1,2-Dimethylhydrazine: Reflections on Its Legacy, Risks, and the Road Ahead
Historical Development
In the mid-20th century, researchers searching for reliable chemical models to study carcinogenesis landed on 1,2-dimethylhydrazine. Back then, the industrial boom drove a race to understand and control hazardous chemicals. This compound joined a notorious family—hydrazines—already known in rocket propellants and pharmaceuticals. Yet, its chief notoriety came from its link to colon cancer research. Laboratories worldwide leaned heavily on 1,2-dimethylhydrazine for its consistent cancer-inducing ability in rodents, allowing generations of scientists to explore the stages and genetics of tumor development. This focus shaped much of modern toxicology’s early thinking about environmental carcinogens, risk factors, and intervention strategies.
Product Overview
1,2-Dimethylhydrazine doesn’t show up on supermarket shelves, and for good reason. This compound belongs to a class of chemicals more famous for their hazards than their household perks. Used mainly in research or specialized industry, it usually appears as a clear, oily liquid—volatile, flammable, and highly toxic. From a practical viewpoint, the compound’s main appeal lies in its chemical reactivity and predictable biological effects, especially its unique ability to cause DNA damage in living organisms. Its reputation among scientists walks a fine line between respect for its research value and serious concern about its hazards.
Physical & Chemical Properties
Handle 1,2-dimethylhydrazine in a lab, and you’ll soon notice its pungent odor and tendency to evaporate quickly. The clear liquid flashes a warning not just by smell but by volatility and combustibility. Chemically, its structure seems simple: two methyl groups attach to a hydrazine backbone. Yet that simplicity hides a molecule ready to react with oxygen, acids, and other compounds. Small molecular weight and easy vaporization mean inhalation risk climbs fast. Storage often involves careful controls—unbreakable bottles with proper venting, locked cabinets, and regular monitoring for leaks. It doesn’t mix safely with just anything; spontaneous reactions with oxidizers or exposure to heat can turn dangerous in a blink.
Technical Specifications & Labeling
Purchase from a chemical supplier, and the label reads more like a cautionary tale than a shopping list. The United Nations assigns a hazardous materials number right up there with other reactive, toxic substances. Labels warn of acute toxicity, environmental danger, and flammability. Even shipping requires adherence to strict international codes—hazmat teams won’t touch it without training. In practice, these warnings serve as reminders of the need for gloves, eye protection, and positive pressure hoods in professional settings. Where chemicals like ethanol beg only for responsible handling, 1,2-dimethylhydrazine demands a thorough hazard assessment before every use.
Preparation Method
Most 1,2-dimethylhydrazine synthesis routes rely on well-guarded chemical procedures. Industrial labs typically choose controlled alkylation of hydrazine or sequence methylation with methyl halides. In the old days, processes required strict temperature and pressure control, since unwanted by-products could lead to toxic exposures or even fires. Today’s synthesis methods use as little raw hydrazine as possible and emphasize containment, recycling waste, and minimizing emissions. Each step hinges on decades of incremental safety improvements born from hard-learned lessons.
Chemical Reactions & Modifications
As a building block, 1,2-dimethylhydrazine is valued for how easily it hands off its methyl groups or forms new bonds under the right conditions. The methylhydrazine backbone reacts with acids to form salts, while stronger oxidizers pitch it toward chain reactions best observed from a safe distance. Chemists appreciate the challenges it brings; for example, modifications often focus on dampening its toxicity while retaining reactivity, pointing toward derivatives with more narrowly tailored uses. Yet, its widespread application in synthetic routes gets limited by the ever-present safety questions.
Synonyms & Product Names
In chemical catalogs and references, 1,2-dimethylhydrazine appears under plenty of names: symmetrical dimethylhydrazine, SDMH, or symmetrical 1,2-dimethylhydrazine. Some researchers stick to simple abbreviations, though the compound’s notoriety means most professionals recognize it regardless of the label. In regulatory filings and technical documents, the full IUPAC name signals more than just nomenclature consistency—it makes plain just what dangers and precautions the user faces.
Safety & Operational Standards
Lab work with this compound feels like a masterclass in containment strategy. Everyone on the team needs training not only in the basics of chemical hygiene, but in emergency protocols for spills and exposures. Safety showers and eyewash stations are non-negotiable features near work spaces. Constant monitoring for vapor leaks, strict ventilation, and the use of chemical fume hoods reinforce the point: here’s a chemical that does not tolerate shortcuts. Regulators in the United States, Europe, and elsewhere classify 1,2-dimethylhydrazine as both acutely toxic and potentially carcinogenic, urging constant adherence to exposure limits and monitoring for contamination—especially given the substance’s strong associations with occupational illness.
Application Area
Almost every significant use for 1,2-dimethylhydrazine falls within scientific research. Its ability to generate colon tumors in animal models shaped decades of biomedical inquiry. Researchers testing preventive drugs or seeking genetic roots for cancer returned again and again to this compound. Outside the lab, there’s almost no commercial use due to its hazards and the availability of safer alternatives in most fields. In rocket propulsion, its structurally similar cousin, unsymmetrical dimethylhydrazine, gets more attention. That limitation speaks to its singular reputation; chemical companies rarely produce it unless a research contract demands it.
Research & Development
My own work in toxicology has often circled back to 1,2-dimethylhydrazine, especially during early studies on DNA repair and the mechanisms underlying colon cancers. The compound gave researchers an almost reliable way to induce tumors and test interventions—something that advanced cancer biology by leaps and bounds. These early forays emphasized sharp experimental control and drove labs to isolate genetic mutations linked to both disease onset and progression. In recent years, most research shifts toward understanding the exact metabolic pathways that turn this compound from an oily liquid on a shelf into a mutagenic force of nature inside cells. This quest opened up genomics, molecular diagnostics, and even preventive medicine strategies with roots in early hydrazine studies.
Toxicity Research
Studying 1,2-dimethylhydrazine forced an entire generation of scientists to face the tough realities behind chemical safety. Animal studies painted a clear picture: repeated exposure causes cancer, especially in the large intestine. At a cellular level, the compound fragments DNA, sets off uncontrolled cell division, and overrides the very systems the body relies on to tamp down rogue mutations. Occupational exposures haunt the historical record, with reported cases of acute poisoning, coma, and even deaths—most years ago, as regulator standards tightened and awareness improved. Some early researchers didn’t appreciate the risk until symptoms set in, underscoring the value of today’s strict safety culture.
Future Prospects
Look forward, and it’s clear that 1,2-dimethylhydrazine’s era in mainstream research is winding down. Few new laboratories adopt its use, and most funding agencies favor safer, more precise model compounds for toxicity and cancer studies. That said, the compound’s legacy endures in the libraries of data, the lessons about carcinogenesis, and the safety culture it helped inspire. In the coming years, researchers might leverage data from past studies to refine in silico models for predicting environmental carcinogens, replace animal studies with better cell-based systems, and push for substitutes that limit risk without compromising insight. As green chemistry matures, one hopes that legacy chemicals like 1,2-dimethylhydrazine serve more as historical signposts than present-day risks.
What is 1,2-Dimethylhydrazine used for?
What 1,2-Dimethylhydrazine Does in the Lab
Many folks outside research circles haven't run into the name 1,2-dimethylhydrazine, but scientists know it well, especially in cancer research. This compound, classified as a hydrazine derivative, plays an unusual yet critical role: it helps researchers study how cancer, especially colon cancer, develops. The main way it does this is by acting as a chemical that causes tumors in animal models during experiments.
In research projects, labs inject 1,2-dimethylhydrazine into rodents, most often rats or mice, to trigger tumor growth. This process creates models that mimic human colorectal cancer. Through these models, scientists try to pinpoint why healthy cells begin to grow out of control, test the effectiveness of new treatments, and improve early-detection methods. This approach has led to discoveries in how certain genes interact with carcinogens, and why one person might be more at risk than another.
Why such a Risky Chemical?
Few people ever touch 1,2-dimethylhydrazine directly, and with good reason. This substance is toxic, quite volatile, and even small doses can cause harm to humans. There’s no medical use for it. In fact, it’s considered a strict laboratory tool, far away from any hospital or household shelf. Still, its unique ability to reliably induce colon cancer in small animals allows for research that wouldn’t be possible otherwise.
Some history helps clarify the situation. Decades ago, scientists noticed the pressing need for models that imitate human cancer. Until then, gaps slowed new treatments because they couldn't test with enough accuracy. Chemicals like 1,2-dimethylhydrazine met that demand. Most research centers have to comply with strict handling procedures, wearing heavy-duty protection and ventilating their workspaces thoroughly. There’s no room for shortcuts.
The Bigger Conversation About Research Chemicals
Every time I read about a substance like this, it reminds me how progress in medicine often depends on tools we'd never use outside the lab. Society has good reason to set boundaries on how we use hazardous compounds. Still, cancer continues to be one of the toughest challenges, and using tricky chemicals has paved the way for new screening techniques, drugs, and even dietary guidance that cuts cancer risk.
One example stands out. Through animal testing with 1,2-dimethylhydrazine, scientists found a clear link between high-fat, low-fiber diets and colon cancer. By running experiments with different diets in these models, they showed how what we eat can impact cancer risk, a message that's reached millions. New medicines, too, have moved from theory to real hope thanks to studies that relied on this hazardous chemical.
Room for Improvements
Tough questions have surfaced about ethical issues and alternatives. More labs have started to replace animal testing with advanced cell cultures or computer-based simulations. Some researchers use genetically engineered mice so that fewer chemicals like 1,2-dimethylhydrazine are needed. A handful of labs now focus almost entirely on reducing animal use and improving safety standards for everyone involved.
There’s always a need to strike a balance between pushing scientific boundaries and protecting both people and animals. Public transparency, independent review boards, and strict government oversight help keep the use of 1,2-dimethylhydrazine where it belongs—deep in the safety of the research lab, working in the service of better health outcomes for all.
What are the health hazards associated with 1,2-Dimethylhydrazine?
Hazardous by Nature
1,2-Dimethylhydrazine often sits out of public view. Most people never hear about it, but lab workers and industrial chemists know it’s dangerous. It’s used in cancer research to induce tumors in lab animals, so its risks aren’t just theoretical – they’re proven science.
Breathing in even small amounts of its vapors can knock you out with headaches, dizziness, or worse. Exposure can strip your airways and lungs, leaving you coughing and gasping for air. I remember walking past a poorly ventilated lab store as a grad student and getting one whiff: I reeled back, eyes burning. It’s more than a bad smell. It’s poison in the air. Skin doesn’t get off lightly either. Touching it directly can cause burns. If you get it in your eyes? You could lose vision, plain and simple.
Cancer Risks That Don’t Stop in the Lab
Scientists rely on 1,2-dimethylhydrazine to create colon tumors in animals for experiments. It’s a textbook carcinogen. If workers get sloppy, or if containers leak, the risk extends straight to the people handling it. I read a study from the Occupational Safety and Health Administration: even chronic low-level exposure raises cancer risk. The molecule attacks DNA, leading to mutations. It’s not just a rare lab oddity — it’s a molecule that can genuinely threaten someone’s long-term health.
Environmental Fallout
Spills matter. 1,2-Dimethylhydrazine escapes easily into air and water. If a drum tips over or a pipe leaks, local soil and streams pay the price. Wildlife isn’t immune. Studies show aquatic animals exposed to runoff develop tumors and reproductive problems. Water treatment plants struggle to break this chemical down, so trace amounts persist in the environment.
Historically, places that used to manufacture rocket fuel (where hydrazines get used) have trouble cleaning up their soil. No amount of sun and rain can erase the mess. Neighboring residents sometimes report odd smells and headaches, only to learn years later that chemicals like 1,2-dimethylhydrazine have invaded their neighborhood groundwater.
What Can Be Done?
Safe handling isn’t optional. Labs need sealed hoods, chemical-resistant gloves, and airtight storage. Industry workers should wear proper masks or full respirators. I learned early on: don’t assume ventilation alone will protect you. Regular checks on equipment and strict protocols help stop accidents before they happen.
Disposal gets tricky. Dumping isn’t an option. Incinerators certified for hazardous chemicals can break it down safely, but they’re expensive. Community awareness needs to grow. Often, local governments and nearby residents don’t know what’s stored next door. Regular public reporting and independent audits of facilities can prevent quiet disasters from building up over time.
Replacing 1,2-dimethylhydrazine in research and manufacturing remains tough. Scientists push for safer alternatives, but change moves slowly. Better funding for green chemistry and stricter regulation make a difference. The health cost of standing still gets paid by real people – it’s a bill no one deserves.
How should 1,2-Dimethylhydrazine be stored and handled safely?
Understanding the Risks
1,2-Dimethylhydrazine packs a real punch. As someone who’s spent years near lab benches and industrial storage rooms, the dangers become obvious pretty quickly. This compound ranks among the more aggressive chemicals, both flammable and toxic. Small spills can release vapors sharp enough to make eyes water and breathing tough. There’s cancer risk with even small exposures—enough of a concern for most regulatory bodies worldwide.
Temperature, Ventilation, and Containment
Anything flammable should stay cool, but 1,2-dimethylhydrazine calls for extra care. Most facilities I’ve seen use dedicated refrigerators with explosion-proof seals, keeping temperatures just above freezing. Leaving a drum or bottle too warm cranks up the vapors, so that’s a line no one wants to cross.
Rigorous ventilation comes next. Standard lab fans don’t move enough air. Walk into storage designed for this chemical, and the exhaust hum stays constant, with ductwork leading outside. No corners cut—every cubic foot of air gets changed out often.
Physical containment matters, too. Metal shelves with spill trays below every unit, raised lips at the edge, and chemical-resistant coatings help catch any leaks. Do not put it over wooden shelves or set on a basic countertop—liquid can chew up surfaces.
Protecting People: Gloves, Goggles, Suits
I’ve watched new staff handle bottles wearing surgical gloves. It’s nonsense. Rubber nitrile gloves, chemical splash goggles, and flame-resistant lab coats, plus sometimes full-face shields, give layers of protection. Accidents happen when people treat the risks casually or use worn-out equipment.
An emergency shower and eyewash station should always stand nearby, never behind a locked door. That extra fifteen seconds makes the difference between a minor scare and a lasting injury. I make it a point to remind people: treat every splash as an emergency, not just an inconvenience.
Fire Safety and Spill Control
For flammable materials, most shops load up on CO2 or dry chemical extinguishers. Sprinklers with water don’t always cut it, since 1,2-dimethylhydrazine reacts with oxidizers and can spread toxic vapors. I’ve seen Class B fire extinguishers stationed only a couple steps from every storage door.
Spill kits come packed with absorbent pads suited for hydrazines, not just any generic chemical mat. Sealable waste drums, labeled for hazardous waste, stand close to every work area. Store everything on near-ground level, since lifting heavy containers from overhead raises the odds of a drop or spill.
Training and Procedures Win Every Time
Best practices rely on muscle memory built through regular safety drills. Workers memorize evacuation routes, spill protocols, and emergency numbers long before any incident happens. Print out those protocols, keep them visible at eye level. Companies that try to cut costs skip this step, and injuries stack up fast.
I prefer facilities that double down on buddy systems—no one handles or transports 1,2-dimethylhydrazine alone. A lone worker faces trouble if a container ruptures or a vapor cloud forms. Two sets of hands and eyes lower the risk dramatically.
Documented inspections finish the loop: check seals, labels, expiration dates, and the structural integrity of each storage drum or bottle. Trust fades fast once someone finds corroded lids or leaking seams.
Better Designs for Safer Workplaces
The future points toward automated inventory systems, zero-spill shelving, and improved detection sensors. Activity monitors and leak alarms already show up in modern sites. No single trick solves every problem, but every layer added shaves down risk.
In my years around these hazards, I’ve seen the gap between shortcuts and real safety. The priority runs deeper than meeting a checklist—it’s about respecting the very real risk this chemical brings into a space and making sure everyone goes home at the end of the day.
Is 1,2-Dimethylhydrazine carcinogenic?
Looking at the Risks with Eyes Wide Open
People don’t walk around thinking about chemicals like 1,2-dimethylhydrazine, unless the word “cancer” steps into the conversation. Used in labs and industry, this chemical keeps showing up in studies connected to tumor growth. Researchers flagged it as a powerful carcinogen decades ago. This isn’t just split hairs over rat studies; many labs worldwide tested it, and the pattern held strong. These animal studies matter because if a compound regularly triggers colon cancer in mice and rats, most scientists take notice. That’s the world I came up in—one where repeated outcomes in controlled settings guide safety decisions.
The International Agency for Research on Cancer (IARC) calls 1,2-dimethylhydrazine “possibly carcinogenic to humans” (Group 2B). That phrase leaves room for scientific caution, but the studies behind it show real danger. This chemical produces DNA mutations, chromosomal breaks, and tumors in lab animals even at low doses. My training in environmental health drilled into me that consistent DNA damage is a flashing sign. The worry isn’t just some bureaucratic label—companies, researchers, and policymakers trust these large toxicology bodies to sift through piles of evidence most of us never see firsthand.
Sneaky Ways Exposure Can Happen
Most people never run into 1,2-dimethylhydrazine at work or at home. But it isn’t locked away in a vault. Scientists use it to create experimental colon cancer in animals, and some industries use chemicals from the same hydrazine family. Accidents, spills, careless storage, or improper disposal could mean real-world exposure. The compound evaporates easily—one whiff, and the lungs and bloodstream can pick it up. Based on what’s come out in case studies, the body absorbs it quickly, and then the damage starts. We trust regulatory controls to keep these substances away from groundwater or food, but leaks happen.
Where Science Meets Policy
I always come back to how exposure gets controlled. Banning a chemical isn’t always practical, especially in research, but adopting airtight storage, strict transport rules, and solid education for any staff working nearby seems like the bare minimum. Strong personal protective equipment—think gloves, goggles, and top-of-the-line ventilation—can make a world of difference.
The U.S. Occupational Safety and Health Administration (OSHA) hasn’t set a Permissible Exposure Limit for 1,2-dimethylhydrazine, but NIOSH’s recommended exposure limit drops to the lowest level practically possible. That tells you how nervous the experts are about even trace amounts. Those rules exist for a reason; small lapses add up, putting chronic exposure on the table.
How Society Should Respond
Public trust depends on keeping people safe from invisible risks like this one. That means staying clear-eyed about the evidence. No company or government agency should cut corners when science calls out a clear danger. Anyone working with chemicals similar to 1,2-dimethylhydrazine, even if not exactly the same, benefits from a culture that prizes safety. That’s the lesson from every case where regulators waited too long to sound the alarm—think asbestos or benzene. Every safeguard we put in place spares people from dangers they might never even know exist.
What are the emergency procedures in case of 1,2-Dimethylhydrazine exposure?
Understanding the Risk
1,2-Dimethylhydrazine turns up in the headlines every so often, often attached to stories of chemical spills or lab accidents. This chemical isn’t like accidentally touching bleach or breathing a bit of dust. It’s highly toxic and can hit the body hard in small doses. I’ve worked in research labs for years and seen how a whisper of the word “hydrazine” snaps people’s attention. No one wants to take the gamble.
Emergency Steps That Make the Difference
If someone breathes in vapors or fumes from 1,2-Dimethylhydrazine, you move them to fresh air as quickly as possible. Forget finishing the experiment. Get them out, loosen tight clothes, make sure there’s nothing constricting their breathing. People sometimes think about “waiting it out” if symptoms seem mild, but this chemical isn’t predictable. Watch for signs like dizziness, headache, and trouble breathing. It hits the lungs and nervous system. This isn’t a job for over-the-counter medicine or coffee—call emergency services straight away. Paramedics have the gear and training to handle these cases.
For skin contact, speed matters. Peeling off any contaminated clothing is the first thing to do — including shoes and jewelry. Don’t wait until you’re “in the clear”; scrub with soap and water right away, for at least 15 minutes. I remember a colleague who tried to wash off a chemical burn with just water in a rush—soap actually makes a difference because it helps break down the oily texture of many chemical spills. Don’t use solvents like alcohol—these can pull the toxin deeper into the skin.
If the chemical gets into someone’s eyes, get to an eyewash station immediately. Flood the eyes with water for at least a quarter hour. It sounds tedious but it’s crucial. I’ve seen training exercises where folks flinch at the cold water, but there’s no time for comfort. Removing contact lenses, if they’re there, should be done carefully so you don’t spread the chemical further.
Accidental swallowing? It’s rare, but it sometimes happens with lab mistakes. Don’t try to induce vomiting or give anything to drink unless the emergency team tells you to. My old workplace forbid eating and drinking in the lab for this exact reason—chemicals linger on benches and clothing long after “cleanup.”
Preventing a Disaster
Proper ventilation, gloves, goggles, and lab coats are more than window dressing. Chemical fume hoods suck up dangerous vapors before they reach your lungs. Double-layering gloves, switching to goggles that seal around your face, and keeping a respirator nearby all add layers of safety. Facilities that respect these rules see fewer accidents—simple as that. The story after the headline spills rarely mention the countless close calls avoided by following these steps.
Why This Matters
Knowing the steps matters because lives and futures are on the line. 1,2-Dimethylhydrazine is no one’s friend and doesn’t give second chances. Ignoring training, skipping personal protective gear, or hesitating to call for medical help isn’t just careless—it’s dangerous. In the labs and industries using these chemicals, it’s everyone’s job to stay sharp, protect their coworkers, and remember that emergency steps save lives before the professionals even arrive.
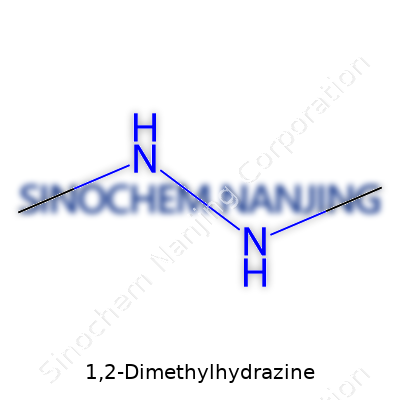
| Names | |
| Preferred IUPAC name | 1,2-dimethylhydrazine |
| Other names |
Sym-Dimethylhydrazine UDMH UN 1163 |
| Pronunciation | /ˌwaɪ.tuː.daɪˌmɛθ.əl.haɪˈdreɪziːn/ |
| Identifiers | |
| CAS Number | 540-73-8 |
| Beilstein Reference | 1361085 |
| ChEBI | CHEBI:34961 |
| ChEMBL | CHEMBL15733 |
| ChemSpider | 69578 |
| DrugBank | DB04230 |
| ECHA InfoCard | 03d6aef8-bf04-4d63-9a51-698a4a16de3c |
| EC Number | 200-143-0 |
| Gmelin Reference | 8214 |
| KEGG | C06572 |
| MeSH | D004107 |
| PubChem CID | 1576 |
| RTECS number | UB0700000 |
| UNII | 6QI9V4520S |
| UN number | UN1163 |
| Properties | |
| Chemical formula | C2H8N2 |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless liquid with an ammonia-like odor. |
| Odor | Ammonia-like |
| Density | 0.829 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | 0.13 |
| Vapor pressure | 40 mmHg (20°C) |
| Acidity (pKa) | 7.6 |
| Basicity (pKb) | pKb 3.46 |
| Magnetic susceptibility (χ) | -27.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 0.81 cP (20 °C) |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 198.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 79.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2835 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | V03AW32 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H350 |
| Precautionary statements | P210, P260, P264, P270, P271, P280, P301+P310, P302+P352, P304+P340, P308+P311, P311, P312, P321, P330, P361, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2-Po |
| Flash point | 35 °C (95 °F; 308 K) |
| Autoignition temperature | 228 °C |
| Explosive limits | 2.5% - 23% |
| Lethal dose or concentration | LD50 oral rat 79 mg/kg |
| LD50 (median dose) | LD50 (median dose): 79 mg/kg (oral, rat) |
| NIOSH | NIOSH=PA8050000 |
| PEL (Permissible) | PEL: 0.5 ppm (2 mg/m³) |
| REL (Recommended) | 0.09 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,1-Dimethylhydrazine Hydrazine Monomethylhydrazine |

