The Story and Significance of 1,2-Epoxybutane
Historical Development: A Walk Through Chemical Progress
Looking back over the history of 1,2-epoxybutane, people often overlook how simple molecules like this have shaped entire corners of the modern world. Chemists first started to get a handle on epoxides around the early 20th century, when the broader push for industrial solvents and intermediates turned laboratory curiosities into commercial priorities. Instead of chasing after academic puzzles, industries needed chemicals that could keep new materials flowing into factories. Researchers discovered that 1,2-epoxybutane could break open its three-membered ring with a little coaxing, making it more than just a laboratory oddity. Over time, this made it attractive for producing surfactants, specialty polymers, and even pharmaceuticals. It's not a glamorous compound, but in my view, its reliability and flexibility have carved out a steady niche, especially as alternative starting materials have become harder to source or regulate.
Product Overview: A Backbone for Synthesis
1,2-epoxybutane stands as a clear, colorless liquid with a faint, ether-like odor. Its main power lies in its structure—a strained oxirane ring that wants to react. Chemists value this property for selective ring-opening reactions. As someone who has spent time in process chemistry, I see this compound less as a final product and more as a launchpad for building specialty molecules. 1,2-epoxybutane ends up in everything from ion-exchange resins to chemical reagents. Even now, companies look for reliable sources of this stuff because of its effectiveness in making custom surfactants and glycol ethers, products no modern city could function without. The fact that it keeps showing up on order sheets year after year proves its staying power.
Physical & Chemical Properties: Why Epoxybutane Works the Way It Does
At room temperature, 1,2-epoxybutane keeps things interesting for both chemists and engineers. Its boiling point sits at around 63°C, making storage and transport less demanding than higher-boiling solvents. The density remains lower than water, which can create some handling challenges in multi-phase processes, but it also simplifies some separations. The molecule’s structure—a small ring strained to a breaking point—means it reacts easily with acids, bases, or nucleophiles. I remember a time in the lab when improper venting during heating turned a routine transfer into a learning experience, as vapors built up more quickly than expected. This volatility and reactivity define most approaches to handling, transport, and use.
Technical Specifications & Labeling: Keeping Safety Front and Center
Label accuracy plays a direct role in safety with 1,2-epoxybutane, given both its physical hazards and reactivity. Flammability stands out, with flash points low enough to make careful control of static or open flames critical. Whether transferring from drums or working at scale, clear hazard symbols and composition details help avoid accidents in even the busiest plant settings. I’ve seen too many slips in documentation lead to confusion, so rigorous labeling is more than regulatory box-ticking—it’s the thin barrier between routine operation and emergency. Proper chain of custody matters too, especially since mistaken identity with similar-looking solvents can result in disastrous process deviations.
Preparation Method: Turning Raw Materials into Useful Epoxides
Making 1,2-epoxybutane isn’t a single-step affair. Commercial manufacture usually takes butene and runs it through a reaction with hydrogen peroxide or an organic peracid to produce the epoxide ring. Catalysts like silver or specific lewis acids help drive efficiency, but small tweaks in process controls can mean big differences in yield or purity. From my own time touring chemical plants, the scale of these operations always struck me—dozens of cubic meters producing a product you wouldn’t look twice at in a glass bottle, but which underpins an astonishing range of specialty chemicals. Waste management and byproduct minimization keep creeping up in importance, partly due to rising environmental standards and community scrutiny.
Chemical Reactions & Modifications: A Gateway to Complexity
What makes 1,2-epoxybutane valuable is its willingness to react almost on cue. It can undergo acid- or base-catalyzed ring opening, forming glycols or alcohols tailored for various systems. In skilled hands, its reactions open doors to chiral building blocks, a vital resource for drug development and modern materials. I’ve seen synthetic teams in both academia and industry lean on the predictability of the epoxide ring, trusting it to provide the backbone for more elaborate molecules. Modifications might add functional groups or create custom side chains, driven by the growing demand for performance chemicals or active pharmaceutical intermediates. The versatility here isn’t theoretical; it’s something I’ve experienced first hand, mapped out on process flowcharts and validated by real-world production runs.
Synonyms & Product Names: Not Getting Lost in Translation
Different catalogs and supply chains list 1,2-epoxybutane under a jumble of names—epoxybutane, butylene oxide, 1,2-butylene oxide. Chemists know to watch for these differences, since ordering the wrong isomer or variant can set a project back by weeks. I remember an incident early in my own lab days where a shipment labeled “butylene oxide” contained the wrong regioisomer, causing a cascade of troubleshooting before we tracked the problem down. Industry players need to insist on clarity in procurement, since a simple synonym swap can cost tens of thousands in lost production time.
Safety & Operational Standards: Mitigating Everyday Risks
Conversing about safety with 1,2-epoxybutane, there’s no getting around its fire risk, acute toxicity, and potential for corrosive reactions. It won’t forgive shortcuts on ventilation or flame-proofing. Working in facilities where solvents like this get used daily, I’ve come to trust checklists and double-checks more than any single warning label. Regular monitoring for leaks, ignition sources, or improper storage saves lives—no one should treat these steps as optional. Personal protective equipment, including gloves and eye protection, helps stave off exposure, since the epoxide ring is reactive against human tissue as well. On a broader level, training and up-to-date material safety data reinforce a safety culture that reaches from the storeroom to the lab bench and out to shipping docks.
Application Area: Practical Relevance Across Industries
Anyone surprised by the reach of 1,2-epoxybutane probably just hasn’t followed the trails between industrial plants and consumer products. This molecule doesn’t draw headlines, but it sits near the foundation of everything from adhesives to rigid plastics. In my career, I’ve seen it specify the backbone for ethers used in glycol-based antifreeze, key surfactants, and even bespoke latexes that end up in specialty coatings. Research groups still find new routes to modified pharmaceuticals by leveraging its unique chemistry. For the auto industry, for instance, 1,2-epoxybutane-derived chemicals raise performance under the hood and in interior materials, handling both heat and cold with reliable durability. Even water treatment benefits from derivatives that help ion-exchange capacity or reduce fouling.
Research & Development: Pushing Boundaries—With Caution
Research teams working with 1,2-epoxybutane continue to tweak its chemistry, aiming for new niche applications where its ring strain and reactivity add unique value. In recent years, academic groups and private companies have explored tailored catalysts that tune selectivity in ring-opening, opening up opportunities for custom monomers or fine chemicals. The demand for cleaner and more sustainable production loops pushes innovators to look past old methods, seeking catalysts and starting materials with lower toxicity or carbon footprints. I attended a symposium just last year where groups showcased continuous flow systems using 1,2-epoxybutane intermediates, aiming for safer scale-up and improved process safety.
Toxicity Research: Keeping Up with Hidden Risks
Lab stories and formal toxicology reports agree—exposure to 1,2-epoxybutane comes with health risks. Scientists have documented its potential for causing skin, eye, and respiratory irritation, alongside more worrisome long-term risks like possible carcinogenicity. My own experience reinforces the need for strict handling controls. Even a few parts per million in air demand respect, making regular air sampling and worker monitoring non-negotiable in responsible operations. Regulatory agencies keep an eye on epoxides for good reason, and plants that cut corners run the real risk of regulatory shutdowns or worse, worker injury. Confusing 1,2-epoxybutane with its close cousins can be another trap—closely related compounds sometimes carry different toxicity profiles, so research and communication must stay specific.
Future Prospects: Navigating Growth and Responsibility
Looking to the future, 1,2-epoxybutane will probably see more attention both for its versatility and the challenges it brings. On one hand, demand for high-performance polymers and specialty surfactants appears set to grow, especially as emerging markets invest in new industries. More researchers are also experimenting with green chemistry principles to tame the environmental downsides of making and using this compound. Life-cycle analysis, greener catalyst development, and improved waste processing will reshape both how 1,2-epoxybutane is produced and where it can be safely used. For companies willing to innovate—and regulators willing to hold feet to the fire—there’s a real chance to keep this humble molecule at the heart of progress without repeating past mistakes. From experience, trust in science-balanced by tough oversight-keeps compounds like 1,2-epoxybutane available for the next round of breakthroughs, without slipping back on safety or responsibility.
What is 1,2-Epoxybutane used for?
The Story in Industry
Factories and labs use plenty of odd-sounding substances. 1,2-Epoxybutane stands out as a staple. In my early days working alongside industrial chemists, I saw this chemical mostly in plastic plants and coatings shops. It’s not one of those household names. Still, its role sneaks into far more corners of daily life than most folks realize.
What Makes 1,2-Epoxybutane Useful?
At its core, 1,2-Epoxybutane works as a building block. Chemists call it an intermediate. It helps form links in polymer chains, especially epoxies—those tough resins sealing garage floors, making up glues, patch kits, even coatings on electronics. The trick lies in that ring-shaped group of atoms, what’s called an epoxide. This part likes to react quickly with other chemicals, speeding up the process of making large, durable plastics.
Paint shops use this chemical to create water-resistant finishes. I’ve talked with painters who prize these formula tweaks, since their finishes hold up in high traffic, wet rooms, and even outside on boat hulls. Without the chemistry behind 1,2-Epoxybutane, these kinds of rugged coatings would cost more or fall apart under stress.
Out of the Factory, Into the Field
Pharmaceutical researchers depend on this compound to build more complex molecules. It serves as a key step in synthesizing certain drugs and lab tools. Specialty labs tap into its reactivity to attach sugar groups, tweak solvents, or create custom molecules aimed at cancer research or pest control. While I haven’t worked in that corner much, I regularly hear from biotech folks who keep it stocked for small-batch synthesis.
Some gas and oil companies use it to treat fuels or clean up contamination. The chemical helps break apart certain byproducts or stabilize tricky blends. During an industrial visit, I noticed tanks tagged with “Epoxybutane—Caution,” and the safety manager explained they used it sparingly for specific cleanup tasks.
Risks and Responsibility
Like a lot of industrial chemicals, 1,2-Epoxybutane comes with health concerns. Inhalation or skin contact can irritate or harm workers. The World Health Organization keeps an eye out for its potential as a carcinogen, yet officials have laid down clear handling rules to keep people safe. One overheard millworker joke, “If your hands smell like almonds, that’s a bad day”—a reminder about strict personal protective gear.
Waste from the manufacturing process needs care, too. Local regulations call for proper disposal and exhaust filtering. Without these controls, run-off could damage waterways or air quality. Factories that respect these protocols protect both workers and the neighbors downwind.
Looking Ahead
Cleaner production practices show promise already—better ventilation, sealed systems, smarter chemical recycling. If research on greener substitutes pans out, future generations might see less reliance on risky chemicals like this one.
For now, people in industry need to respect what 1,2-Epoxybutane does and manage its risks. Working with chemists who’ve seen both sides—the benefit in materials, and the need for hazard reduction—gives perspective. Science won’t ever stop pushing for better, safer ways to build what people use every day.
What are the safety precautions when handling 1,2-Epoxybutane?
Why This Chemical Matters
Anyone who’s spent time around industrial labs or chemical plants knows that 1,2-Epoxybutane has a strong presence in manufacturing. Used for making chemicals and coatings, it stands out for its volatility and health effects. Living through a near-miss years ago taught me that it doesn’t take much to get into trouble with this liquid—one slip with handling, and symptoms show up quick.
Common Hazards: What Goes Wrong
Handling 1,2-Epoxybutane means dealing with a substance that catches fire easily and irritates skin, eyes and lungs. Vapors build up fast without good ventilation. Workers I’ve known who got careless have dealt with coughs, rashes, and stinging eyes after short exposure. Studies from OSHA and NIOSH have linked long-term inhalation to nerve problems and possible cancer risks. It pays to take safety rules seriously, not only for yourself but for everyone nearby.
Real Safety Precautions That Work
The best protection starts before opening a drum or bottle. I always read the Safety Data Sheet (SDS) myself and push others to do the same. The SDS gives emergency steps, lists incompatibles, and covers storage temperatures. For instance, even leaving 1,2-Epoxybutane near a heat source or sunlight kicks fire risks up fast.
Based on firsthand experience and what I’ve seen enforced in solid companies, the main safety measures break down like this:
- Wear the right gear: Nitrile gloves, chemical goggles, and a long-sleeved lab coat block most splashes. For big volumes, a face shield and chemical apron matter too. Old-timers sometimes skip this, but burns and blisters remind them quick.
- Keep the air moving: Working with 1,2-Epoxybutane in a closed room is an invitation for trouble. Fume hoods pull away vapors fast. If building ventilation falls short, explosions and toxic exposure follow. Always check airflow before pouring or transferring the solvent.
- Skip open flames and sparks: This liquid flashes at low temperatures, so even a cellphone battery or a static jolt can set it off. I’ve seen a spark from synthetic clothing do damage without warning. Use grounded gear and inspect tools for signs of wear.
- Trap spills fast: If a spill hits the floor, use adsorbent pads made for organic solvents, and seal the waste up for disposal. Leaving a puddle for “later” lets fumes build up and causes slips or burns. Post emergency clean-up kits near storage areas.
- Store it safe: Keep 1,2-Epoxybutane in a flammables cabinet, tightly sealed and labeled clear as day. Order only what you’ll use soon—less waste, less risk sitting on the shelf.
Learning from Mistakes: Training Never Ends
The most reliable labs I’ve seen treat safety as a living process, not a box to check. Regular drills, honest talks about near-misses, and practice runs with spill kits keep people sharp. I have walked new hires through personal stories, not horror tales—just facts. A strong safety culture grows from real habits, reinforced by management’s support and daily reminders.
Next Steps: Build Safety into Every Routine
Laws like OSHA’s worker protections don’t just exist on paper. They reflect real risks and hard-earned lessons. Demand up-to-date training and don’t let shortcuts become acceptable. People who speak up about gaps in safety, whether it’s a broken vent or missing gloves, often prevent injuries before they happen. Respect for 1,2-Epoxybutane—or any hazardous material—comes from understanding its power to hurt and the wisdom to use it responsibly.
What are the physical and chemical properties of 1,2-Epoxybutane?
The Look and Feel
1,2-Epoxybutane comes out as a colorless liquid. Pour it into a clear vial and it’s tough to spot unless the light catches just right. The smell gives itself away—sharp, almost ether-like. Anyone who’s spent time around basic industrial chemicals might pick up on it quick. This compound’s boiling point sits at about 63°C, so it starts shooting vapors upward before water even thinks about simmering. Leave the cap off in a warm spot, and it’s gone before you blink.
This liquid likes to run, not stick. Its viscosity is low. If you spill some, it will snake quickly across lab benchtops. Work gloves keep it from sinking in since it gets through skin fast. This ease of absorption calls for real caution. The density lags behind water, coming in at around 0.87 g/cm³—so it floats if you try to mix it with a cup of water.
Reactivity and Sharp Corners
A big part of 1,2-Epoxybutane’s character ties to its epoxide ring. That triangle shape in the molecule packs a punch. Stress builds up there, so it wants to snap open when it meets acids or bases. Mix it with a strong acid or a shot of hydroxide and it won’t sit quietly. You get heat, you get new products, and often you get pressure that can build in sealed bottles. Add this stuff to a synthesis and watch for sudden changes.
One story from grad school: A friend tried to store a big sample in a warm storeroom. Next morning, the stopper lay across the room. Epoxides act fast in the wrong conditions.
It dissolves well in most common organic solvents—ether, acetone, benzene—so it slides right in with other laboratory chemicals. Most water won’t break it down, but strong mineral acids or alkalis chew through its backbone with ease. The molecule moves through air too, making indoor handling without a hood a risk. People working with it talk about the sharp, stinging sensation it leaves in the nose and throat.
What Makes It a Hazard?
Chemists respect 1,2-Epoxybutane for a reason: it’s classified as a possible carcinogen. The International Agency for Research on Cancer links exposure to increased cancer risk. Absorption through skin and lungs puts techs at risk, so anyone using it goes heavy on gloves, goggles, and good ventilation. No one wants to mess around with this level of chemical unless it’s necessary.
A splash can cause redness and burns. Inhalation means head-spins and lung irritation at low concentrations. Spills get wiped up fast with absorbents designed for organics, and most labs have a waste plan—including sealed containers and neutralizing agents—ready to keep exposure down.
Real Uses and Smarter Practices
Most labs use 1,2-epoxybutane as a building block—making pharmaceuticals, polymers, or specialty chemicals that call for its unique structure. Its reactivity unlocks access to more complex molecules. The rush to get results shouldn’t mean cutting corners, though. Routine glove checks, fume hood work, monitoring spill response kits—these skills turn into muscle memory. In my lab days, keeping each other accountable about safety meant no one ended up in the emergency room.
People working with this compound benefit from regular training. Incorporating safety posters, eye wash checks, and spill drills into the lab routine help. Firms producing materials on an industrial scale can set up closed systems, limit airborne exposure, and use continuous environmental monitoring. These aren’t extras—they’re baseline expectations rooted in experience and science.
How should 1,2-Epoxybutane be stored?
Understanding the Risks
1,2-Epoxybutane brings a lot of hazards into the mix. Its vapors can catch fire at room temperature, so improper handling invites trouble fast. Exposure harms health too—irritation, headaches, and dizziness are real risks, even without a full-blown accident. Over years working with chemicals, I’ve seen complacency cause more injuries than any equipment failure. People start thinking, “I’ve handled this before, so it’ll be fine,” until one day it isn’t.
Choosing the Right Storage Location
Flammable chemicals like 1,2-Epoxybutane do not belong anywhere near heat sources or direct sunlight. The smartest move is to use a dedicated flammable storage cabinet, away from the regular bustle of foot traffic. Fireproof cabinets provide a physical line of defense, keeping people and property safer. In labs where space gets tight, I’ve seen people store cans wherever they fit. Quick access might seem useful, but quick access won’t matter if you’re dealing with a flash fire.
Temperature Control Really Matters
This compound reacts to temperature shifts. It needs a cool, dry spot—ideally below 30°C (86°F). Higher temperatures push it closer to ignition, and humidity messes with the container seals. In facilities without strong air circulation, I’ve found that temp logging tools help. Checking temperature every day helps prevent surprises. Sometimes, people rely on air conditioning alone and forget mechanical breakdowns don’t care if a chemical is sensitive or not. Using an added thermometer or a digital monitor inside the storage spot keeps things on track.
Container Considerations
Steel or polyethylene containers work best for storing most organic chemicals, and 1,2-Epoxybutane is no exception. Containers should stay tightly sealed to keep vapors from escaping. Overfilling creates extra pressure—and that spells disaster, especially in the heat. Labeling is another area where shortcuts invite mistakes. Even old timers grab the wrong bottle sometimes if a label is faded or missing. I keep a roll of heavy-duty adhesive labels on hand, and I train new staff to check every lid and label at the end of each shift.
Ventilation and Spillage
Even inside a cabinet, the surrounding area needs solid ventilation. Spilled liquid creates a trail of fumes. At home, cracked windows are enough for paint, but inside a facility, mechanical vent hoods need to carry the fumes out. Dropping a bottle on concrete can kick up a cloud that spreads through a building within minutes. Emergency kits that neutralize spills should stay close by—preferably on the same rack as fire extinguishers. Training everyone on where to find and how to use them carries just as much weight as installing a fancy HVAC system.
Training and Accountability
Even with safety posters everywhere, it’s easy to slip back into bad habits. Regular drills and hands-on walkthroughs help people remember the stakes. New hires benefit from shadowing experienced techs for a few days instead of watching a video. The risks involved with 1,2-Epoxybutane require eyes wide open, common sense, and consistent enforcement of safe storage. By protecting each other, we keep small mistakes from turning into full-scale disasters.
Is 1,2-Epoxybutane hazardous to health or the environment?
Looking Beyond the Chemistry Book
Most folks won’t bump into 1,2-epoxybutane at the grocery store. Still, it turns up in more places than you’d expect: paint thinners, adhesives, maybe even in the warehouse down the street. This clear liquid works well as a building block in the chemical industry thanks to its reactivity. That’s good news for manufacturing, but not so great when it comes to health.
The Real-World Risks to People
Breathing in vapors from compounds like 1,2-epoxybutane doesn’t just cause a strong, unpleasant smell. Short-term exposure can make your nose and throat burn, sometimes so badly even basic tasks feel tough. Eyes water up, heads pound, skin gets red and irritated. There’s evidence from workplace safety reports that longer or repeat exposure could affect nerves and lungs. It’s also listed as a possible cancer risk by agencies like the US National Institute for Occupational Safety and Health (NIOSH), and animal studies have shown some concerning results for tumors.
This shouldn’t be a surprise—epoxide chemicals often show up in stories about toxic workplace conditions. Companies with strong safety cultures invest in protective gear, good ventilation, and regular health checks, because they’ve seen people get sick before. I used to cover safety training for a chemical handling facility, and any material like this set off alarm bells. If someone felt lightheaded or itchy or irritated, we would check if they’d been exposed and if their masks had leaks. There's a reason government rules put strict limits on the vapors workers can breathe on the job.
Environmental Effects Aren’t Just On Paper
1,2-Epoxybutane doesn’t stick around in soil or water very long, which may seem like a relief. Some would say fast breakdown means low risk, but that has more to do with luck than smart stewardship. Spills traveling down a storm drain can reach streams before the breakdown happens. Fish and invertebrates draw in dissolved chemicals like this through their gills and skin, and even short-term exposure can damage delicate tissues. Typhoons and major floods sometimes move stored vessels or waste barrels, causing sudden releases that would never be easy to explain to nearby farmers or parents.
Out in the wild, these solvents can cause headaches for the folks responsible for local water testing. Labs sometimes scramble after an accident, setting up extra sampling days and warning systems. Communities who rely on well water or who fish nearby rivers have every right to push for detailed spill protocols and real enforcement of safety plans. I know a few local activists who have spent years mapping out the source of periodic chemical odors after storms, often with little help from the companies involved until media got involved.
Pushing for Solutions That Put People First
Rules and safeguards can lower the odds of exposure but rarely wipe out risk. Stronger leak detection, real-time air monitoring, and emergency drills at facilities move the dial. Most importantly, all workers—especially temps and contracted staff—need to get the same safety education as full-timers. Factories that want to protect neighborhoods invest in community notification tools and regular transparency instead of reactivity after a spill. Science keeps learning more about these compounds. Both workers and community members deserve honest answers, not fine print or spin.
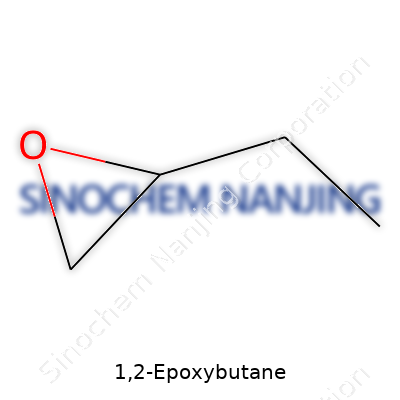
| Names | |
| Preferred IUPAC name | 2,3-Epoxybutane |
| Other names |
1,2-Butylene oxide 1,2-Butene oxide Epoxybutane |
| Pronunciation | /ˌwʌnˌtuː ɪˈpɒksiˌbjuːteɪn/ |
| Identifiers | |
| CAS Number | 106-88-7 |
| Beilstein Reference | Beilstein Reference: **1209242** |
| ChEBI | CHEBI:42511 |
| ChEMBL | CHEMBL48648 |
| ChemSpider | 8037 |
| DrugBank | DB14019 |
| ECHA InfoCard | EC000415 |
| EC Number | 1.2.2.4 |
| Gmelin Reference | 8227 |
| KEGG | C06327 |
| MeSH | D001822 |
| PubChem CID | 7846 |
| RTECS number | KX2450000 |
| UNII | 31I1PA9454 |
| UN number | UN2023 |
| Properties | |
| Chemical formula | C4H8O |
| Molar mass | 72.1057 g/mol |
| Appearance | Colorless liquid |
| Odor | ether-like |
| Density | 0.867 g/mL at 25 °C |
| Solubility in water | partly soluble |
| log P | 0.68 |
| Vapor pressure | 29.3 mmHg (20 °C) |
| Acidity (pKa) | 18.5 |
| Basicity (pKb) | The basicity (pKb) of 1,2-Epoxybutane is 13.33 |
| Magnetic susceptibility (χ) | -59.8·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4000 |
| Viscosity | 0.64 mPa·s (25 °C) |
| Dipole moment | 2.42 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 260.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −81.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2622.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02 GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H319, H335, H351, H315 |
| Precautionary statements | P210, P261, P280, P301+P310, P304+P340, P305+P351+P338, P308+P311, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1,2-Epoxybutane: "2-3-2 |
| Flash point | “−28 °C (−18 °F; 245 K)” |
| Autoignition temperature | 443°C |
| Explosive limits | 1.7% (LEL) - 14.1% (UEL) |
| Lethal dose or concentration | Lethal dose or concentration (LD50, oral, rat): 233 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1,2-Epoxybutane: "1130 mg/kg (oral, rat) |
| NIOSH | RN 106-88-7 |
| PEL (Permissible) | PEL = "50 ppm (240 mg/m3) TWA |
| REL (Recommended) | 2 ppm |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
1,2-Ethanediol Butan-2-ol Butan-1-ol Propylene oxide Epoxypropane |

