sign to step back. I learned to always work with chemicals like this in a fume hood, or at least near a source of good ventilation. One small whiff in a closed room will convince anyone to take air flow seriously.
Storage and Handling: Small Habits, Big Difference
Store 1,2-Propanediamine away from heat and open flames. This chemical isn’t as explosive as some, but fires love to grab onto its fumes. Use tightly sealed containers, and never pour from a big drum into a smaller bottle without a plan for spills. Messy storage leads to messes on the floor.
Once, in a poorly organized supply room, a half-closed jug tipped off a shelf and sent fumes across the building. All because someone thought they’d “fix it later.” Clean storage reduces these headaches. Sturdy shelves, clear labels, and regular checks make a real difference. Flammable safety cabinets aren’t glamorous, but they save money—and trouble—in the long run.
Immediate Response Matters
Spill kits should sit nearby, ready to go. Absorbent pads, neutralizing agents, gloves—these tools aren’t just for big emergencies. Even a small splash on a workbench can send vapor into the air. Quick response keeps the problem small. If the chemical hits skin or clothing, wash immediately with water and peel off contaminated clothing. Sticking to this routine limits the damage; every experienced hand knows how fast an accident can spiral.
Keep emergency contacts visible. Posting poison control and local safety officers’ numbers where everyone can see them saves precious time if something goes wrong. In my experience, new staff learn fast when someone reviews emergency protocols face to face, not just on paper.
Training and Shared Awareness
Periodic safety training works better than annual forgettable lectures. People remember stories and examples far more than slideshows. Supervisors set the tone—if they skip gloves or rush through clean-ups, others notice. Safety takes hold when everyone shares responsibility and pays attention to each other’s habits.
Handling 1,2-Propanediamine brings out the basics: respect the material, choose the right gear, store it properly, and act fast when something goes wrong. Nobody wins awards for doing things right every day, but everyone stays healthy. That’s what counts.
How should 1,2-Propanediamine be stored and transported?
The Importance of Handling 1,2-Propanediamine Correctly
Anyone working around chemicals knows sloppy handling can trigger health risks or blow a company’s compliance. With 1,2-Propanediamine—a sharp-smelling, clear liquid often used to make drugs, resins, and specialty chemicals—mistakes can get expensive fast. I’ve seen more than one operation deal with damaged drums or a leaky tote just because someone cut corners on storage. This chemical is flammable and causes nasty burns if you get it on your skin or breathe in the fumes. Forgetting basic safety can land people in the hospital, halt production, or bring fines from safety inspectors. These are real costs you can avoid by handling it smartly from the start.
Storing 1,2-Propanediamine: What Actually Matters
Poor storage does more than just cause messes. 1,2-Propanediamine catches fire fast and eats through metal if you let water or air mix into it. Always put it in well-sealed, clearly labeled steel drums lined with plastic or high-density polyethylene containers. Old, rusty barrels won’t cut it—moisture sneaking in triggers rust, which speeds up corrosion. Keep containers out of direct sun and away from heat sources. I still remember a warehouse guy taking a shortcut by stacking containers near a hot air vent. They softened and nearly ruptured. Keep temperatures steady, under 25°C, and keep the area well-ventilated—vent fumes outside since the vapors make people dizzy or sick even in small amounts.
Never store acid or oxidizers in the same area. If these mix with 1,2-Propanediamine, the reaction kicks off heat, sometimes enough to start a fire. I always check that ventilation systems work and that staff wear splash-proof goggles and chemical gloves every time someone breaks open a fresh drum. Spills need clean-up kits nearby, not hidden in another part of the building, since most leaks need a response right now, not later.
Transport: Getting It from A to B Without Trouble
Transport brings another set of headaches. Some companies try skimping on approved packaging, but 1,2-Propanediamine’s labels and containers fall under global rules—think UN recommendations and the US Department of Transportation guidelines. All containers have to earn that UN mark and sit inside secondary containment, so a sudden leak doesn’t turn into a disaster on the road or at the dock.
Truckers don’t need surprises; I always review documentation and check that each drum and tote is sealed tight before anyone drives off. If the product leaks in transit, vapors can fill the air and endanger workers or first responders. Loading crews should always handle drums with secure straps and use forklifts carefully—the tiniest puncture sends fumes into the air.
Building an Ongoing Safety Culture
Training goes hand-in-hand with any good storage or transport system. New hires won’t know all the risks unless someone points them out. Monthly drills or even simple walk-throughs teach people what to do in a spill or fire. I’ve seen major accidents avoided just because a staff member remembered the right way to contain a leak before it spread.
Regular audits help too. A walkthrough with a solid checklist often turns up forgotten risks—open doors, blocked vents, damaged spill kits—that can build into bigger problems. Chemical handling calls for respect: never let routine lull folks into mistakes. A safe workplace doesn’t happen by luck or good intentions, but by real habits, clear rules, and watching out for each other every day.
What is the purity specification for commercial 1,2-Propanediamine products?
Understanding Purity in the Chemical Industry
Inside a chemical facility, every percentage point of purity grabs the attention of both the safety team and the business manager. Take 1,2-Propanediamine, a colorless liquid used for making pharmaceuticals, resins, and coatings. Manufacturers, especially those that follow guidelines from organizations like the American Chemical Society or ISO, target purity specifications for a reason—they want consistency, predictable reactions, and above all, product safety. For 1,2-Propanediamine, most commercial grades offer a minimum purity of about 98 percent. This number didn’t appear by chance. Chemical suppliers keep that bar high because even tiny traces of water or other amines can mess with downstream reactions or introduce new hazards in a facility that deals with reactive chemicals.
Impurities and Why They Get Noticed
It’s tempting to scoff at a few tenths of a percent. But customers who trust their ingredient list don’t take much for granted. Typical impurities in commercial 1,2-Propanediamine batches include traces of water, higher molecular weight amines, and, occasionally, aldehydes. Water content usually sits under 0.5 percent for a standard technical grade. Aldehyde levels also get a hard look because these byproducts can trigger unwanted reactions or affect the safety profile of the finished goods. From my time consulting for a coatings company, I remember production suddenly stalling over a spike in iron content—less than 20 parts per million, but enough to cause headaches in the final product's color and stability. Small numbers, big impact.
The Testing Game
Reliable suppliers invest in regular testing using gas chromatography, titration, and spectroscopic analyses. These methods don’t just verify the main specs; they also flag problem materials early. The process seems tedious—shipping samples, waiting for lab reports—but it saves bigger headaches later. Certificates of analysis come with every drum or tote, and purchasing departments read them closely. Discrepancies between labeled and actual purity threaten both trust and budgets. I’ve seen contracts lost over inconsistent documentation and companies spending extra to rework a batch that missed the mark by half a percent.
What Buyers Should Watch Out For
Whether ordering truckloads for pharmaceuticals or smaller batches for research, buyers need to know what their process can tolerate. Sourcing teams work closely with quality assurance to review detailed specs before shaking hands with a supplier. If a company makes resins, it might push for 99 percent or higher, paying a premium to avoid any hiccups. Facilities handling bulk fertilizer might settle for technical grade material, betting that minor impurities won’t impact their end use. Regulatory compliance matters, particularly for pharmaceuticals—here, standards from organizations like the FDA or European Pharmacopoeia offer specific limits on related substances and metallic impurities that might go even tighter than commercial suppliers offer off the shelf.
Better Outcomes Through Clear Communication
Open channels between suppliers and buyers help avoid supply chain drama. Over the years, the best relationships I’ve seen run on transparency—everyone shares their data, nobody hides subpar lots behind jargon. Companies improve outcomes by demanding third-party test results, asking for full impurity profiles, and, where budgets allow, running their own spot checks on incoming drums. If issues pop up, buyers spell out the impact on their process. Suppliers respond with either a solution or adjustments to future production runs.
Why Consistency Drives Confidence
No one benefits from mystery ingredients. Clear purity specs support better yields, safer operations, and lower costs from wasted batches or recalls. Instead of gambling on what’s inside each shipment, teams can focus on product development and business growth. In my experience, those who invest in monitoring and improvement see fewer surprises—and end up with a better reputation in a business that leaves little room for error.
| Names | |
| Preferred IUPAC name | propane-1,2-diamine |
| Other names |
1,2-Diaminopropane Propylenediamine 1,2-Propylenediamine PDMA Propane-1,2-diamine |
| Pronunciation | /ˌwʌnˌtuː.proʊˈpeɪn.daɪ.əˌmiːn/ |
| Identifiers | |
| CAS Number | 78-90-0 |
| 3D model (JSmol) | `/opt/ibm/webshare/jmol/models/0108/01083205.xyz` |
| Beilstein Reference | 622175 |
| ChEBI | CHEBI:40568 |
| ChEMBL | CHEMBL1507 |
| ChemSpider | 7866 |
| DrugBank | DB01990 |
| ECHA InfoCard | 03c6b67e-1f2d-46b7-93a1-135a4fcf7ff3 |
| EC Number | 203-865-4 |
| Gmelin Reference | **8113** |
| KEGG | C02296 |
| MeSH | D01889 |
| PubChem CID | 9547 |
| RTECS number | TY9625000 |
| UNII | 7C7829671D |
| UN number | UN2382 |
| CompTox Dashboard (EPA) | DTXSID6020939 |
| Properties | |
| Chemical formula | C3H10N2 |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Ammonia-like |
| Density | 0.872 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -1.5 |
| Vapor pressure | 11.8 mmHg (20 °C) |
| Acidity (pKa) | 10.47 |
| Basicity (pKb) | 4.20 |
| Magnetic susceptibility (χ) | -9.1·10⁻⁶ |
| Refractive index (nD) | 1.431 |
| Viscosity | 8.2 mPa·s (20 °C) |
| Dipole moment | 1.38 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 311.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -38.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1761.8 kJ/mol |
| Pharmacology | |
| ATC code | C01CA08 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H314, H332 |
| Precautionary statements | P260, P261, P264, P270, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P310, P311, P312, P321, P330, P337+P313, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 63 °F |
| Autoignition temperature | 410 °C |
| Explosive limits | 6–16% |
| Lethal dose or concentration | LD50 oral rat 1470 mg/kg |
| LD50 (median dose) | LD50 (median dose): 298 mg/kg (oral, rat) |
| NIOSH | UY0710000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1,2-Propanediamine: 2 ppm (9 mg/m³) |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,3-Propanediamine Ethylenediamine Isopropylamine |

