1,2-Phenylenediamine: A Closer Look at Its Story and Real-World Impact
Historical Development
Stories about chemicals like 1,2-phenylenediamine often start in dusty old laboratories where curious scientists chased the unknown. The late nineteenth century shaped much of modern organic chemistry. Those early researchers probably didn't realize their work with anilines and benzene rings would echo through so many industries. 1,2-Phenylenediamine came out of a period when dyes powered global trade and chemists everywhere played with coal tar and aromatic amines. The link between its discovery and the burgeoning synthetic dye industry highlights just how intertwined chemistry and commerce have always been. The stuff has tangled roots in industrial revolution progress, showing that old-school curiosity sometimes leads to large-scale practical results, even if at the time no one thought beyond that flask or test tube.
Product Overview
1,2-Phenylenediamine is usually a solid, with a telltale strong smell and off-white-to-pale purple look fresh out of a bottle. It doesn't jump out visually, but anyone familiar with its use in chemical labs, dye manufacturing, or in the formulation of certain pigments and hair dyes, would recognize its weight. Chemists often rely on stable, versatile building blocks, and 1,2-phenylenediamine fills that spot in countless syntheses. Over decades, its basic structure—a benzene ring with two amine groups sitting side-by-side—has turned it into a wildly convenient player for those building more complicated molecules.
Physical & Chemical Properties
The melting point stands at roughly 104 degrees Celsius, which lands well within the range for handling in standard labs without fancy cooling or heating. Its moderate solubility in water means it spreads out enough to handle various reaction conditions. But the property that stands out most is reactivity, owing to its two adjacent amine groups. This molecular arrangement allows chemists to mold it into a range of more complex compounds, going far beyond dyes. The aromatic structure with nitrogen handles on both sides lets it coordinate with metals or participate in cross-coupling, which opens up even more downstream chemistry. Most users will recognize the classic pungent odor, a reminder that chemical functionality often carries a sensory dimension.
Technical Specifications & Labeling
Quality control on 1,2-phenylenediamine needs to be tight, especially where pharmaceutical or high-grade technical processes rely on a steady supply of known purity. Batch labeling in factories usually includes assay values, residual moisture, and checks for regulated impurities like related amines. The real challenge often comes from the way regulations shift around chemicals tied to health hazards. Users must trust what the label claims and concurrently know the characteristics to look for, whether that’s color, melting point, or spectral signals. Anyone using it on a routine basis has developed a keen eye for small variances, since unexpected colors or residual contamination can upend whole syntheses or end up triggering safety alerts.
Preparation Method
Producing 1,2-phenylenediamine typically starts with nitrobenzene derivatives followed by reduction, echoing century-old procedures but scaled up with more efficient controls. Classic approaches use catalytic hydrogenation, swapping out harsh acids or metals for safer, recyclable options where possible. Factories often integrate process controls that monitor temperature, pressure, and pH tightly because side products can crop up suddenly and complicate purification. Many researchers remember the balancing act involved in reduction chemistry: too much force and the molecule breaks; too little and the conversion stalls. Modern plants use catalytic methods that favor high yields, but the principle remains—move electrons without losing the delicate structure of adjacent amines.
Chemical Reactions & Modifications
1,2-Phenylenediamine participates in all sorts of transformations, often as a core precursor to more complex dyes, pigments, or pharmaceuticals. Chemists can hook up those amine groups to aldehydes to create Schiff bases, a reaction anyone who’s taken an organic synthesis lab might remember for its colored byproducts and sharp odors. The molecule is a solid foundation for heterocyclic ring formation, often serving as a backbone for quinoxalines or other nitrogen-rich scaffolds, showing up in ligands for metal complexes and biochemically active compounds. The versatility comes straight out of its structure, and chemists value materials like this because they know a single tweak in a substituent or linkage opens up a handful of new applications.
Synonyms & Product Names
In catalogs, 1,2-phenylenediamine often goes by names like o-phenylenediamine or ortho-phenylenediamine, using the language that points to the amine positions. Some call it OPD on lab benches, and older literature throws in terms like 1,2-diaminobenzene. Anyone working internationally knows that catalog numbers and trade names can get in the way, but in the end most chemists and technical staff remember the look, the feel, and the unmistakable sharp smell more than any branding.
Safety & Operational Standards
Safety concerns with 1,2-phenylenediamine can’t be hand-waved away. Lab veterans talk about the sharp warning labels and personal protective equipment that always come out before the bottle opens. It’s known to cause skin irritation and poses risks through inhalation, so gloves, fume hoods, and tightly sealed balances are the rule. Over the years, increasing evidence connected aromatic amines like this one to health risks, including possible carcinogenicity, which led to tighter rules on storage and handling. Outside research labs, factory workers must deal with even greater risks from exposure, underscoring ongoing debates about industrial hygiene and chemical substitution where safer alternatives exist. Steady vigilance and real-time training matter, and anyone who’s worked a few years in chemical manufacturing has seen regulations evolve in step with new toxicity data.
Application Area
The story of 1,2-phenylenediamine stretches from the practical to the imaginative. Its roots lie in dye and pigment production, particularly for textile and cosmetic applications. As time passed, it carved out a spot in photography chemistry, the development of sensors and indicators, and even corrosion inhibitors. The scientific literature brims with examples of the molecule’s flexibility, including uses in electrochemical detection, polymer modification, and metal complexation. In some countries, it even still finds its way into hair dye formulations, although bans in several regions show the push and pull between utility and concern for public health. In pharmaceutical chemistry or research, it stands as a critical intermediate for specialty heterocycles, supporting antimicrobials and other therapeutic classes.
Research & Development
Ongoing research into 1,2-phenylenediamine usually focuses on finding safer ways to make and modify it or new applications that take advantage of its structure. Academic groups keep building on classic reactions, aiming for greener chemistry and reduced waste. There’s a lot of energy spent on catalytic processes and polymer-bound versions that can cut down on environmental impact and exposure risk. Some groups dig into ways to alter the molecule to craft novel ligands for asymmetric catalysis or sensing technologies. Others explore functionalizing its skeleton to improve biological selectivity or material utility. In my experience, the most long-lived research questions arise from problems in actual lab or industrial practice, like improving selectivity or making processes safer, which pushes scientists back to the molecule with renewed focus.
Toxicity Research
Toxicology circles back to 1,2-phenylenediamine like it does for so many aromatic amines. Evidence points to mutagenic and carcinogenic risks, especially with chronic exposure. Studies with animal models have shown concerns over liver and kidney impacts, and regulators in Europe and North America keep the compound on restricted lists for consumer goods. Hair dye use drove some of the largest studies, linking skin contact with absorption and urging cosmetics companies to shift to safer alternatives. Toxicologists still look at metabolic pathways to understand exactly how this molecule causes DNA damage, which can nudge replacement chemistry and public health advice. Workers in production environments take these risks seriously and ongoing monitoring and health screening have become essential to reduce exposure and cut down on occupational diseases.
Future Prospects
As more industries wrestle with tighter safety regulations and green chemistry targets, the future of 1,2-phenylenediamine will depend on how fast researchers and manufacturers can innovate. Chemists are searching for alternatives in hair dyes and pigments, hoping to develop colorants without the same health baggage. On the academic side, efforts to harness the reactivity of 1,2-phenylenediamine for new materials or medical applications still spark interest, especially in building blocks for polymers or advanced sensors. My experience says adoption doesn’t just hinge on scientific feasibility—social trust, regulatory clarity, and supply chain transparency all play a part in whether a material survives or fades out. For now, 1,2-phenylenediamine remains a reminder of the balancing act between chemistry’s power and responsibility, with every gram measured against safety, utility, and evolving societal expectations.
What is 1,2-Phenylenediamine used for?
What Is 1,2-Phenylenediamine?
For most people, the term 1,2-Phenylenediamine doesn’t mean much—unless you’re into chemistry, dye manufacturing, or deal with hair colorants. It’s a molecule with two amine groups connected to a benzene ring. That simple structure works its way into many industries and, honestly, into daily routines in some surprising ways.
Key Uses That Touch Daily Life
Anyone who’s colored their hair has probably encountered this chemical without realizing it. 1,2-Phenylenediamine stands at the foundation of many permanent hair dyes. Dyes using this compound create long-lasting shades because the molecule reacts easily in oxidation processes. Strong, vivid reds and blacks in hair dye often depend on these kinds of reactions. I once colored my hair in college seeking a new look, and spent hours reading ingredient labels. Nearly every box for darker tones listed phenylenediamines, a sure reminder that chemistry shapes the fashion industry in subtle but far-reaching ways.
Hair color isn’t the only area where this chemical plays a big role. Factories producing synthetic dyes for textiles, inks, and plastics all benefit from 1,2-Phenylenediamine. Not only do these dyes create vibrant shades, but they also last through repeated washes, sunlight, and general wear. The ability to hold color without bleeding or fading has shaped everything from the durability of school backpacks to the look of home furniture.
Industrial and Laboratory Applications
1,2-Phenylenediamine goes beyond color alone. In labs, researchers use it to build other useful chemicals. Some pharmaceuticals rely on it as a building block. Its structure makes it easy to modify, allowing chemists to create molecules for treating a wide variety of conditions. Its versatility keeps it in demand for synthesizing antioxidants and corrosion inhibitors, which help extend the life of metals and machinery. My neighbor, who works in a paint factory, always tells me how chemicals like this extend the lifespan of metal bridges and pipes, saving cities millions in repairs.
Health and Environmental Considerations
No story about this compound ends without talking safety. Exposure, especially in large quantities, can cause skin and eye irritation. More concerning, some research links it to allergic reactions and possible long-term health issues. Stylists who spend a career working with hair dyes containing this chemical need to take serious precautions—gloves, good ventilation, and strict hygiene routines. Policies exist in most countries to limit exposure in workplaces and ensure clear labeling for consumer products.
Factories must control waste carefully. Getting this substance into water systems creates problems for aquatic life. Regulations require filtration systems and safe disposal methods. I recall a local workshop years ago that flouted rules, only to find themselves responsible for a costly cleanup after a spill. That cautionary tale serves as an example of why chemical literacy and sound practices matter in every community.
Moving Toward Safer Alternatives
People want results from colorants, but they also expect safe products. Modern research looks for substitutes—dyes and precursors from renewable or less toxic sources. Some salons now promote “PPD-free” or “ammonia-free” dyes, appealing to consumers worried about allergic reactions. These options show that companies can listen to customer concerns and invest in science, reducing risk without sacrificing style.
The story of 1,2-Phenylenediamine shows how closely science, industry, and daily life intertwine. From boosting the boldness of a T-shirt to powering the success of a new look, its presence is felt every day. Just as important, the call for safety and responsibility grows louder, challenging everyone to keep pushing for better solutions that protect health while still meeting modern needs.
Is 1,2-Phenylenediamine hazardous or toxic?
What Is 1,2-Phenylenediamine?
1,2-Phenylenediamine shows up in all sorts of products. Some people might spot it in hair dyes, certain inks, or even as a building block for making pharmaceuticals. Its ability to add color or help build more complex molecules keeps it in circulation in different industries. If you figure it’s mostly hidden away from day-to-day life, think again. Even salons and home hair dye kits are common places to run across this chemical.
Recognized Risks and Why They Matter
Touching or breathing in 1,2-phenylenediamine can set off all kinds of trouble. People with sensitivities might break out in rashes or, in more serious instances, wind up facing hives or swelling. Allergic reactions from hair dye often lead back to ingredients like this one. Sometimes folks forget that the body doesn’t care how small the amount is—a reaction can build up after repeated use.
The toxicity isn’t limited to skin issues. Data from the U.S. National Toxicology Program has connected this substance to problems like anemia after prolonged contact or mishandling. There’s animal research linking it to cancer, though those findings come at high exposure levels. That has led the European Chemicals Agency (ECHA) to list it as a substance of very high concern.
Eyes can also take a hit. Short bursts of exposure sometimes bring burning or stinging, especially if the chemical splashes by accident. In industrial settings, people working with powdered forms could end up inhaling fumes or dust. Extended contact may end up damaging internal organs, specifically the kidneys and liver.
Everyday Experiences—Why Being Aware Counts
As someone who worked in a lab for several years, I got familiar with all sorts of warnings taped to chemical containers. The stuff that demands gloves and eye protection every single time tells a story. 1,2-Phenylenediamine landed on that list. Colleagues who ignored the rules came away with itchy hands and bloodshot eyes. That doesn’t mean the chemical always drags people down, but it makes taking precautions non-negotiable.
For regular folks using products at home, labels can leave out details. Sometimes, chemicals hide behind alternate names, so allergy-prone users need to do some homework, especially with hair dyes or tattoo inks. Reports from dermatologists show plenty of patients turning up with painful reactions that trace back to this ingredient.
Helping People Stay Safe
One solution comes from more education. Clear, honest labeling—spelling out names consumers recognize—puts power in people’s hands. Manufacturers can share side effect risks in plain language, not just obscure chemical jargon.
Protective gear for workers makes a difference in labs and on factory floors. Gloves, goggles, and good ventilation turn a risky job into a manageable one. When companies check for safer alternatives, sometimes substitutes do the same job without risking so many health problems.
On the regulatory side, government agencies keep pushing to test and limit hazardous exposures. Setting lower limits on how much of this ingredient ends up in consumer products helps cut down on long-term risks.
Once more people start talking about these risks—at the doctor, at work, even at the hair salon—it gets easier to avoid the kinds of trouble this chemical sometimes brings.
What are the storage and handling precautions for 1,2-Phenylenediamine?
Eyes Wide Open: The Real Hazards
1,2-Phenylenediamine isn’t just another brownish powder tucked away in a chemical cabinet—its toxicity deserves real respect. Skin rashes, eye irritation, even trouble breathing: these are just a few risks that come from cutting corners during storage or handling. Chemical burns and allergic reactions can hit hard, even if you don’t expect them, so it pays to take protection seriously at every step.
The Right Gear: Shielding Yourself
Working with this compound means pulling out proper gloves and protective eyewear every single time. I once saw someone think a quick transfer would be fine “just this once.” It wasn’t. Nitrile gloves and tight goggles block the worst of splashes, keeping skin out of harm’s way. Lab coats and well-fitted respirators add another layer of safety, especially if dust might get airborne.
Smart Storage: No Compromise
A big mistake I’ve witnessed is stuffing chemicals wherever there’s an open shelf. 1,2-Phenylenediamine needs a dry, cool spot, far from any sunlight or heat source. A tightly closed container—preferably labeled with hazard indicators—should be non-negotiable. Anything left open even for a short time can pull in humidity, starting chemical changes that lead to even more dangerous byproducts.
No Surprises: Avoiding Bad Mixtures
Mixing unknowns leads to trouble. This substance reacts easily with strong oxidizers and acids. If it’s sitting near bleach, hydrogen peroxide, or similar substances, you’re risking fire or toxic gas. I’ve moved containers before in shared lab spaces and found corrosive bottles an inch apart. That’s sheer luck, not a plan. Good practice keeps containers separated based on compatibility charts, which are easy to keep posted above chemical shelves.
Clean Workspaces: Small Spills, Big Risks
Tiny spills can go unnoticed but build up on gloves, benches, and tools. Years back, a missed spill led to allergic reactions on several people from cross-contamination. Cleaning as you go with approved absorbents, not just paper towels, saves a lot of trouble later. Dedicated spill kits on hand and a habit of double-checking for residue both make a difference.
Health Monitoring and Education: Looking After Each Other
Most people remember training for a few weeks, then slip into casual routines. Regular refreshers about the specific risks of 1,2-Phenylenediamine keep safety sharp. Routine medical checks for those working with the compound catch health problems before they worsen. Don’t skip ventilation checks—the compound’s dust or fumes affect everyone in the room.
Legislation and Industry Standards: Following the Letter & Spirit
Local and global regulations, like OSHA and REACH, require chemical-specific handling protocols. They make sense: these rules came from years of accidents and cases. Supervisors should lead by example, updating Standard Operating Procedures (SOPs) regularly and locking away chemicals when not in use. A logbook by the chemical cabinet, tracking check-outs and returns, stops confusion about who’s using what, and when.
Practical Solutions: Prevent the Preventable
Clear labeling, organized storage, protective gear, regular inspections: these actions cost little compared to the price of a medical emergency. Sharing personal stories about close calls, encouraging a ‘speak up’ culture, and rewarding safe behavior go a long way. Safety with 1,2-Phenylenediamine demands determination and daily care, but that care guarantees a safer workspace for everyone.
What is the chemical structure of 1,2-Phenylenediamine?
Chemical Structure Up Close
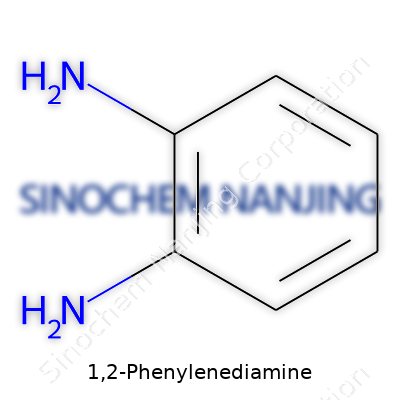
1,2-Phenylenediamine stands as one of the simplest molecules in the aromatic amine family. Imagine a benzene ring as the skeleton—six carbon atoms, arranged in a hexagon, bonded to alternating hydrogen atoms. Swap out two adjacent hydrogens on this ring for amine groups (–NH2) and you get the core of 1,2-Phenylenediamine. The chemical formula appears as C6H4(NH2)2.
Chemists draw this molecule as a hexagon with two –NH2 groups sticking out at positions 1 and 2. That arrangement means both amine groups sit next to each other—hence the name "ortho." In lab slang, they often call this substance o-phenylenediamine. This small shift makes a big difference in reactivity and usefulness compared with its siblings (like the meta- and para- versions) where those amines sit farther apart.
Why This Structure Matters
This simple placement of two amine groups next to each other brings a college chemistry textbook to life. The closeness of those groups creates unique opportunities for chemical reactions—everything from ring closures (making new rings) to double bindings with metals. Industry makes good use of these features. Dyes, polymers, and pharmaceuticals get built from this starting point.
A deeper look at the molecule’s electron cloud offers a solid example of why positions matter in chemistry. Those two amine groups push electrons into the benzene ring, supercharging it for further reactions. That little tweak enables manufacturers to create azo dyes, hair coloring products, and antioxidants. Color developers for photography also rely on the unique reactivity born from this arrangement.
Risks and Real-World Experience
Experience in a working lab brings a grounding sense to safety talk. I spent hours pipetting solutions containing aromatic diamines—always with gloves, never forgetting the potential hazards. The U.S. National Institute for Occupational Safety and Health and the International Agency for Research on Cancer both flag compounds like 1,2-Phenylenediamine as possible health risks. This isn’t empty theory: These chemicals can irritate skin, and some animal studies tie them to tumor growth. Safety must anchor every experiment or industrial application.
Many hair dyes on supermarket shelves owe their impact and long-lasting colors to this class of chemicals. Salon professionals know about allergies connected to aromatic diamines. Some countries place strict rules on concentrations, insisting on warnings and patch testing. Product reformulation and frequent monitoring keep risks low—but the structure that lends utility also brings danger if handled without care.
Building a Safer Future
Relying on 1,2-Phenylenediamine in industry calls for more transparency and research. Researchers seek safer replacements, especially in consumer products. Work proceeds on better ventilation systems, safer glove materials, and next-generation alternatives that maintain useful properties without the same toxicity. Developers and regulators both benefit from real-world reports and careful toxicity studies. Keeping up with new findings stops unnecessary exposure before it starts.
This is one molecule, but its structure provides a springboard to real technological, economic, and health conversations. Familiarity with its chemistry isn’t just for scientists. It keeps everyone from the chemist to the consumer a bit safer and much more informed.
What are the common applications of 1,2-Phenylenediamine in industry?
Coloring Everyday Life
Most folks never think about where the deep black of printer ink or the rich tones in permanent hair dyes come from. That color traces back to 1,2-phenylenediamine, an aromatic compound first introduced to chemistry labs before finding a place in so many products we use. I remember my first job in a print shop, covered in ink and watching sheet after sheet roll out—every finished piece was a silent nod to the molecules doing their invisible work.
Ink and dye manufacturing takes up a good chunk of the world’s use of this chemical. For hair dye, it reacts with other ingredients to lock in color through oxidation. The European Commission tracks this closely, pointing at allergic reactions and making sure products stick to strict safety rules. Even so, without 1,2-phenylenediamine, you wouldn’t get the same durable shades.
Making Medicine Possible
This compound has a big job in pharmaceuticals. Drug synthesis often demands building blocks that react in unique ways, and 1,2-phenylenediamine fits the bill. Chemists use it to build antibiotics, antimalarials, and other drugs that end up treating real people. When I lost a relative to malaria, I learned how many lives depend on steady supplies of these raw materials. Manufacturing plants in India and China churn out tons each year, quietly backing up the fight against disease.
Rubber and Plastics Get a Hand
Rubber products surround us, from car tires to shoe soles. Additives help these materials last longer and resist cracking, especially under heat or sunlight. 1,2-phenylenediamine acts as an antioxidant, giving tires a longer road life and saving drivers the shock of a flat. Without reliable additives, engineers and buyers would face more frequent material breakdown. That translates to higher costs (and headaches) for companies and everyday consumers.
Testing: Behind the Scenes Science
Water utilities and labs count on sensitive detection methods for traces of metals and other impurities. 1,2-phenylenediamine shows up in testing kits and labs where accuracy means safety. Researchers mix it with water samples to spot iron, copper, or other ions. In college, I spent a semester watching subtle color changes in beakers, a faint purple here or there revealing something that could switch a report from “safe” to “unacceptable.”
Challenges and Safer Paths Forward
Workers in manufacturing plants and users in salons can both end up exposed to this compound. Reports from regulatory bodies such as the U.S. EPA stress that overexposure carries risks, from skin irritation to longer-term health issues. Some companies have started to research safer alternatives, while others use more stringent ventilation and gloves. From what I have seen, information and transparency help drive better decisions, both in factories and on store shelves.
Safer substitutes are not always easy to develop, especially when so many industries depend on a stable supply. Ongoing research is vital. Stronger worker training, coupled with modernized equipment, reduces exposure without pausing production. Policy updates and independent safety testing both play a role in keeping health concerns in check while supporting industries that millions rely on.
| Names | |
| Preferred IUPAC name | benzene-1,2-diamine |
| Other names |
o-Phenylenediamine 1,2-Benzenediamine OPD o-Diaminobenzene |
| Pronunciation | /ˌwʌnˌtuː fɛˌniːlɪnˈdiːəmiːn/ |
| Identifiers | |
| CAS Number | 95-54-5 |
| Beilstein Reference | 120922 |
| ChEBI | CHEBI:17604 |
| ChEMBL | CHEMBL1400 |
| ChemSpider | 736 |
| DrugBank | DB13145 |
| ECHA InfoCard | DTXSID5020720 |
| EC Number | 202-542-8 |
| Gmelin Reference | 81424 |
| KEGG | C01417 |
| MeSH | D010673 |
| PubChem CID | 7069 |
| RTECS number | SS8050000 |
| UNII | 1L8WYL5F2P |
| UN number | UN1673 |
| Properties | |
| Chemical formula | C6H8N2 |
| Molar mass | 108.15 g/mol |
| Appearance | white to light purple solid |
| Odor | Ammonia-like |
| Density | 1.021 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | 0.12 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | 14.24 (as conjugate acid) |
| Basicity (pKb) | 7.64 |
| Magnetic susceptibility (χ) | -29.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.569 |
| Viscosity | 1.27 mPa·s (20 °C) |
| Dipole moment | 1.50 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 65.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 53.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3224 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX17 |
| Hazards | |
| Main hazards | Harmful if swallowed, toxic in contact with skin, causes serious eye damage, may cause allergic skin reaction, may cause cancer. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS05,GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Harmful in contact with skin. Causes skin irritation. Causes serious eye irritation. May cause an allergic skin reaction. Suspected of causing genetic defects. |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P310, P405, P501 |
| NFPA 704 (fire diamond) | 1,2-Phenylenediamine NFPA 704: **"Health: 3, Flammability: 1, Instability: 0"** |
| Flash point | 137°C (279°F) |
| Autoignition temperature | 546°C |
| Lethal dose or concentration | Lethal dose or concentration (LD50, oral, rat): 670 mg/kg |
| LD50 (median dose) | LD50 (median dose): 44 mg/kg (oral, rat) |
| NIOSH | SS7700000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | REL (Recommended): 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 mg/m3 |
| Related compounds | |
| Related compounds |
Benzidine p-Phenylenediamine o-Nitroaniline o-Phenylenediamine dihydrochloride Aniline |

