1,2-Epoxypropane: A Hard Look at Its Legacy and Role in Industry
Historical Development: The Story Behind 1,2-Epoxypropane
Long before today’s world where plastics and chemicals touch nearly every part of daily life, researchers wanted ways to make materials both stronger and lighter. 1,2-Epoxypropane, more commonly called propylene oxide, didn’t just pop up by accident. Early synthetic chemists saw the epoxide structure and recognized something special: a strained three-membered ring that practically begged to react. In the early twentieth century, propylene oxide became commercially relevant, thanks to work at major chemical companies looking for new routes to glycols and polyurethanes. The timing aligned perfectly with the postwar boom, as industries from automotive to textiles demanded new polymers. The chemical’s scale-up brought with it waves of innovation, tighter safety standards, and lessons—both good and bad—about managing hazardous materials in bulk. Each chapter in its history spotlights the ongoing dance between technological ambition and responsibility to protect both workers and the planet.
Product Overview: Why 1,2-Epoxypropane Matters
Propylene oxide stands out not just for its reactivity, but for what it makes possible. If you’ve ever handled polyurethane foam in seat cushions, played with plastic toys, or even used antifreeze in your car, there’s a solid chance propylene oxide had a role to play. This chemical acts as a backbone for polyether polyols, which then form the flexible or rigid foams manufacturers shape into a dizzying array of products. Beyond that, it’s a starting material for producing propylene glycol—a substance that goes into everything from food-grade solvents to pharmaceuticals. The versatility keeps demand steady, but also raises questions about sustainable production and safe handling, especially as countries update environmental and health regulations.
Physical & Chemical Properties
Take a walk into a lab and you quickly learn that 1,2-epoxypropane is not the safest compound on the shelf. It’s usually found as a colorless, highly volatile liquid, giving off a sharp, somewhat sweet odor that tends to linger longer than most care for. The low boiling point, at around 34 degrees Celsius, makes it tough to contain; the vapors rise quickly and, if they catch a spark, ignite with frightening ease. The molecular weight hovers around 58, thanks to three carbons, six hydrogens, and an oxygen locked in that tense, triangular ring. The small epoxide ring brings both promise and peril—on one hand, it opens readily when struck by acids or bases, leading to a variety of reaction products. On the other, that same instability means leaks, spills, and vapors can turn into emergency situations, especially if ignored for even a few minutes. Keeping it contained takes vigilant engineering controls, decent ventilation, and training that goes well beyond glancing at a safety poster.
Technical Specifications & Labeling
Chemical labels for propylene oxide don’t mince words. Flammable liquid, severe irritant, and carcinogen all appear somewhere on the bottle or drum. Supply chain experts fuss over packing in airtight, secure containers—usually steel drums—marked not only for transportation hazard classes but with strict instructions for emergency response. Regular lab users respect its volatility by storing it remote from heat and direct sunlight. Chemical purity (often above 99 percent for industrial uses) matters as even trace contaminants can trigger side-reactions or interfere with polymer production. Batch tracking, barcode systems, and secondary containment regulations keep first responders and plant operators ready for both big and small problems, with compliance audits acting as constant reminders that a slip in labeling could carry a steep price.
Preparation Method
Let’s talk synthesis. Most commercial production these days uses either the chlorohydrin process or the newer hydroperoxide route. In the chlorohydrin method, propylene reacts with chlorine and water under carefully controlled conditions to give propylene chlorohydrin, which then gets dehydrochlorinated with lime. This route produces a lot of waste brine, and environmental regulators keep a close eye on both emissions and byproducts. The alternative—oxidizing propylene with an organic hydroperoxide—yields propylene oxide and a co-product, often alcohol, with fewer environmental headaches. Both methods require purpose-built reactors, well-designed separation units, and robust monitoring, since flammable intermediates and toxic byproducts mean mistakes can’t get swept under the rug. From working in pilot plants, it’s clear the focus has shifted toward cleaner, less wasteful routes—driven by tougher laws and public scrutiny.
Chemical Reactions & Modifications
Chemists value 1,2-epoxypropane for its tendency to react with just about anything eager enough to pry open that epoxide ring. Acids, bases, and nucleophiles all take a crack at it. This property lets manufacturers tailor the molecule to make everything from glycols to alcohols. For example, tossing it in with water yields propylene glycol, a workhorse chemical in industries from food to de-icers. Polyurethanes get built up by treating propylene oxide with polyols and isocyanates, creating foams that shape our mattresses and insulation. Advances in catalysis have let researchers steer reactions to favor one product over another, reducing the waste and energy consumed along the way. As a synthetic tool, 1,2-epoxypropane opens up opportunities for by-design chemistry, though every shortcut or improved selectivity has to pass not just a technical, but increasingly a social and regulatory, litmus test.
Synonyms & Product Names
In the world of chemicals, 1,2-epoxypropane goes by more names than most casual users realize. Old-timers still call it propylene oxide; some labels shorten it to PO, especially in technical documents. Propene oxide and methyl oxirane also crop up in various reference materials. This jumble of names sometimes confuses newcomers, especially when cross-referencing safety data sheets from different suppliers or regulatory bodies. Standardizing terminology doesn’t just make procurement easier—it helps everyone, from truck drivers to emergency crews, know what they’re really facing, and that helps save lives and prevent costly mistakes.
Safety & Operational Standards
If you’ve never sweated through a safety drill in a plant that handles chemicals like propylene oxide, consider yourself lucky. An accidental spill or vapor leak can turn deadly in minutes. Stringent safety protocols rule operations, starting with regular leak checks and continuing through mandatory personal protective equipment—think gloves that actually stop permeation, chemical splash goggles, and proper respirators. Automated sensor networks track air levels. Ventilation systems run almost non-stop, pushing fresh air through and contaminated air out. Frequent staff training, not only in spill response but in hazard recognition, keeps complacency at bay. Storage tanks house double containment and overpressure relief. Regulators and insurers serve as constant watchdogs, as any cut corner gets uncovered by surprise inspections. Industry groups share lessons learned after incidents—pushing for engineering corrections and safer operational standards with each close call.
Application Area: Where 1,2-Epoxypropane Ends Up
Take a look around your office or living room. Much of the foam puckering beneath your sofa fabric owes its bounce to 1,2-epoxypropane-derived polyols. Out on city streets, insulation boards in buildings and refrigerators rely on rigid foams. Beyond that, the chemical’s footprint stretches into brake fluids, spray adhesives, and lubricants. In agriculture, it gets used as a fumigant and, less commonly than before, in sterilizing medical supplies. Every time industry finds a new use for lightweight yet tough plastics or stable emulsifiers, propylene oxide seems to enter the conversation. Each use case brings not just profits, but scrutiny about end-of-life disposal, leaching, and potential environmental damage.
Research & Development: New Directions
Lab conversations about propylene oxide rarely stay basic for long. Teams keep searching for greener, safer synthesis routes, like direct oxidation over novel catalysts. Collaborative projects explore bio-based synthesis, nudging the molecule away from petrochemical feedstocks. Polymers built from propylene oxide blocks get tailored for specialty uses in electronics, coatings, and even adhesives that self-heal under the right conditions. Analytical chemists dig deep to detect trace contaminants down to the parts-per-billion level—partly to satisfy regulators, partly to keep final products as pure, safe, and predictable as possible. Increasingly, research zones in on life-cycle analysis, weighing not just technical attributes and price, but the broader environmental impact, from climate emissions to recycling bottlenecks.
Toxicity Research: Ongoing Concerns
Discussing personal safety around propylene oxide is more than just a bureaucratic requirement. Decades of studies link chronic exposure to cancer risk and DNA damage, especially in lab animals and workers facing long-term low-level exposures. Short-term high exposures vaporize the moisture in eyes and throats, and skin blistering becomes a real threat. Regulatory agencies—OSHA, EPA, IARC—debate how strict limits should get, with the science pushing standards lower as new risks surface. Dedicated teams run epidemiological studies, measuring cancer rates in industrial workers, and the legal landscape shifts with every new finding. Protective gear, local fume extraction, and monitored air quality never go out of style. Public awareness, usually spurred by the latest headline, drives stricter workplace controls and product reformulations.
Future Prospects: Where Do We Go from Here?
Looking down the road, the spotlight falls squarely on sustainable chemistry. Several pilot plants are working on greener propylene oxide production, cutting down both carbon footprints and noxious waste. Researchers push for methods using hydrogen peroxide and environmentally safer catalysts instead of old-school chlorine-based processes. Calls for biodegradable polymers sometimes clash with the performance promise of propylene oxide derivatives, nudging innovators to marry toughness with ecological responsibility. Governments ratchet up recycling and emissions standards; chemical engineers tinker with process intensification techniques, making each kilogram counted and each watt saved. Open questions remain about exposure privacy, community environmental justice, and how to truly close the loop from sourcing to final product disposal. What stays clear is that 1,2-epoxypropane, for all its hazards and all its possibilities, serves as a flashpoint for larger conversations about how industry balances ambition, accountability, and respect for both people and the planet.
What is 1,2-Epoxypropane used for?
What 1,2-Epoxypropane Does Behind the Scenes
1,2-Epoxypropane, also known as propylene oxide, works quietly in the background of modern life. Its sharp smell takes over laboratories and factories, but most people go through their days without ever coming across it—at least not directly. This clear liquid helps make things smoother, foamier, and even safer in some cases.
How 1,2-Epoxypropane Shapes Everyday Products
Step inside any home improvement store, and there’s a good chance a lot of what you see owes its shape—or its soft touch—to 1,2-Epoxypropane. The biggest share of this chemical serves the plastics industry. Polyurethane foams, which puff up sofas, car seats, mattresses, and insulation panels, start with a reaction involving propylene oxide. These foams let companies tailor comfort, safety, and energy savings for dozens of everyday uses. The construction industry, in particular, leans on these foams to make buildings warmer and more efficient.
It doesn’t stop at comfort and insulation. Propylene oxide sits at the base of the production of propylene glycol. That’s the substance tucked into bottles of antifreeze, cleaning products, deicers, and even food—as a thickener or preservative. As a result, every winter drive and every bite of a packaged snack may owe something to this molecule, even if the story gets lost in the chemistry.
Mixing Chemistry with Health and Safety
I’ve spent hours reading up on chemical safety sheets. Propylene oxide gets flagged again and again for its health risks. Short visits around open containers leave a strong scent in the air and a tingle in the throat. Researchers link repeated or high exposure to increased cancer risk, and regulatory agencies in many countries keep a close watch. Workers in production plants rely on strict controls—from ventilation to full safety suits—to handle the stuff. There’s no shrugging off the real dangers. Everyone in the chain has to take this chemical seriously, whether blending, shipping, or storing.
Despite these concerns, propylene oxide remains an important part of how industries keep pace with demand. Modern life depends on synthetic foams and antifreeze. Cutting off the supply overnight would leave furniture factories and auto shops scrambling. Still, health and environmental groups keep pushing for safer workplace standards and tighter rules on emissions. They’ve got reason to worry. In 2024, studies traced propylene oxide in industrial air samples from surrounding neighborhoods, sparking debates about what counts as safe. Those who live or work near chemical plants feel the tension between economic jobs and health worries every day.
Looking for Better Balance and Better Chemistry
The story of propylene oxide fits into a bigger question: how should society handle chemicals that feel essential but come with risks? My own take lands on the side of transparency. Communities deserve to know what’s in the air. Factory workers deserve both fair pay and protection. Scientists keep searching for alternatives and safer production methods, and industry can help by investing in new technology. Meanwhile, regular inspections and honest reporting play a role in keeping everybody safe. Some companies have begun testing greener chemistry approaches, hoping to replicate all the benefits without putting health on the line. A world with better balance seems possible if industry, regulators, and communities don’t let up pressure or lose track of real-world consequences.
Is 1,2-Epoxypropane hazardous to health?
Why Knowing the Facts Matters
Folks working in factories, labs, or warehouses often come across chemicals with names that sound like they belong in an advanced chemistry textbook. 1,2-Epoxypropane falls squarely into that category. Also known as propylene oxide, this substance pops up in all sorts of places, from the production of plastics to things like antifreeze. Based on over a decade of working closely with manufacturing equipment, I’ve learned that handling chemicals loosely classified as “harmless unless misused” leaves a lot of room for misunderstanding and error.
Direct Contact Carries Real Risks
Propylene oxide doesn’t just sting the eyes or make you cough. Inhaling even low amounts can lead to headaches, skin irritation, nausea, and in uncontrolled situations, can eventually damage your lungs. Years ago, a coworker of mine experienced redness and blistering after a spill. Medical journals back up these types of observations. The International Agency for Research on Cancer (IARC) points to evidence linking long-term exposure in animals to lung and nasal tumors. For people on the job, that signals more than a minor inconvenience.
Environmental and Long-Term Concerns
Folks living near major factories sometimes ask what happens to chemicals like 1,2-Epoxypropane that escape containment. Air and water quality begin to suffer if these leaks go unchecked. Communities downstream of industrial plants often report more cases of respiratory discomfort. While data is strongest for industrial workers, neighborhoods situated close to chemical operations deserve honest answers. US Environmental Protection Agency (EPA) research confirms that frequent exposure comes with a cancer risk. Given evidence of DNA damage in laboratory tests, the concern isn’t wild speculation.
Safety Protocols: What Actually Works?
Most workplace injuries involving chemicals boil down to simple mistakes: a missing glove, a loose vent, or too little attention paid to leaks. Regular health checkups and air quality monitoring help spot trouble before symptoms turn severe. Proper engineering controls—solid ventilation, well-maintained piping, sealed containers—make a difference. Health authorities stress that even short bursts of high exposure can cause nerve problems and liver damage, so there’s no excuse for cutting corners. In my years supervising junior staff, clear signage and easy-to-follow procedures proved just as crucial as fancy protective gear.
Looking for Safer Alternatives
Lately, a growing number of companies have pivoted to safer chemicals whenever possible. This comes from pressure by watchdog groups, consumer awareness, and stiffer regulations by governments. Some plastics manufacturers substituted less toxic epoxides, which cut the hazard without derailing production. It’s not magic, and it never happens overnight, but industries that invest in worker training and safer production show fewer accidents and happier employees.
Everyone Bears Responsibility
Ordinary people, regulators, and business owners all influence how chemicals like 1,2-Epoxypropane are handled. Asking questions, requesting safety data, and keeping tabs on local air and water is not just for scientists. I’ve seen lives improved by open communication between plant workers and local residents. Real change starts with small, consistent efforts—an extra mask, up-to-date safety drills, faster leak repairs—pushed by people who won’t settle for the bare minimum.
How should 1,2-Epoxypropane be stored?
Understanding the Baggage Behind the Bottle
Working with chemicals like 1,2-Epoxypropane, better known as propylene oxide, never feels routine. It’s clear from its sharp, ether-like odor and reputation on safety sheets: this isn’t something you leave on a windowsill. Classified as a highly flammable and reactive liquid, it’s blacklisted in most safety circles for cancer risk and fire hazard, so every step of handling keeps you on your toes.
Containers and Why They Matter
Metal drums—lined and sealed properly—get used, but not every drum will do. Containers that leak or corrode mean risk of vapor escaping, which can ignite from something as minor as static electricity. Glass rarely offers the resilience for volume, so laboratories and warehouses stick to tested metal with self-closing valves. From personal experience, a drum stored improperly in hot weather bulged and nearly spewed its contents: not a lesson you want to repeat.
Temperature Isn’t Just a Number
1,2-Epoxypropane boils at around 35°C, meaning even moderate heat can send vapor pressure skyrocketing. Warehouses keep the bottles tucked away in cool, shaded rooms—never near steam lines, sunlit windows, or boiler rooms. If the indoor thermometer pushes past 25°C, alarms go off, since any spike inches the place closer to a toxic cloud or a fireball.
No Room for Clutter
Crowding drums on a back shelf, wedging them between stacks of unrelated chemicals—this sort of shortcut risks disaster. Spills find their way into forgotten corners and blend all too easily with acids and bases, some of which can spark an explosive reaction. Segregating 1,2-Epoxypropane from anything flammable, oxidizing, or acidic turns storage from a guessing game into a safety practice. Safety data highlights that incompatible storage links directly to major chemical incidents.
Ventilation: Not Just a Fancy Feature
The smell alone reminds you why good air flow belongs in every storage room. One summer day, a faint odor seeped from storage, tripped the alarm, and had everyone scrambling for respirators. The reason? Poor ventilation didn’t catch the small leak before it built up. Proper fume extraction and routine air monitoring are central—not just suggestions.
Labeling and Routine Checks
Handwritten tags fade, and sometimes digital logs don’t match what’s on the shelf. Dates, hazard warnings, and contents in bold, water-resistant print stop mix-ups. Regular walk-throughs with a checklist, not just trusting digital systems, catch swelling drums or odd smells early. In my time, a quick daily check kept a minor crack from turning major.
Training: Knowledge Over Routine
Even smart folks skip safety steps when the job feels repetitive. Ongoing training—tailored for new staff and old-timers alike—drives home the life-or-death stakes of this work. The stories of close calls, not just the textbook rules, stick with colleagues long after training ends.
Room for Improvement
Technology brings options: automatic leak detectors, remote temperature sensors, digital checklists linked to safety alerts. But none of these beat a culture where staff respect the risks, maintain equipment, and speak up about unknowns. Inspections from local authorities catch obvious faults, but it’s the crew on the ground that sets the standard.
Treating Storage Like the First Line of Defense
1,2-Epoxypropane storage doesn’t just belong in the safety manual. It’s a frontline defense against outright disaster. Whether across industry or in research, the lessons are the same—resilience, attention to detail, and the will to ask questions keep everyone safe and prevent a routine day from turning upside down.
What precautions should be taken when handling 1,2-Epoxypropane?
Understanding the Risks
Most people won't meet 1,2-Epoxypropane, or propylene oxide, outside chemical plants or research labs. Folks who do handle it know the stakes are serious. Sitting in the middle of the chemical's warning sheet? The cancer risk, mainly through inhalation. The fumes sting the eyes, burn the throat, and leave a scratchy taste that sticks for hours. You breathe just a little too close, and the effects hit hard — headaches, dizziness, sometimes nausea. I remember my first workday with this chemical: the facilities manager never stopped talking about respect for the process. He'd seen smart people pay the price for small slip-ups.
Beyond health hazards, 1,2-Epoxypropane starts fires before a spark even hits. Its low flash point means vapors can ignite room air far from the liquid itself. This isn’t the kind of risk that improves with practice; it demands constant attention, no matter how many years you've been near the stuff.
Steps for Safer Work
The knowledge everyone learns early: suit up and never cut corners. Proper gloves (not just latex), full-face respirator or supplied-air mask, sleeves down, goggles on top of safety glasses — all must fit snug, without gaps. Common nitrile gloves swell and crack after repeated contact, so switching gloves frequently stays burned into anyone’s muscle memory. Once, after a round of batch mixing, we found a puncture in a glove only after the end-of-shift check. No rash, just luck that day, but the lesson stayed.
Ventilation is where a lab’s character shows. A cramped, stuffy space makes mistakes more likely — vapors linger, leaving workers breathing a soup of invisible threat. In a well-designed setup, the exhaust roar always hides just beneath the sound of work. Fume hoods vented outdoors offer some relief. Good supervisors schedule regular airflow tests; bad ones gamble with their teams’ lungs. If you ask an experienced handler about hot summers, many remember workers fainting or stumbling because stale air built up after an air handler failed.
Spill cleanup routines bring teamwork front and center. No one skims over training. Sand, vapor-absorbing pads, and neutralizing agents sit close to workbenches, not locked deep in a storeroom. Spills go from event to memory only when staff jump straight to their checklists: seal the area, clear people out, call for an immediate response, manage waste containers tightly closed. Waste from these actions ends up tracked until disposal, with no shortcuts.
Accountability and Reducing Risk
Backing up safety with honest communication goes a long way. Veterans know someone always fights the temptation to “just get it done.” Those moments cause slips, literal and figurative. Regular refreshers and walk-throughs help new folks feel comfortable bringing up close calls or near misses. Supervisors set the pace: reward questions, don’t shame mistakes, stay ready for upgrades when systems fall behind.
Sensors and detectors, although easy to forget in daily routine, can signal trouble before noses and throats warn us. Most labs that take safety seriously invest in digital monitors for airborne concentration, with set alarms that will not be ignored. In one blistering July, an exhaust fan shut down without notice, and a monitor’s shrill tone scattered everyone from the mixing room. Nobody got cocky with protocols again for the rest of the year.
Safer work comes with investment: in gear, in trustworthy training, and in room for honest communication. When managers budget tightly, teams pay the price with higher odds of exposure or disaster. Professionals who work with propylene oxide track near misses, call for stricter controls, and pass along lessons learned at every turn. It’s these ground level safeguards that separate near-failures from true disasters — every shift, every season.
What are the physical and chemical properties of 1,2-Epoxypropane?
The Shape and Feel
1,2-Epoxypropane goes by a catchier name in labs—propylene oxide. Pour it into a glass beaker and you’ll notice a clear, colorless liquid similar to rubbing alcohol. The smell isn’t sweet or mild; it comes across as sharp and ether-like, so working without good ventilation stings the eyes and nose. At normal room temperature, this stuff doesn’t solidify. It boils at around 34°C, which is lower than the tap water you use for a cup of tea. So, it evaporates quickly, making spills or leaks something nobody wants to ignore. If you think of water as heavy when lifting a bucket, this liquid feels lighter. Its density lands around 0.83 grams per cubic centimeter, so it floats on top of water rather than sinking in.
How It Mixes and Moves
People handling chemicals often get frustrated by substances that refuse to mix, but propylene oxide blends well with most organic solvents—alcohol, ether, acetone, you name it. In my early days in the lab, we used it to clean up stubborn residues left behind from glues and paints; its solvent power helps break them down where soap and water fail. On the other hand, it doesn’t dissolve in water easily. You get two layers if you pour it straight into a glass of water, which means any spill on the ground isn’t easily washed away by a puddle or rain.
The Reactive Side
Chemically, this molecule has a three-membered ring, with an oxygen atom holding it tightly. That makes the structure pretty strained—think of a tightly wound spring—so it snaps open if it meets the right trigger. Mix it with acids or bases and the ring opens, leading to reactions that form other substances, like glycols used for making antifreeze. I learned about that the hard way, watching a runaway reaction foam out of a flask after a careless mix. Keeping it cool and away from anything reactive turns out to be a lesson you remember. Unstable materials are rarely forgiving.
Safety and Challenges
Propylene oxide isn’t just flammable; it catches fire almost as easily as gasoline. A static spark can set off a blaze, so grounding tanks and lines in storage is a must. From a safety perspective, inhaling the vapors burns your throat and lungs and might raise the risk of cancer. The International Agency for Research on Cancer calls it possibly carcinogenic, and that label means real-world consequences in workplace rules. Protective goggles, gloves, and breathing gear aren’t just guidelines—they’re basic survival.
What Keeps People Using It?
With all the risks, factories still choose it to make polyurethane foams for furniture and car seats, among other uses in plastics and fumigants. It allows big companies to turn out products that touch everyday life—beds, insulation, packaging, and even food processing tools. Major producers run tightly controlled plants. I once visited a facility where every valve was tagged and monitored. The process uses steel pipes, containment systems, and round-the-clock staff trained for emergencies. Controlling emissions, preventing leaks, and using air scrubbers keep the bad stuff out of lungs and away from the water table.
Looking Ahead
Stricter rules for handling, better plant design, and switching to closed-loop production systems all help reduce risks. More manufacturers invest in training and detection tech, pushing to cut accidental releases. Alternatives pop up for some applications, but for now, propylene oxide’s mix of properties means it stays in heavy rotation across industries. Understanding exactly how it looks, feels, and behaves keeps both workers and the public safer as long as we respect the science and set clear guard rails.
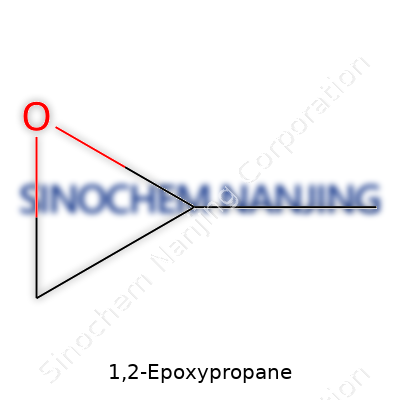
| Names | |
| Preferred IUPAC name | 2-Methyloxirane |
| Other names |
Oxirane Propylene oxide Methyl oxirane 1,2-Propylene oxide Methyloxirane |
| Pronunciation | /waɪ.tuː.ɪˈpɒk.siˌproʊ.peɪn/ |
| Identifiers | |
| CAS Number | 75-56-9 |
| Beilstein Reference | 1209281 |
| ChEBI | CHEBI:28697 |
| ChEMBL | CHEMBL14438 |
| ChemSpider | 6824 |
| DrugBank | DB00160 |
| ECHA InfoCard | 13b44fa1-70b1-43a8-ba3a-aceb4b0a780b |
| EC Number | 200-879-2 |
| Gmelin Reference | 136162 |
| KEGG | C00432 |
| MeSH | D007926 |
| PubChem CID | 6367 |
| RTECS number | TX2940000 |
| UNII | 6I6RNZ57FD |
| UN number | UN1605 |
| CompTox Dashboard (EPA) | 'DTXSID8022204' |
| Properties | |
| Chemical formula | C3H6O |
| Molar mass | 58.08 g/mol |
| Appearance | Colorless liquid with an ethereal odor |
| Odor | Ether-like |
| Density | 0.830 g/cm³ |
| Solubility in water | soluble (completely miscible) |
| log P | 0.14 |
| Vapor pressure | 400 mmHg (20°C) |
| Acidity (pKa) | 16.9 |
| Basicity (pKb) | pKb = 9.52 |
| Magnetic susceptibility (χ) | -9.07×10⁻⁶ |
| Refractive index (nD) | 1.357 |
| Viscosity | 0.43 mPa·s (20 °C) |
| Dipole moment | 2.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 276.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -20.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –2013 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D08AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS04, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H319, H332, H351 |
| Precautionary statements | P210, P273, P280, P301+P310, P303+P361+P353, P305+P351+P338, P308+P311 |
| NFPA 704 (fire diamond) | 2-4-1 |
| Flash point | -36 °C |
| Autoignition temperature | 455°C |
| Explosive limits | 2% (LEL) - 37% (UEL) |
| Lethal dose or concentration | LD50 oral rat 960 mg/kg |
| LD50 (median dose) | 150 mg/kg (rat, oral) |
| NIOSH | NL |
| PEL (Permissible) | PEL: 100 ppm |
| REL (Recommended) | 400 |
| IDLH (Immediate danger) | 400 ppm |
| Related compounds | |
| Related compounds |
Propylene glycol Propylene oxide Propylene carbonate 1-Chloropropan-2-ol |

