1,2-Ethylenediamine: A Deep Dive Into Its Story, Science, and Use
Historical Development
Years back, people were searching for compounds to knit together growing fields of chemistry. 1,2-Ethylenediamine showed up in the 19th century, born from basic organic reactions as chemistry itself found its feet. During the big industrial push, companies in Europe sought practical amines—those that could grab hold of acids, stabilize metal ions, and build polymers. Chemists kept refining their approach, producing larger and purer volumes. For decades, this compound played a role in synthesizing chelating agents and stabilizing industrial solutions, where other amines just couldn’t keep pace. The world of pharmaceuticals, textile manufacture, and polymer production owes a lot to this development journey. Whenever industry required a strong but compact diamine, this became the go-to.
Product Overview
1,2-Ethylenediamine, known as ethylenediamine or simply EDA among chemists, sits among basic building blocks for modern synthesis. A gaze at its clear, water-white liquid form gives no hint of its far-reaching impact. Industry leans on it for everything from production of resins to complexing metal ions used in agriculture and photography. Even though it wafts up a strong ammonia-like odor, its presence threads through many essential products in daily life—bleaches, lubricants, drug intermediates, and even in coolant systems. Behind the lab doors, it’s often the silent fixer when a reaction calls for a small, bidentate chelating agent.
Physical & Chemical Properties
You pick up a bottle and immediately notice the pungent, fishy smell. 1,2-Ethylenediamine shows up as a colorless liquid, volatile enough to sting the nose, and it boils at around 117°C. Its structure is simple: two amino groups attached to the ends of a two-carbon chain. The molecular formula, C2H8N2, tells most of the story—two nitrogen atoms willing to reach out for anything acidic. The density usually measures just below water, near 0.9 g/cm³ at room temperature, and it pulls moisture from the air because it’s quite hygroscopic. Water mixes with it at any ratio; it holds its own with ethanol and many organic solvents. Thanks to those two dangling amines, it forms stable complexes with a laundry list of metals, making it indispensable when a robust yet flexible ligand is needed.
Technical Specifications & Labeling
Industrial-grade ethylenediamine ships in metal drums or large intermediate bulk containers; you’ll see labeling that warns of toxicity, flammability, and the need for careful ventilation. Standard purity for most commercial processes goes up to 99%. Impurities like water, ammonia, and higher ethylene amines get monitored closely, since these can throw off downstream reactions—especially in pharmaceuticals or high-performance resins. Manufacturers post hazard warnings under GHS (Globally Harmonized System) standards, flagging it as corrosive to skin and eyes. Producers record batch numbers, UN shipping codes (UN 1604), and emergency response data right on the packaging.
Preparation Method
Most big producers use the reaction of ammonia with ethylene dichloride. Heated under pressure, these two feedstocks churn out a mixture of ethylenediamine, water, and several close cousins—diethylenetriamine, triethylenetetramine. Careful distillation isolates EDA, and equipment must manage its strong tendency to vaporize and absorb atmospheric water. Older methods, like treating monoethanolamine with ammonia over a catalyst, don’t see widespread use anymore, since large-scale production favors the ethylene dichloride route for higher throughput and simpler separation.
Chemical Reactions & Modifications
A good chemist can tweak EDA in a hundred directions. Those two amine groups make it a champ at forming Schiff bases with carbonyl compounds, producing strong chelators. Acid chlorides react readily, swapping in amide functionalities—useful for building pharmaceuticals and polymers. In the lab, EDA acts as a nucleophile, picking up alkyl groups to make secondary and tertiary amines. Oxidation yields aziridine, itself an important industrial and research intermediate. Coordination chemistry makes good use of its ability to grab two positions on a single metal center, leading to stable complexes for everything from dye manufacture to analytical chemistry. These modifications shape EDA into specialty chemicals with targeted properties for adhesives, resins, or corrosion inhibitors.
Synonyms & Product Names
Ethylenediamine goes by many names depending on who is using it: EDA, 1,2-diaminoethane, and sometimes just “diaminoethane” in older European literature. Companies brand it under trade names for specific applications, such as Dethamine or Enamine-102, especially in global markets. Chemical supply catalogs list it plainly under its CAS number 107-15-3. Safety data sheets use the most formal name, but buyers still ask for “EDA” whether it's for a bulk shipment or a specialty lab supply.
Safety & Operational Standards
Anyone who’s worked with ethylenediamine remembers the stinging sensation if gloves fail or goggles slip. Direct skin contact causes burns, and inhalation irritates the respiratory tract. Every facility handling EDA follows OSHA and local safety protocols: mandatory eye protection, solvent-proof gloves, lab coats, and active ventilation. Eyewash stations stay handy. Storage requires cool, dry areas with chemical-resistant containment, since EDA attacks metals and soft plastics over time. Emergency response teams train for accidental release because even a small spill fills the room with pungent vapors, requiring proper respirator use and rapid cleanup before anyone suffers lasting effects. Chemical workers keep MSDSs on hand and review handling standards with every new shipment. Safety is not negotiable with this material.
Application Area
Agriculture needs EDA for chelating micronutrients in fertilizers; these complexes help keep essential metals soluble and available to crops in alkaline soils. The textile sector uses it in dye manufacture and for conditioning synthetic fibers during spinning. EDA-based adducts turn up in epoxy curing agents, essential for tough composites in construction and marine repair. Pharma relies on it to build antihistamines, local anesthetics, and antifungals by linking simple molecules through those active nitrogen atoms. Factories dealing with lubricants and coolants turn to EDA for stable, long-chain additives that improve performance under pressure. Even gas treatment operations, such as removal of acidic contaminants from natural gas, pull it into scrubber solutions for sulfur and carbon dioxide fixation.
Research & Development
Researchers spent decades studying new complexes built from EDA, seeking ways to harness its bidentate grip on metals for sensors, catalysts, and electronics. Academic labs publish papers on its role in stabilizing transition metal oxidation states, a field that grew with the rise of inorganic chemistry. Drug companies keep hundreds of patents covering EDA-based scaffolds, tweaking the core structure as a backbone for new therapeutic candidates. Most recently, material scientists work on functionalized ethylenediamine derivatives for use in nanomaterials, polymer networks, and membranes for water purification. Every step forward in advanced resins or controlled drug release methods sparks renewed interest in reengineering the EDA molecule to chase higher durability or better biocompatibility.
Toxicity Research
Toxicologists highlight that 1,2-ethylenediamine presents moderate to high acute toxicity when inhaled or when it makes contact with skin or eyes, causing irritation, burns, and—in poorly controlled settings—respiratory distress. Chronic exposure links to sensitization effects, triggering allergic reactions among plant and laboratory workers. Animal studies show that high concentrations cause organ toxicity. Regulatory agencies monitor workplace exposure, and public health reports from manufacturing zones stress the need for regular air and surface monitoring. Environmental fate studies track its breakdown in soil and water, where it doesn’t persist for long, but can still cause acute toxic effects if improperly released. Workers receive regular health screenings, and modern synthetic facilities install containment and detection systems to tackle buildup before reaching harmful levels.
Future Prospects
Looking ahead, demand for EDA grows with the expanding market for advanced polymers, high-performance coatings, and specialty chemicals. Green chemistry shapes the way manufacturers redesign the synthesis process, searching for safer, less energy-intensive routes with better atom economy. The growth of battery technologies and composite materials for vehicles opens new spaces where EDA and its derivatives could improve lifespan and efficiency. Regulatory pressures to lower toxicity and emissions spur research into safer handling, with hopes for downstream modifications that keep the functionality but reduce risk. As sectors like agriculture and water treatment chase higher crop yields and cleaner supply, EDA stands ready to form the backbone of new innovations as long as safety and sustainability keep pace with demand.
What are the main uses of 1,2-Ethylenediamine?
A Closer Look at Ethylenediamine in Everyday Life
Most people never see a drum of 1,2-ethylenediamine, but the impact of this basic chemical trickles into daily life more than you’d guess. It’s a clear, ammonia-smelling liquid—definitely not something you’d want to knock over—but the chemical world leans on it for big reasons. Truth is, it helps patch together different segments of industry, from medicine to agriculture to cleaning up water.
Behind Pharmaceutical Developments
Whenever I think of disease treatment in modern times, chemistry sits right at the core. 1,2-ethylenediamine helps build many medicines, especially those involving antibiotics. Pharmaceutical firms use it as a building block, giving chemists a way to shape more complex molecules. For example, aminophylline—a drug used to help people breathe easier—needs ethylenediamine in the manufacturing process. It improves how some drugs dissolve, turning a powder into something the body’s actually able to absorb, which definitely benefits patients.
Crop Protection and Agriculture
Modern farming owes a lot to the chemicals that protect crops from bugs and mold. 1,2-ethylenediamine anchors itself in the pesticide and fungicide business. Manufacturers use it when making metal chelates that deliver nutrients to plants. Chelates let micronutrients reach a plant’s roots without getting stuck in the soil like clumps of minerals. The result—a higher yield, healthier plants, and less wasted input, which translates to more food reaching market shelves.
Industrial Chemistry and Manufacturing
Factories working with plastics, resins, and fibers see ethylenediamine as a raw material. It forms part of certain epoxy resins—tough, durable coatings found on everything from electronics to concrete garage floors. I’ve watched people repair boats with these same resin systems. Epoxy strength and resilience depend on ingredients, and ethylenediamine often brings the right stickiness and toughness to the mix. The textile industry also uses it to introduce softening agents and anti-static coatings, making synthetic fabrics more comfortable to touch.
Cleaning Up Water Supplies
Water treatment plants face never-ending challenges. Ethylenediamine acts as a chelating agent here too, grabbing onto stray metals in the water so they can’t cause harm. When heavy metals like lead or mercury show up, chelating agents keep them from reaching taps. This reduces risk for families, because contaminated water causes long-term health problems. Municipalities sometimes blend ethylenediamine derivatives into treatment cocktails for this reason.
Specialty Chemicals and Additives
Paints, dyes, and lubricants sometimes get a boost from ethylenediamine. It helps manufacturers tweak colors, making shades last longer and stay true. If you’ve ever noticed how older cars’ paint jobs stay vivid, chances are there’s some good chemistry behind it, including additives based on ethylenediamine.
Staying Safe With Ethylenediamine
Like a lot of industrial compounds, 1,2-ethylenediamine demands careful handling. It can cause skin and respiratory irritation. Strict safety measures in factories protect workers, including gloves, goggles, and fume hoods. Continuous monitoring and the right training hold accidents down, and workers have a strong reason to take these rules seriously.
Improving the Story Going Forward
Safer alternatives and better containment can soften the downsides of using ethylenediamine. Companies invest in researching substitutes that break down faster or pose fewer health risks, aiming to keep both workers and the environment safer. Keeping a close watch on emissions and favoring closed-loop systems, where chemicals recycle instead of escaping, also helps limit impact on the outside world.
What safety precautions should be taken when handling 1,2-Ethylenediamine?
Understanding the Risks
A bottle of 1,2-ethylenediamine on a laboratory shelf doesn’t look much different from any other clear liquid, but the hazards hiding inside demand respect. This chemical, which folks might see during polymer or pharmaceutical production, comes with a punch: strong smell, irritating vapors, burns on skin, and potential for fire. Even brief exposure can leave skin stinging for hours. In my early days on a college research team, one splash showed me the importance of thinking ahead.
Gear Up and Lock Down
For a long time, goggles seemed like overkill — until a mist of ethylenediamine stung my eyes. Forgetting gloves or eye protection is just about the most painful shortcut anyone could take. Nitrile or butyl gloves work best against this chemical. Rubber aprons and splash-resistant lab coats deliver another layer of armor. Face shields aren’t just for show — they keep the vapors and splashes away from your skin.
Breathing the fumes turns a tough day into a medical emergency. The vapor can irritate the nose and lungs, sometimes causing a cough or, after repeated exposure, longer-term respiratory problems. Proper fume hood use isn’t just a guideline, it’s the minimum. Even with smaller batches in open spaces, the smell fills the air. Trying to open a bottle without vent fans running can make your eyes water and your chest tight. Ventilation fans or fume hoods draw away the invisible dangers.
Safe Handling: No Shortcuts Allowed
Prepping and labeling each container beats playing a guessing game. Most trouble with chemicals starts with unmarked bottles or confusing names. All it takes is a simple label with the full chemical name, hazard symbol, and date. I’ve seen a new technician fill a waste drum labeled only “organic waste” with leftover amine, only to end up sick from the fumes. That mistake left the whole crew revisiting storage rules in a hurry.
Transferring the liquid should always be slow and steady. Never pipette by mouth — this should go without saying, but someone new to the work can feel pressured to rush. Using pipettes, pumps, or syringes with splash guards keeps the liquid where it belongs. Bringing a friend or colleague along when handling larger quantities might feel awkward, but two sets of eyes catch mistakes much faster.
Storage Smarts: Think Ahead
Storing 1,2-ethylenediamine in a tightly sealed, chemical-resistant container is the safest bet. Keep it away from strong acids, oxidizers, and most metals. It’s best off in a dedicated corrosives cabinet with secondary containment, just in case of leaks. Temperature swings and direct sunlight should be avoided; heat makes it more volatile, raising the fire risk.
Fire is a real concern here. The vapors can build up and ignite with a spark. Make sure extinguishers rated for chemical fires are close by. I once watched a small lab fire grow fast after a spill found an old extension cord.
Emergency Response
If a spill happens, evacuate folks not involved in cleanup and contain the area right away. Use absorbent pads and neutralize with weak acid if possible. Skin or eye contact means flushing with water for at least 15 minutes and seeing a doctor right away.
Training is not a formality. Anyone handling this substance should get clear coaching from someone experienced. Peer-to-peer training sticks with you more than any online test.
Building a Safety Culture
For anyone spending time with chemicals like 1,2-ethylenediamine, real safety comes from shared habits and honest conversations. The best teams I’ve worked with ask questions and give reminders, because one person’s shortcut can become everyone’s disaster. Simple routines save hands, eyes, and lives every week.
Is 1,2-Ethylenediamine hazardous to health or the environment?
Human Experience With 1,2-Ethylenediamine
Working in labs, you notice specific chemicals get handled with extra gloves and splash goggles. 1,2-Ethylenediamine stands out as one of those. Even before checking the safety data sheet, a whiff of the strong ammonia-like odor sends a clear signal: this isn’t something to brush off lightly. Handling it, you quickly learn spills irritate the nose and eyes. Colleagues sometimes complain about headaches or coughing after working near it too long. The local health and safety regulations—never short on detail—treat it as a skin and respiratory hazard.
The science matches these everyday signals. Short-term exposure irritates the eyes, skin, and lungs. People splashed with this liquid at work sometimes deal with redness, burning, and allergic reactions. NIOSH, the US health agency, recommends not breathing in its vapors or letting it contact the skin, as it penetrates gloves faster than you’d hope. Inhalation in confined lab spaces can lead to dizziness or worse for folks who already have breathing problems like asthma.
Environmental Impact
Ethylene diamine’s threat doesn’t stop at how it feels on your skin. Its chemical activity, which makes it so useful in making medicines, dyes, and cleaning agents, gives it a knack for moving quickly in the environment. It mixes with water easily and, in large accidental releases, can get into rivers, ponds, or the ground. Scientists trace how it breaks down, and while bacteria sometimes handle the job if the quantity isn’t too high, big spills overwhelm these natural clean-up crews.
Aquatic life fares poorly with this compound. Studies show it harms fish and water insects even at low concentrations. Plants growing in damp areas near factories using it sometimes show leaf burn. Some small communities near chemical plants have voiced concerns after seeing strange colors or dead fish in creeks. Their worries aren’t unwarranted.
Supporting Facts
OSHA classifies ethylenediamine as both an irritant and a possible cause of allergic skin reactions. NIOSH and similar agencies recommend limiting workplace exposure, and their recommendations are rooted in studies showing increased asthma and eczema among factory workers. A European Chemicals Agency assessment concluded that it can cause long-term harm to aquatic life, labeling it as hazardous under REACH rules. Because of these findings, chemical plants in several countries must install special filters and emergency systems to catch leaks or spills before they escape into the environment.
What Can Be Done
Simple steps make a difference. Factories can use closed systems and reliable scrubbers to capture vapors and leaks at the source. Emergency training and personal protective equipment protect workers from exposure. Good ventilation, easy-to-reach eye wash stations, and swift response plans for spills shrink the chance of accidents turning serious. Wastewater from plants processing this substance needs careful monitoring, with treatment tanks and checks before it heads into public water systems. On a bigger scale, local communities benefit when regulators push for regular checks and transparency, making data about workplace safety and emissions public. People working with or living near industry sites feel safer and can respond quickly if something goes wrong. These changes help everyone breathe easier.
What is the chemical formula and structure of 1,2-Ethylenediamine?
The Basics: Breaking Down Its Chemical Formula
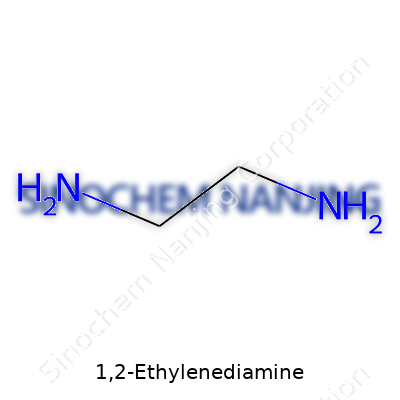
1,2-Ethylenediamine goes by the formula C2H8N2. Every molecule contains two carbon atoms, eight hydrogens, and two nitrogens. Both nitrogen atoms attach to separate carbon atoms, giving the compound a simple but impactful arrangement. Folks in labs and industry sometimes call it ethylenediamine, and it serves as a core building block for a lot of chemistry around the world.
Peering Into the Molecular Structure
Looking at its structure, it’s easy to visualize. Think of a two-carbon chain, with each carbon holding a –NH2 group. The chemical shorthand often reads H2N–CH2–CH2–NH2. So, you’ll find amino groups at both ends of the short chain. This symmetrical structure fuels much of ethylenediamine's usefulness in synthesis and industry.
Why the Structure Matters in the Real World
My time working in a research lab convinced me the details of a molecule’s structure always shape its value. Each –NH2 group is eager to grab onto other atoms and ions, so 1,2-ethylenediamine locks down metals or acids in chemical reactions. You find it balancing roles from cleaning up heavy metal spills to forming stable complexes that make catalysts hum.
Its ability to bond at two ends sets it apart from chemicals with a single active site. In making pharmaceuticals or polymers, for example, this dual-bonding power leads to longer, more flexible chains and increased efficiency in product yields. Chemists learn to rely on ethylenediamine when they want new materials that can perform under pressure or in harsh conditions.
Real-World Uses and How We Handle Risks
Factories produce thousands of tons of ethylenediamine each year for things like antifreeze, synthetic fibers, and even treatments for mining waste. Still, its strong smell and irritating vapors can sting the eyes and nose. I remember one project that drilled into the importance of proper fume hoods and gloves—with ethylenediamine, safety means everything. Breathing in just small amounts causes discomfort, so training and well-kept equipment become non-negotiable.
Environmental groups sometimes raise flags about spills and accidents involving diamines. Straightforward handling protocols and quick spill response teams help keep these concerns in check. Investing in smart ventilation, rigorous education, and sealed containment also brings down risk to almost zero. Lessons from past exposure problems led to stricter standards, especially for workers who use this chemical day in and day out.
Looking Ahead: More Than Just a Chemical
Ethylenediamine's role keeps shifting as science and industry look for greener alternatives and safer working conditions. New formulations and tighter regulations can trim down risks and side effects. Bigger attention to workplace monitoring picks up leaks before they grow into hazards. For students and professionals, understanding the chemical structure of 1,2-ethylenediamine means unlocking ideas for new medicines, stronger plastics, and safer industrial blends. Knowledge is the first step to smart application and responsible stewardship.
How should 1,2-Ethylenediamine be stored and disposed of?
Why Safe Storage Matters
1,2-Ethylenediamine gets used a lot in chemical production and lab settings. People who work in these environments know the stuff smells strong—almost like ammonia—and it can create big problems if handled poorly. Even small spills or leaks might start up fires or let off fumes that irritate the nose and eyes. In my early lab days, ignoring those warnings led to headaches and some red eyes. It drove home the importance of rules around this chemical.
Safe Storage Tips Backed by Facts
This substance likes to react with air, moisture, and lots of other chemicals, so storage choices have a direct effect on safety. The right move involves steel or polyethylene containers with tight-fitting lids. Glass containers, on the other hand, break too easily and don't offer real protection. Keeping storage rooms cool and dry stops the chemical from evaporating or reacting with water vapor. The flash point sits at only 38°C (100°F), so even a warm storage closet poses a clear danger during summer.
Label both the container and the storage area with hazard symbols and the substance name—not just for the compliance officer, but for the coworker in a rush who grabs the wrong container. From my own time in shared lab spaces, labels saved countless messy situations. Eye wash stations and fire extinguishers belong close by, not at the end of a hallway. Emergencies don't wait for a long jog.
Personal Protection Can't Be Skipped
Skin easily absorbs 1,2-Ethylenediamine. Gloves, safety goggles, and a lab coat or apron become essential instead of optional. I once saw a veteran scientist try to skip gloves while cleaning up; his hands broke out in a rash for days. No one repeats that mistake twice. Good lab practice puts safety first, not after convenience.
Disposal Isn't Just a Box to Tick
Throwing this chemical down the drain or into regular trash means risking harm to both people and environment. 1,2-Ethylenediamine reacts with common cleaning agents and metals, making it especially tricky. It contaminates water if it enters the sewer, which can cause headaches for any wastewater facility and pollute rivers. EPA guidelines say to neutralize it by mixing with an acid under controlled conditions, but that only works in labs with the right setup and people trained for the job.
For most workplaces, reputable chemical waste disposal services do a much better job. These companies know how to collect, transport, and either incinerate or treat the waste for safe long-term storage. Keeping waste logs helps track what and how much gets disposed of—useful if an inspector visits or an accidental spill occurs. Missing a signature or a date throws a wrench in that process.
Solutions Start with Training and Communication
Information-sharing builds a solid culture around chemical safety. I remember learning more from coworkers’ stories and warnings than from posters or manuals. Short training sessions show everyone how to handle, store, and dispose of materials using real-life examples. Regular walk-throughs keep safety practices at the front of people’s minds, so bad habits don’t creep in. In my experience, these human-focused methods work better than paperwork alone for building reliable habits.
Building a Safer Workplace, Step by Step
Clear rules, clear labels, and personal responsibility make a big difference with hazardous chemicals. Taking a little extra care during storage or disposal keeps coworkers safe, the lab running smoothly, and the local environment out of harm’s way. Making these practices part of the routine means fewer accidents, happier teams, and healthier communities.
| Names | |
| Preferred IUPAC name | ethane-1,2-diamine |
| Other names |
1,2-Diaminoethane Ethylenediamine EDA Aethylenediamin Dimethylenediamine |
| Pronunciation | /ˈɛθɪliːndiˌæmiːn/ |
| Identifiers | |
| CAS Number | 107-15-3 |
| Beilstein Reference | 82163 |
| ChEBI | CHEBI:28968 |
| ChEMBL | CHEMBL590 |
| ChemSpider | 14169 |
| DrugBank | DB03160 |
| ECHA InfoCard | 03b4a8c0-91cd-4bbc-9701-9f42ba7c7cd7 |
| EC Number | 203-468-6 |
| Gmelin Reference | 8257 |
| KEGG | C00137 |
| MeSH | D004821 |
| PubChem CID | 13968 |
| RTECS number | KR6300000 |
| UNII | 8NLQ36F6MM |
| UN number | UN1604 |
| Properties | |
| Chemical formula | C2H8N2 |
| Molar mass | 60.10 g/mol |
| Appearance | Colorless to yellowish liquid with an ammonia-like odor |
| Odor | Ammoniacal |
| Density | 0.899 g/cm³ |
| Solubility in water | Complete miscible |
| log P | -2.78 |
| Vapor pressure | 0.8 kPa (at 20 °C) |
| Acidity (pKa) | 7.56 |
| Basicity (pKb) | 2.89 |
| Magnetic susceptibility (χ) | -32.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.446 |
| Viscosity | 10 mPa·s (20 °C) |
| Dipole moment | 2.320 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 51.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -38.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1987.8 kJ/mol |
| Pharmacology | |
| ATC code | N07BB56 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS02,GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. May cause an allergic skin reaction. May cause respiratory irritation. |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P310, P337+P313 |
| NFPA 704 (fire diamond) | 3-3-0-A |
| Flash point | 38 °C |
| Autoignition temperature | 385 °C |
| Explosive limits | 3% to 55% |
| Lethal dose or concentration | LD50 oral rat 930 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2070 mg/kg (oral, rat) |
| NIOSH | NN0715000 |
| PEL (Permissible) | PEL: 10 ppm (parts per million) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethanolamine Diethylenetriamine Triethylenetetramine Piperazine Dimethylethylenediamine |

