Looking Beyond the Label: The Real Story of 1,2-Dichloropropene
Tracing Its Roots: A Chemical Born of Necessity
The story of 1,2-dichloropropene reflects the tangled path of agricultural chemistry over the last century. Early on, researchers noticed their crops withering thanks to pests in the soil. As chemical giants searched for solutions, they homed in on this molecule, synthesized as a byproduct during the manufacturing of allyl chloride and propylene oxide. It moved quickly from laboratories to the furrows of farm fields, echoing a time when few asked tough questions about what really went into pest control. Farmers saw it as a breakthrough, saving them weeks of labor and boosting yields. Not until communities began to ask what price went along with those easy wins did the conversation begin to shift.
Physical Identity and Chemical Behavior—Not Just a Generic Liquid
Crack open a drum of 1,2-dichloropropene and the sense memory lingers—a sweet, sharp scent with a weight that hangs in the air. It boils at around 104°C, slips easily between liquid and vapor, and has a density heavier than water. It doesn’t cling to a simple destiny; volatile yet persistent, soluble enough to move through moist soils, yet reactive enough to surprise. Its structure, two chlorine atoms clinging to a three-carbon backbone, makes it ready to join all sorts of chemical shuffles, whether breaking bonds in the environment or inside the bodies of organisms it touches.
Labeling Tells Just Part of the Story
Looking at a label, you’ll find nothing poetic. Trade names crowd out the plain chemical title, and product descriptions keep things basic. Regulations demand that containers flash their warnings—hazard statements, pictograms, and emergency instructions. Yet behind those symbols, most users don’t see the processes that shape these guidelines, nor the studies that drive governments to tighten or relax regulatory nooses. Interpret labels is a learned skill, not just for safety, but to cut through the technical talk and see the broader risks and responsibilities at play.
How It’s Made and What That Means for Industry
Chemists make 1,2-dichloropropene mainly by chlorinating propylene. It’s an efficient process, but also a reminder of how closely our agriculture and heavy industry intertwine. The production brings a mix of isomers and other byproducts, which separations and purifications sort into saleable material. Each step adds complexity and cost, and each brings opportunities for leaks, spills, or worker exposures. I remember walking through a plant’s alleyways, smelling that faint chlorinated aroma, and thinking how easy it would be for a valve or seal to falter, releasing more than anyone expected.
Chemical Workhorse: Reactions and Adaptations
1,2-dichloropropene isn’t just a pest-control tool. Its double bond makes it reactive, a sort of chemical multitool every industrial chemist knows. In the soil, it unravels nematode populations, yet that same reactivity means it doesn’t sit quietly underground. Microbes and the environment break it down—sometimes into harmless fragments, other times into substances that raise their own concerns. In the lab, it gets used to prepare other chemical products, but outside controlled settings, it’s hard to pin down what new compounds will form and whether any are more toxic or tenacious than the original.
Names, Names, and More Names
Few chemicals collect as many aliases. Folks may know it as Telone or Nematox, and still others by the simple "soil fumigant." These synonyms reflect both branding ambitions and regulatory requirements, each trying to bottle up a compound with many hats. Sorting through the names is more than an academic exercise—different markets, different crops, and different regulators may treat a product in their own way, and the paperwork can quickly snarl up anyone trying to trace how, where, and why the chemical moves across borders and counties.
Staying Safe: Standards That Came Late
Like so many industrial chemicals, operational standards for 1,2-dichloropropene evolved after big mistakes. Workers exposed in poorly ventilated fields or storage sheds showed regulators the gaps between what was supposed to happen and what actually did. National agencies now dictate ventilation, protective gear, and restricted entry intervals after use—but these rules came on the heels of sickness, not foresight. Even today, rules vary sharply, and enforcement doesn’t always keep pace with paperwork. Walking with farmworkers during application, it's clear that written protocol sometimes bumps up against old habits, cost-saving shortcuts, or the rush to beat the rain.
Fieldwork and Beyond: Where 1,2-Dichloropropene Shows Up
From carrot farms in California to tobacco fields in the Southeast, 1,2-dichloropropene pulls its weight as a go-to soil fumigant. For crops struggling with nematodes and certain fungal threats, it offers a fast fix that few alternatives can match, especially where high-value plants grow in rotation or on tight margins. Occasionally, it finds a role in chemical manufacturing outside agriculture, but its identity has been shaped by the seasons and rows of commercial farms. As organic and regenerative methods spread, some regions turn away from it—but for many commodity growers, it remains the most practical choice on the market, despite tough questions about its long-term impact.
Not Just an Industrial Relic: Research Points Toward Change
Academic and corporate labs keep revisiting 1,2-dichloropropene, hunting for ways to make it less hazardous or unnecessary altogether. Recent studies dig into microbial degradation in soil, trying to pinpoint strains that might clean up residues before they reach water supplies. Others examine alternative compounds, or even plants engineered to resist nematode damage naturally. Still, a lot of research gets tangled up in funding debates, with big players hesitant to lose a product that’s profitable and politically sensitive. If a better option emerges, it will likely come from a mix of regulation, public demand, and genuine innovation.
Chasing Safer Science: What Toxicity Studies Show
Toxicologists have poked and prodded 1,2-dichloropropene for decades and the story remains complicated. Acute exposure can irritate lungs, skin, and eyes in workers. At higher doses or chronic exposure, animal studies raise red flags about cancer, mutagenesis, and organ toxicity. Real-world risk depends on how and where people encounter it—so drift from fumigated fields worries nearby residents, not just applicators. Monitoring wells near use sites sometimes detect residues, and that keeps health departments vigilant. Researchers keep gathering data, sometimes reaching conflicting conclusions, since field realities rarely match the tidy boundaries of a study.
By the Numbers: Technical Realities and Regulatory Gaps
Looking at the technical side, manufacturers list specifications like purity, isomer ratio, and stabilizer additives. Over time, batches got cleaner, but impurities remain an issue. Regulators try to keep a lid on unsafe levels, especially in food crops and groundwater, yet loopholes persist. Some regions have banned or curtailed use after local contamination episodes, pressuring growers to find substitutes, while others lag behind. It’s tough to find a uniform approach between states or countries, and traders often shuffle materials to markets with looser controls, keeping the cycle alive.
What’s Next: Pushing the Conversation Further
Moving forward, the fate of 1,2-dichloropropene will ride the wave of public health research, environmental activism, and agroscience breakthroughs. New soil treatment methods—solarization, biofumigation, or biological control—offer promise, but none have wiped out the need for chemical fumigants, especially where profit margins push riskier decisions. More attention to worker protection, buffer zones, and transparent labeling would help, but the deepest changes depend on shifting the balance away from reactive fixes and toward prevention through soil health and crop rotation. For those who work the land or watch their kids breathe the dust downwind, the story of 1,2-dichloropropene feels like a test of how much risk a society accepts in the bargain for abundant harvests. Real progress will depend on keeping facts at the front, holding industry and regulators accountable, and demanding solutions that leave fewer scars for the next generation.
What is 1,2-Dichloropropene used for?
What Farmers Count On
1,2-Dichloropropene is no household name, though people in agriculture have relied on it for decades. For those growing carrots, potatoes, or tobacco, fields need more than sun and rain. Nematodes—microscopic worms—wreak havoc under the soil, damaging roots and eating into yields. Farmers look for tools to fend off these invisible enemies, and they often turn to 1,2-Dichloropropene. It works as a soil fumigant, injected directly into the ground before planting. The chemical targets pests that burrow deep, where few other treatments reach. Without that help, big portions of crops could disappear, threatening a farmer’s whole year.
The Chemistry Behind the Scenes
This compound doesn’t sound special, but it’s got a backbone of two chlorine atoms tacked onto propane, and that makes it effective. Chemistry textbooks might talk about reactivity, but farmers see results in the form of healthier, stronger plants. Other lines of work, like turf management for golf courses, have also found uses for it. Healthy soil means more than just green grass. A professional in the business once told me, “Root knot nematodes don’t care if it’s a farm or fairway—they just eat.” The demand comes from a simple truth: if people want reliable harvests, they look for solutions that work below ground before any seed goes in.
Health and Environmental Questions
Using 1,2-Dichloropropene comes with safety questions. The chemical moves fast in soil and air, and while it breaks down over time, there’s always a risk it might drift after application. Respiratory issues and skin irritation can be real problems for workers handling it directly. California’s Department of Pesticide Regulation, for example, sets strict limits on use, requiring buffer zones and training for applicators. Researchers track its presence in water and air, finding it when people—or pets—could be at risk. No one wants to see a jump in hospital visits during fumigation season, and oversights don’t just endanger the person spraying. Fact: the US Environmental Protection Agency reviews soil fumigants every few years, weighing science against community health and farm needs. Figuring out the right balance isn’t easy.
Alternatives and a Way Forward
Many growers talk about finding ways out of chemical dependence. Cover crops like mustard or marigolds add a natural barrier to pests, choking off nematodes before they start. New tech offers biological controls using beneficial bacteria or fungi. Integrated pest management—using a mix of natural and chemical tools—gives farmers breathing room to rotate away from heavy-duty chemicals. Switching takes patience and often brings a learning curve. One organic grower told me, “I lost a lot those early years, but the soil looks better now, and there’s less worry for my crew.”
People want safe food and healthy land. Decisions about chemicals like 1,2-Dichloropropene need the input of scientists, farmers, consumers, and regulators. By leaning into field data and the daily realities of growing food, progress stays grounded. Healthy farms grow from both good science and daily grit.
Is 1,2-Dichloropropene hazardous to human health?
What is 1,2-Dichloropropene?
1,2-Dichloropropene often shows up in agriculture as a soil fumigant. Growers use it to kill nematodes and other pests before planting crops. Anyone who has spent time around farmland knows about the drive to keep pests at bay to protect yields. That chemical edge comes with risk, though, and not just for bugs.
Long-Term Health Risks
Many chemicals drifting across fields linger in the air, soil, or water for longer than most of us realize. This one vaporizes easily and drifts off target. Farmers and workers can breathe vapors or pick up residue on their skin, especially during application seasons. The World Health Organization and U.S. Environmental Protection Agency both see its long-term effects as serious. Studies link it with lung and stomach irritation, headaches, and dizziness at first. Animal tests suggest ongoing exposure may lead to liver or kidney damage. Some data tie it to higher cancer risk, particularly for folks who work around it as part of their job.
Concerns For The Wider Community
Rural families living near fields treated with fumigants often share their stories about odor complaints and unexplained health symptoms. Children playing outside, older folks sitting on porches, even pets: none of them sign up for chemical drift, but they still get exposed. Water sources that run near treated fields can carry traces of 1,2-dichloropropene. That means communities relying on wells face added risks if groundwater gets contaminated.
Regulation and Reality
Regulators in different countries judge these risks and set rules on how much farmers can use, how close to homes or water sources application happens, and how long people must wait before reentering a treated field. California and the European Union keep stricter controls because their health agencies reviewed available data and found patterns that raise red flags.
I’ve known farm workers who don protective suits and respirators before applying soil fumigants. Even with that gear, stories circulate about headaches and nausea, especially after a long day in midsummer heat. The EPA classifies this chemical as a possible carcinogen. That label alone grabs the attention of both growers and consumer advocates.
Better Solutions For Safer Fields
Addressing this hazard means more than just tighter regulations. Some growers look for ways to reduce reliance on chemical fumigants altogether. Rotating crops breaks pest cycles naturally; cover crops help restore soil health. Biological controls—relying on beneficial organisms to beat back nematodes—keep gaining ground. Technology helps, too: precision application techniques and better weather forecasting limit drift and cut down on waste.
Education plays a big role here. Workers get trained on safe handling. Neighbors learn about scheduled applications, so they can stay inside or close windows. Some communities push for buffer zones or outright bans, especially near homes or schools.
Everyday Choices Matter
Labels on produce or local ordinances can seem disconnected from everyday life. In reality, the stories behind them matter. Public pressure, consumer demand for safer foods, and new policies all grow out of concern for what we breathe, drink, and eat. My own visits to farming communities left me certain: folks care about protecting their health and the land they live on. By acknowledging the risks of chemicals like 1,2-dichloropropene, we push towards solutions that keep both harvests and people safe.
What are the proper storage and handling precautions for 1,2-Dichloropropene?
Why Care About 1,2-Dichloropropene Safety?
Anyone who works on a farm or in a chemical plant may come across 1,2-Dichloropropene. It often shows up in soil fumigation, especially in the fight against nematodes. This colorless liquid packs a punch: health risks, environmental dangers, and the kind of volatility that makes a splashy headline after an accident. Folks sometimes overlook chemical storage because it seems straightforward, yet this is where trouble often starts. I’ve seen sheds, barns, and back rooms turned into risky spaces because a drum or container leaked or overheated.
Proper Storage: More Than Just Putting a Barrel on a Shelf
Chemicals like 1,2-Dichloropropene do not forgive sloppy storage. Keep containers locked tight and make sure the labels are clear and legible—no peeling or fading. Conditions matter just as much as containers. This liquid does not like heat. Temperatures above 40°C drive up the pressure inside drums, and leaks or ruptures happen fast. Store the containers in a cool, shaded spot out of direct sunlight. Concrete floors and metal shelving work better than wood, which soaks up any spills.
Avoid stacking containers unless a chemical safety consultant gives the green light. Store this chemical away from oxidizers, acids, alkalis, and food storage. I once heard about two barrels—a fertilizer and a fumigant—parked side by side in a shed, and it didn’t end well. Separate storage really does save lives.
Personal Protection and Ventilation
Cracking open a drum or transferring product raises real health risks. 1,2-Dichloropropene can irritate the eyes, nose, and respiratory tract. Prolonged exposure links to more serious effects, including damage to the liver and kidneys and a possible cancer risk. Don’t rely on shortcuts such as wrapping a scarf over your face. Workers need splash-proof goggles, chemical-resistant gloves, long sleeves, and, whenever vapor is present, a proper respirator rated for organic vapors.
Good airflow keeps vapors from building up to dangerous levels. Work outdoors or in well-ventilated areas. Indoor work? Local exhaust ventilation systems make all the difference. There’s nothing worse than a hidden vapor cloud sneaking up on someone fixing a leaky valve.
Emergency Response Matters
Spills happen, even with the best plan. Quick action keeps a minor leak from turning into a costly cleanup or medical disaster. Powdered absorbents such as vermiculite can lock up a spill, but don’t sweep vapors into drains or water sources. Every worker should know where to find emergency showers, eyewash stations, and spill kits. If symptoms start—cough, headache, dizziness—move to fresh air and seek medical attention right away.
Fire risk remains at the top of the list. This chemical flashes at relatively low temperatures, so keep fire extinguishers handy—rated for liquid chemicals. Never use water jets, as they tend to spread the material or drive vapors into the air.
Building Safer Habits
Rules and warning labels become second nature with enough practice and real talk among coworkers. Training—real, hands-on training, not a dusty binder on a shelf—teaches people to spot trouble and respond before a situation spins out of control. All it takes is one moment of carelessness to see how dangerous shortcuts can be with 1,2-Dichloropropene. Safe storage and smart handling protect workers, communities, and the land we all rely on.
What are the main routes of exposure to 1,2-Dichloropropene?
The Fumigant in the Field
Working in agriculture means dealing closely with soil treatments. 1,2-Dichloropropene, or 1,2-DCP, shows up in this world as a strong soil fumigant, mostly to control nematodes before planting crops. For anyone walking a field right after application, the chance to breathe in vapors runs high. Farmers and workers pulling hoses, checking plastic tarps, or handling empty containers pick up the bulk of their exposure through what they breathe. There’s no getting around how much open air application increases what ends up in the lungs.
Some folks use fumigants without thinking about the wind or shielding gear. Short term, the irritation comes quick—throat burn, watering eyes, headaches. I remember a neighbor who forgot a mask and spent a whole day feeling like he’d swallowed sandpaper. Long term, there’s data showing risk bumps for respiratory problems and possible cancer. The U.S. Environmental Protection Agency classifies 1,2-DCP as a probable human carcinogen, backing this up with years of animal studies.
Bringing Home Pesticides on the Skin and Clothes
Rural living sometimes blurs work and home life. After finishing in a treated field or at a loading dock, residue can stick to boots, overalls, or bare skin. Rushing to lunch without stripping down lets chemicals hitch a ride onto kitchen chairs and car seats. Dermal exposure happens every time a splash misses a glove, or when mixing the chemical with bare hands. Some people underestimate tiny spills; studies have picked up high concentrations around collars, sleeves, and inside gloves from sweat.
Washing up and changing before coming home helps a lot. Based on what I’ve seen, better locker setups at barns or field stations make a real difference. In places where workers have nowhere to wash, the chemical keeps spreading to families and pets. California’s field studies suggest tracking dust and vapors into homes can add up, almost like secondhand exposure.
Public Exposure Through Air and Water
Living close to treated fields feels different on the nose. On windy days or near application season, a faint, sharp odor sometimes wafts across rural neighborhoods. People inside those homes, especially children, can pick up exposure by breathing indoor air or playing outside right after fumigation. Drift—the spread of chemicals outside target fields—means nobody on the edge of a farm really escapes the risk.
1,2-Dichloropropene can seep below the soil surface and move into water supplies. Studies in farming regions of the Midwest have flagged shallow groundwater with traces of this chemical. The number might be small, but that’s all it takes when families use wells year-round.
What Can Keep People Safer
Wearing protective equipment helps workers most, but employers have to supply it and enforce its use. Harvesters and applicators, from my experience, prefer gear that’s comfortable and easy to put on—otherwise, it sits unused. Laws requiring buffer zones, weather checks, plus strict storage make a dent in unnecessary exposure. Researchers call for safer substitutes, but until then, community awareness and regular medical checks play a huge part. If there’s a responsibility here, it falls on employers, regulators, and neighbors working together, sharing honest information, and taking science seriously.
How should 1,2-Dichloropropene spills or leaks be managed?
Why 1,2-Dichloropropene Poses Real Risks
1,2-Dichloropropene sits among a family of chemicals widely used in farming, especially for controlling nematodes in soil. When spilled or leaked, it doesn’t just disappear. This stuff quickly turns into a problem for both people and the environment. The health impact hits first. A strong smell, watery eyes, headache, and sore throat often show up fast. Frequent or heavy exposure can grind down lungs and nerves over time. The chemical seeps into soil and slips down to groundwater, threatening what comes out of taps or irrigation lines. In the air, vapors can catch a spark and ignite flames no one in a rural or farm setting wants to see. These facts give us enough reason not to treat a spill as just another routine mess.
Handling the Reality: Training and Speed Matter Most
In my time helping out on family fields and working around agricultural chemicals, I saw firsthand how some folks want to “deal with it later.” That approach never ends well. With 1,2-Dichloropropene, fast action isn’t an option—it’s the law if you care about safety. Workers handling the cleanup need real training. Gloves, goggles, and tight-fitting masks make a difference. I once watched a neighbor skip a mask and regret it within minutes. The importance of protective gear can’t be oversold.
Act quickly. Any leak or spill should set off a response plan: get people out of the area, rope off the scene, and call trained help if the spill looks bigger than what you’d handle with a mop and a bucket. Never touch it with bare hands or try to wash it down a drain. No one wants that chemical in local creeks or drinking water.
Stopping Spread and Protecting Water Sources
Soil can hold on to chemicals like 1,2-Dichloropropene, but that doesn’t mean it’s safe underfoot. On a dry day with steady wind, vapors travel. Keeping the chemical contained—using absorbent pads, sand, or dirt—works better than trying to sweep it away. Shovels or brooms should go straight into a chemical waste container after use. If any got loose, especially after heavy rain, testing wells for contamination makes sense. The EPA and local water boards post actual numbers on threats to groundwater, showing that even small spills leave a mark if ignored.
Prevention and Clear Accountability
Accidents slow down with better storage. Tanks and containers deserve regular checks for leaks—no exceptions. Warning signs marking storage areas let everyone know to be careful. In my hometown, a neighbor’s diligence paid off: a leak from a cracked valve got caught early, dodging what could have become a water emergency. That awareness and care helped the whole community.
If 1,2-Dichloropropene gets loose, it’s nobody’s secret. Local and federal agencies—like the fire department or EPA’s emergency spill line—expect quick, honest reporting. All workers should know who to call and what steps to take, long before disaster forces a learning curve.
What Builds Real Trust
Communities put faith in farms and businesses that show respect for health and land. Clear protocols, honest reporting, and respect for federal rules build real trust. Not by talking up safety, but by showing it day after day. I’d trust a neighbor or business who treats chemical safety as more than just paperwork. That’s the neighbor who keeps water clean, air clear, and the local kids healthy.
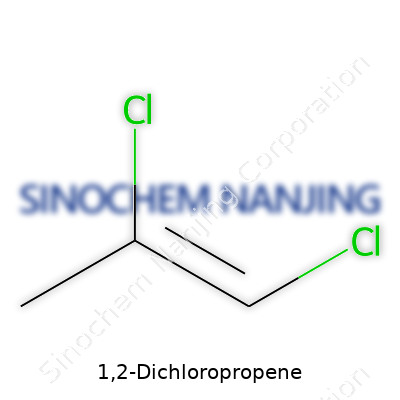
| Names | |
| Preferred IUPAC name | 1,2-dichloroprop-1-ene |
| Other names |
1,2-DCP Telone Nematox Nematocide Propylene dichloride 1,2-Dichloropropylene |
| Pronunciation | /ˌdaɪˌklɔːrəˈproʊpiːn/ |
| Identifiers | |
| CAS Number | 563-47-3 |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:36940 |
| ChEMBL | CHEBI:17444 |
| ChemSpider | 6802 |
| DrugBank | DB11128 |
| ECHA InfoCard | 03c060d7-cc7d-4147-b8fc-50f1af120b1d |
| EC Number | 204-892-4 |
| Gmelin Reference | 100162 |
| KEGG | C01460 |
| MeSH | D004039 |
| PubChem CID | 7906 |
| RTECS number | GZ0350000 |
| UNII | LYG7838PQC |
| UN number | UN2047 |
| Properties | |
| Chemical formula | C3H4Cl2 |
| Molar mass | 110.98 g/mol |
| Appearance | Colorless to amber liquid with a sweetish, chloroform-like odor |
| Odor | sweet pungent odor |
| Density | 1.17 g/mL at 25 °C (lit.) |
| Solubility in water | 2.35 g/100 mL (20 °C) |
| log P | 1.98 |
| Vapor pressure | 341 mmHg (20°C) |
| Acidity (pKa) | 11.72 |
| Magnetic susceptibility (χ) | -63.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.419 |
| Viscosity | 0.855 cP (20 °C) |
| Dipole moment | 2.08 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 290.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −54.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2508.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H331, H341, H351, H410 |
| Precautionary statements | P210, P261, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P501 |
| NFPA 704 (fire diamond) | 3-2-1 |
| Flash point | 23 °C (closed cup) |
| Autoignition temperature | 384°C |
| Explosive limits | 7.0–14.5% |
| Lethal dose or concentration | LD50 oral rat 1235 mg/kg |
| LD50 (median dose) | LD50 (median dose): 300 mg/kg (oral, rat) |
| NIOSH | NIOSH: TQ2690000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,2-Dichloropropene: "1 ppm (5 mg/m3) TWA |
| REL (Recommended) | 5 ppm (18 mg/m³) |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Allyl chloride 1,3-Dichloropropene Epichlorohydrin Chloropropanes Chloroethenes |

