Commentary: 1,2-Dichloroethyl Ether — From Lab Curiosity to Industrial Controversy
Looking at the Past: The Path to 1,2-Dichloroethyl Ether
Digging back through chemical history, scientists have always looked for new compounds that reshape what is possible in industry and medicine. 1,2-Dichloroethyl ether cropped up in the wake of chlorinated organic chemistry’s rapid growth after the mid-19th century. Once researchers understood the basic reactions between chloroethanols and diaryl ethers, it wasn’t long before this molecule hit the laboratory benches. These early discoveries often started as academic projects or government-funded programs, before they spilled over into manufacturing as the reach and curiosity of chemistry widened. The path that 1,2-dichloroethyl ether took — from synthetic curiosity to more commercial use — reflects both the open challenges and willingness to pursue new molecules, despite safety questions that shadowed many chlorine-based organics.
Understanding What It Is: Product Overview
Those who have spent hours in the lab know that 1,2-dichloroethyl ether represents a specific slice of chloroalkyl ethers, a group prized for both their reactivity and volatility. Much of its early adoption tagged along with the rising use of chlorinated solvents that shaped everything from plastics to pesticides. This compound, with two chlorine atoms hanging off an ethyl ether backbone, always carried a certain edge in chemical performance, but at a cost — and anyone who's handled it recognizes that distinct aroma and stubborn persistence on glassware. Industrial chemists saw opportunity in its solvent properties and its behavior as a chemical intermediate, especially in the decades before tougher regulations closed in on volatile and toxic compounds.
What We See and Smell: Physical & Chemical Realities
Handling 1,2-dichloroethyl ether in the flesh brings those textbook facts to life. This molecule stands as a clear, colorless liquid, heavier than water and rolling out a sharp, ether-like odor that lingers in any workspace. Its boiling point hovers at a moderate level, adding a layer of volatility that speaks to its risks and utility. The absence of color often tricks newcomers into underestimating it, but seasoned researchers know not to ignore its chemical punch. In my experience, any substance that doesn’t play nicely with strong bases, acids, or oxidizing agents draws a mental red flag. It has a wicked flammability and not much patience for careless mixing, and that underlines every safety procedure you drill into students or lab staff.
Technical Specs and Real-World Labeling
Out on the market, 1,2-dichloroethyl ether never arrives in unmarked containers — not if you’re following proper standards. Legible, prominent labeling isn’t just regulation compliance; it keeps accidents from turning into horror stories. Technical specifications detail everything from minimum purity (usually above 99 percent for demanding synthesis) to maximum permissible levels of related impurities, stabilizers, and water contamination. There’s always a lot of chatter about bottle size, material compatibility, and shelf life, but the real weight sits with how closely users adhere to the recommended storage conditions, since poor handling leads to unwanted byproducts and dangerous decomposition.
Digging Into How It’s Made
Unlike some compounds that drop out of one-pot wonders, 1,2-dichloroethyl ether calls for a calculated process. Industrial practice leans on the chlorination of ethyl ether, a reaction that walks a fine line — too much heat or miscalculated ratios ramp up the risk of runaway reactions or byproduct messes. The selectivity depends heavily on catalysts, temperature controls, and timing. Research chemists used to tinker endlessly with these variables to dial in better yields or fewer chlorinated byproducts. Every time someone tries to cut corners or skips on monitoring, the process reminds them that chlorinated ethers are not forgiving. It’s the kind of thing you learn from bad experience rather than textbooks.
Chemical Reactions — More Than Just Theory
Get past the preparation and you confront a world of chemical behavior. 1,2-Dichloroethyl ether’s structure — that ether linkage paired with chlorines — pushes it into a versatile, if hazardous, space. In a synthetic setting, the molecule steps up as a potent alkylating agent, introducing its backbone into new organics, especially where other ethers would fall short. This reactivity makes it a double-edged sword: great for pushing reactions forward, terrible if it means stray contamination or unintended modification of sensitive compounds. In all my years, chemists only trust it with proper glass and plenty of ventilation, always staying mindful about which reagents hang around and what byproducts might sneak out.
Other Names — Synonyms and Industry Terms
Look up 1,2-dichloroethyl ether in any chemical index and you’ll see how many languages chemists speak. Names like Ethylene dichloride, Bis(2-chloroethyl) ether, and Cloral essentially trace back to the same basic skeleton, with only small tweaks in attachment or focus. Speaking plainly, anyone who works with chlorinated organics develops a healthy sense of skepticism for synonyms, since they often mask regulatory differences or carry hidden histories of past incidents and research. The variations in nomenclature might seem trivial until someone mixes up their compounds under pressure, and that’s where clear labeling and training earn their keep.
Staying Safe and Getting it Right — Safety & Operational Standards
1,2-Dichloroethyl ether doesn’t forgive inattention. Industrial safety rules, standards from OSHA and European agencies, didn’t appear out of thin air — they answer to a history of injuries and environmental incidents. Personal protective equipment is not a suggestion: full-face shields, chemical-resistant gloves, and impervious lab coats go from luxury to necessity the minute a spill looms. Engineering controls push for fume hoods, tight-sealing containers, and proper waste disposal setups, since vapor inhalation and direct skin contact pose real risks. It’s tempting, especially in tight-budget operations, to try shortcuts with ventilation or storage, but the cost hits sharply when accidents happen. My own experience lines up with this — following procedures strictly pays off in the long run, both for worker health and regulatory peace of mind.
Out in the World — Where 1,2-Dichloroethyl Ether Goes
Industries once reached for 1,2-dichloroethyl ether as a go-to solvent and reaction intermediate, especially before green chemistry standards tightened. It showed up in the synthesis of specialty resins, precision-engineered plastics, pharmaceutical intermediates, even select agrochemicals. Despite regulatory limits, certain sectors persist in controlled uses, driven by either lack of alternatives or specific performance needs. Each new application opens old questions: can processes fit into safer, cleaner frameworks, or does sticking with legacy chemicals mean repeating past mistakes? In labs and on factory floors, anyone over a certain age remembers chems like this not just for their effectiveness, but for the lessons learned in spilled barrels and nervous safety drills.
Research Pushing Forward — R&D Efforts and Trends
Scientists keep searching for safer, more sustainable alternatives, but also for ways to handle legacy chemicals like 1,2-dichloroethyl ether with less risk. University and industry research teams chase after improved synthesis routes that minimize toxic byproducts or ramp up selectivity, often under pressure from environmental watchdogs. Efforts to find drop-in substitutes in high-value reactions tie into the move toward greener chemistry, as do studies into materials and catalysts that can temper this compound’s volatility and toxicity. From where I stand, research into remediation — figuring out how to break down or recycle hazardous chlorinated ethers — matters just as much as discovering the next new reaction, especially as regulatory rules grow tighter year by year.
Facing the Dark Side — Toxicity and Health Impacts
Stories about the dangers of working with chlorinated ethers like 1,2-dichloroethyl ether crop up repeatedly in toxicology literature. Inhalation brings headaches, dizziness, and nausea, but the real scars come long-term, as studies link chronic exposure to liver and kidney damage, disturbances in the nervous system, and possible carcinogenic risk. Animal research, and grimly enough, accident reports, show how insidious low-level exposure proves over months and years. I’ve seen too many cases where minor spills, ignored fume control, or poor personal hygiene added up to serious health issues by the time anyone realized the cause. Real safety comes from more than paperwork; it demands relentless attention and ongoing review as new data arrives.
Looking Ahead — Future Prospects and Hard Choices
Every new round of regulation and research on 1,2-dichloroethyl ether adds pressure for change. As scientific understanding about environmental persistence and health impact deepens, chemical companies and universities face tough calls: phase out legacy chemicals, or invest heavily to engineer away risks? The search for greener solvents and safer intermediates, spurred by REACH and EPA initiatives, stands as both challenge and opportunity. Drop-in replacements remain hard to find for niche uses, but innovation never sits still — catalytic methods advance, and process engineers keep hammering away at containment and remediation. No one in the field doubts demand for cleaner, safer chemistry will keep rising, but for now, legacy chemicals like 1,2-dichloroethyl ether remind everyone of both the progress made and the work left to do.
What are the main uses of 1,2-Dichloroethyl Ether?
Hidden Hand in Industry
People outside labs rarely hear about 1,2-Dichloroethyl Ether. This colorless liquid plays a supporting role in several chemical processes. Its main task involves helping chemists separate and purify materials on a large scale. Many solvents could do the job, but this one stands out for strong solvent power and the ease it brings to reactions where water just makes everything worse. In my time shadowing chemists, I’ve seen it help pull apart sticky, complicated blends that no other solvent could tackle.
Specialty Uses in Production
Few companies advertise their reliance on this compound. In pharmaceutical manufacturing, it assists in extracting active ingredients from complex plant matter. Its chemical properties make it suitable for grabbing and transferring molecules others leave behind. Pesticide workers and agrochemical labs often depend on it during research. Without reliable solvents, half the crop protection products lining hardware store shelves would never make it through testing.
Role in Synthesis and Engineering
Organic synthesis often messes with tricky reactants that refuse to play nice with water. Here, 1,2-Dichloroethyl Ether steps in. It bones up certain reactions, making them run faster and produce purer results. A few specialty plastics and resins owe their existence to processes where this ether makes links between molecules possible. I watched a pilot plant engineer call it their “secret sauce” when coaxing oddball polymers out of finicky chemicals.
Behind the Scenes in Research
Labs don’t use 1,2-Dichloroethyl Ether for show. Chemists choose it to clean up extracts, strip impurities, or cool volatile mixtures during analysis. Synthesizing delicate compounds—think experimental drugs—sometimes fails without a solvent like this. Some test kits for laboratories run smoother with its help, especially on sensitive chromatographs that need separation fast and clean. I’ve seen research teams hesitate to swap it out, worried about losing data quality.
Hazards and Health Concerns
Serious talk comes with these chemicals. 1,2-Dichloroethyl Ether brings dangers, including vapors that irritate eyes, lungs, and skin. Mishandling means breathing risk, fire, or nasty environmental effects. My time in a university lab drilled safety protocol: proper fans, prompt cleanup, and protective gear. No shortcuts. This is partly why you don’t find this solvent outside professional settings—safety matters too much and substitutes are often safer, even if less efficient.
Moving Forward Safely
Many in the chemical world rethink old solvents because the toll on human health and the environment grows clearer every year. Companies push for greening up, swapping out hazardous materials for newer, less toxic choices. Regulations get tighter, urging more caution and transparency. At the same time, research chemists still count on 1,2-Dichloroethyl Ether for several tasks. Future breakthroughs may finally offer a replacement, but for now, its niche tools matter. The challenge comes down to careful use, strict safety, and adapting as better options appear.
What are the health hazards associated with 1,2-Dichloroethyl Ether?
Understanding The Chemical
Most people have never heard of 1,2-dichloroethyl ether. It hides in the long list of industrial chemicals, far away from the kind of thing you’d find in a kitchen or garage. In labs and some manufacturing plants, it’s used as a solvent or as an intermediate to make other chemicals. That purpose means it stays largely out of public sight, but this doesn’t mean it’s harmless. I’ve done enough reading on chemical hazards in workplaces to know that “rarely seen” can sometimes mean “rarely managed,” which leads to bigger problems.
Breathing And Skin Exposure Risks
Let’s talk symptoms. Breathing in vapors or coming into contact with this ether isn’t like walking through a perfume cloud. People exposed to low levels might complain of sore eyes, irritated noses, or scratchy throats. Only a handful of cases get reported, but the science is clear. According to sources like the National Institute for Occupational Safety and Health (NIOSH), higher levels lead to headaches, nausea, dizziness, numbness in fingers and toes, and even problems with coordination. With enough exposure, people have experienced unconsciousness. The skin doesn’t let it pass, either — it can absorb the chemical and react with redness, itching, or swelling. These problems stack up over time if proper protection isn’t used.
Liver And Kidney Worries
Long-term exposure brings other risks. Chemicals like 1,2-dichloroethyl ether tend to put a strain on the liver and kidneys. Doctors and toxicologists have studied similar ethers and found evidence pointing to organ damage in animals. Ongoing workplace exposure, even at low concentrations, pushes these organs to process and filter out toxins day after day. People working with these substances sometimes share stories of feeling run-down or sick without obvious cause, and blood tests can later reveal their organs carrying a heavier chemical burden than the rest of us.
Cancer And Safety Studies
Several old studies have looked at cancer risks, especially since chemical ethers often pop up in research about workplace cancer clusters. The U.S. Environmental Protection Agency classifies 1,2-dichloroethyl ether as a possible human carcinogen. That label doesn’t pop up unless there’s at least some strong suspicion from laboratory studies. It’s not sold for home use, but even people who work nearby can get caught by accident. Unexpected leaks or poor ventilation have been part of safety reports over the last few decades. Long work hours in these environments make things worse, not better.
Reducing The Dangers
The fix isn’t complicated, but it takes real effort. Good ventilation works, not just a fan in the window, but systems that keep vapor levels below federal safety limits. Gloves, masks, and protective goggles matter, especially since direct skin contact adds to total exposure. Regular air testing in these workplaces helps stop problems before they start. Any employer still using this ether should give training sessions that explain both the risks and best ways to avoid accidents. I’ve spoken with safety experts who say that biggest jumps in worker health come from simple measures used every single day. For folks outside the plant, pushing regulators for more transparency in reporting helps, too.
How should 1,2-Dichloroethyl Ether be stored and handled safely?
Recognizing the Real Risks of 1,2-Dichloroethyl Ether
1,2-Dichloroethyl ether doesn’t hang out in the headlines like some other chemicals, yet this liquid brings plenty of hazards to the table. With its strong ether-like odor and potential to cause everything from eye and lung irritation to larger fire risks, storing and handling it deserves more than a shrug. The folks working in labs or warehouses can’t afford to wing it. The stakes aren’t just regulatory—they’re personal.
This chemical evaporates quickly, so it’s not just skin and eyes on the line; even a minor spill puts the whole workspace at risk. Contact may cause nausea, dizziness, and a sense that you can’t catch your breath. In my early twenties, I watched a colleague get lightheaded simply walking through a poorly ventilated storeroom with open solvent containers. Getting lazy about labeling, storing, or ventilating cost him a trip to the ER and gave the whole team a wake-up call.
Setting Up the Right Storage
Plastic and glass resist corrosion, so those containers stand out as best picks for this solvent. Forget improvising with unlabeled jugs or makeshift shelves. 1,2-Dichloroethyl ether belongs in a cool, dry room or storage cabinet designed to block out sunlight and heat. I’ve seen a sunlit window turn a locked cabinet into a slow-motion disaster waiting to happen. This chemical forms peroxides over time, setting up a bigger explosion danger than most expect. A routine for checking and dating containers can save everyone from mixing old compounds or missing subtle leaks.
Vapors catch fire with surprising ease. Store 1,2-Dichloroethyl ether away from strong oxidizers and open flames. Keep bonding and grounding practices on everyone’s checklist—one static spark, all bets are off. I still remember a near-miss in a teaching lab where someone skipped the antistatic mat, proving these aren’t optional rules. Make the fire extinguisher part of the scene, not an afterthought covered in dust.
Smart Handling Habits on the Job
Every time someone opens a bottle, there’s enough risk to care about. No room for shortcuts. Goggles, heavy gloves, and lab coats aren’t just to meet regulations—they set a division between an ordinary day at work and a disaster. Use chemical fume hoods every time, since standard ventilation rarely keeps up with vapors.
Spills count as emergencies, big or small, and they need everyone’s focus. From my own experience, the fastest spill response happens in places where teams rehearse the drill. Plenty of safety showers and eye wash stations help, but speed comes from practice, not just equipment. My advice? Teach the habit of reporting even slight exposure or suspicion—early attention limits long-term damage.
Learning About Waste and Cleanup
Neutralizing waste before disposal takes extra effort for 1,2-Dichloroethyl ether. This isn’t a pour-down-the-drain moment, and hazardous waste bins exist for good reasons. Anyone who handles cleanup should know the chemical’s dangers inside out before getting involved. I once saw a rookie grab a mop instead of spill pillows, only to find out how fast toxic vapors take over a room. Clear training, regular checks, and leadership that cares—those are common threads where things work right.
Final Thoughts on Real-World Solutions
Treat this chemical with the respect it demands, and it rarely leads to surprises. Cut corners, and trouble comes quick. Pay attention, rely on teams, check labels and dates, and keep safety gear in easy reach. There’s no need to guess at the risks, and plenty of reason to avoid giving 1,2-Dichloroethyl ether a chance to prove itself the hard way.
What is the chemical structure and formula of 1,2-Dichloroethyl Ether?
Understanding 1,2-Dichloroethyl Ether
Chemistry isn’t just for the textbooks—it shapes the products in our everyday lives. For folks who spend time in a lab or read labels on cleaning agents, the names of chemicals like 1,2-Dichloroethyl ether may pop up. Learning about its structure and formula takes this knowledge out of the abstract and into the real world, where safety and application rely on more than just a long title.
The Core Structure
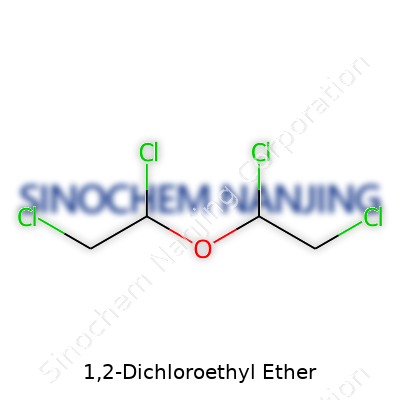
Let’s get right into the molecular basics. The chemical formula for 1,2-Dichloroethyl ether stands as C4H8Cl2O. Looking at those atoms, you get four carbons, eight hydrogens, two chlorines, and an oxygen atom coming together. The “ether” part means there’s an oxygen atom bridging two carbon chains. Here, each of those chains is a two-carbon stretch, and each picks up a chlorine atom on the first carbon, like handles on a jump rope.
If you imagine drawing it on paper, you’d line up the two two-carbon chains (ethylenes). Push one chlorine onto the carbon right next to that connecting oxygen on each side. So, you have ClCH2CH2OCH2CH2Cl. In shorthand, chemists often write it as (ClCH2CH2)2O. These structural details set up the way this compound behaves in practice.
Why Chemical Structure Shapes Reality
A molecule’s blueprint determines its effects in the real world. With 1,2-Dichloroethyl ether, the chlorines don’t just hang around for show. They add weight, change how the substance mixes with water, and shift the risks you might face during handling. These electrons draw reactivity, which affects storage. The presence of ether and chlorinated groups means labs have to keep this chemical far from heat sources, sparks, and any open flames.
In my early university work, I spent hours reviewing Material Safety Data Sheets for solvents. I learned fast that these little details—like the exact spot a chlorine atom sticks to—make a big difference. Some ethers become explosive if handled without care, especially those with halogens. Misreading the structure, or glossing over it, leads straight to lab accidents or ruined batches.
Importance of Responsible Handling
Anyone working with compounds like 1,2-Dichloroethyl ether must think beyond the formula. Its structure poses special risks—think about its volatility or tendency to form harmful byproducts with other chemicals. Ethers in general can develop peroxides over time, becoming dangerous with age. Add chlorine to the mix, and disposal gets tricky, too. Guidelines from groups like the CDC and OSHA emphasize ventilation, diligent personal protective equipment, and strict temperature control. These rules exist because of real chemical behavior, not just policy.
Safer Approaches for the Future
Relying on clear, accurate structural information allows chemists and industry workers to pick the right containment, disposal methods, and emergency protocols. Training new scientists should emphasize structural literacy—learning to break down a formula, picture those bonds, and predict hazards. In my experience, the best classrooms encourage hands-on modeling, not rote memorization. Real practice means fewer surprises in production-scale settings.
Building a culture of safety starts by knowing what’s inside the bottle, atom by atom. With compounds like 1,2-Dichloroethyl ether, clarity on structure won’t just satisfy curiosity. It keeps labs running smoothly and people out of harm’s way.
What precautions should be taken in case of a spill or exposure to 1,2-Dichloroethyl Ether?
Understanding the Real Risks
Working around chemicals carries its own set of risks, and 1,2-Dichloroethyl Ether underlines why respect for safety rules isn’t just about compliance—it’s about protecting lives. This compound isn’t just a name on a label—it’s flammable, and it can be pretty toxic. Breathing in its vapors irritates the lungs and, if there’s enough exposure, things can get worse fast. If it gets on skin or in your eyes, that’s more than a minor problem.
Thinking Beyond Labels: Wear the Right Gear
Lab coats and goggles aren’t just for show. Chemical-resistant gloves, splash goggles, and lab coats actually stand between your skin and a bad day. I remember one mishap with a solvent years ago—I had goggles but wasn’t wearing gloves. A little spill burned my skin for hours. That stuck with me when reading reports about 1,2-Dichloroethyl Ether exposures. The right gloves (like nitrile) hold up, and splash goggles keep your eyes out of harm’s way. If you think regular glasses are enough, trust me, they’re not.
Take Air and Ventilation Seriously
It’s easy to overlook ventilation until someone starts coughing. Vapors from this chemical can hang in the air, especially in tight lab spaces or storerooms. Fume hoods might seem like overkill, but they make a difference. Even a decent fan helps disperse low-level vapors. Air monitoring equipment also gives a heads-up before things get out of hand. OSHA has laid down limits for exposure—those aren’t random numbers, they’re real benchmarks based on health outcomes.
If a Spill Happens—Act Fast and Smart
Spill response kits should never collect dust. Have one at arm’s reach, not locked away in some cabinet. Absorbent materials, like clay or vermiculite, soak up chemicals in seconds. Once, during a night shift, I watched someone try to mop up a spill with paper towels. They got the chemical on their hands and made the spill worse. Dedicated kits and the right absorbent keep you from repeating that mistake. After cleanup, materials should go in labeled hazardous waste containers for disposal—not in a regular trash bin.
Decontamination and What to Watch For
After a spill, contaminated surfaces need soap and water, but not every surface reacts well to water. I’ve found that checking material safety data sheets (MSDS) before reaching for anything saves time and headaches. Any exposed clothing comes off right away and goes into sealed bags for cleaning or disposal. Skin contact? Flush with running water for 15 minutes, no excuses.
Training and Emergency Procedures
No written procedure replaces good training. Regular drills make sure everyone knows their role. You should know where eyewash stations and emergency showers sit. In my former labs, we kept response charts at every entrance, not just for regulations but to cut down on panic. If you feel unwell after exposure—report it. Early symptoms save lives.
Prevention, Not Panic
Chemical safety builds on habit. Review MSDS sheets with your team. Keep kits stocked and check ventilation. Real safety culture means talking about errors, not hiding them. I’ve learned the hard way that simple steps—double-checking labels, clear communication, taking ten seconds for PPE—end up saving much more than just lab time. 1,2-Dichloroethyl Ether isn’t forgiving, and that’s exactly why preparation matters.
| Names | |
| Preferred IUPAC name | 1,2-dichloroethoxyethane |
| Other names |
Bis(1-chloroethyl) ether 1-Chloroethyl ether Ethylene chlorohydrin ether |
| Pronunciation | /ˈwʌnˌtuː daɪˌklɔːroʊˈɛθɪl ˈiːθər/ |
| Identifiers | |
| CAS Number | 111-44-4 |
| Beilstein Reference | 1720239 |
| ChEBI | CHEBI:82252 |
| ChEMBL | CHEMBL43037 |
| ChemSpider | 16424 |
| DrugBank | DB14162 |
| ECHA InfoCard | 100.005.978 |
| EC Number | 1.1.1.1 |
| Gmelin Reference | 8317 |
| KEGG | C19580 |
| MeSH | D003350 |
| PubChem CID | 7904 |
| RTECS number | KK9275000 |
| UNII | G6VB84Y25R |
| UN number | UN1915 |
| CompTox Dashboard (EPA) | urn:cctms:0068e76a-d231-46f5-b2a3-8b3c8d6a1ca3 |
| Properties | |
| Chemical formula | C4H8Cl2O |
| Molar mass | 141.00 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet odor |
| Density | 1.25 g/mL at 25 °C(lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.85 |
| Vapor pressure | 14 mmHg (20°C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | 8.09 |
| Magnetic susceptibility (χ) | -59.0×10⁻⁶ cgs |
| Refractive index (nD) | 1.444 |
| Viscosity | 1.491 mPa·s (20 °C) |
| Dipole moment | 1.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 348.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –233.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -711.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AX02 |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P210, P260, P273, P305+P351+P338, P309+P311 |
| NFPA 704 (fire diamond) | 1-3-0 Health:1 Flammability:3 Instability:0 |
| Flash point | 61 °C |
| Autoignition temperature | 195 °C |
| Explosive limits | 3.3–16% |
| Lethal dose or concentration | LD50 oral (rat): 850 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 460 mg/kg |
| NIOSH | KL3575000 |
| PEL (Permissible) | PEL: 15 ppm |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Bis(chloroethyl) ether Chloroethyl chloroformate 1,2-Dichloroethane Ethylene oxide |

