Reflecting on 1,2-Dibromo-3-Butanone: Science, Risk, and the Road Ahead
Historical Development: From Curiosity to Laboratory Staple
In the world of halogenated ketones, 1,2-dibromo-3-butanone carries a history that traces back to the energetic exploration of organic synthesis in the early and mid-20th century. Often, advancements in synthetic routes paralleled the expansion of organic analytical techniques. 1,2-dibromo-3-butanone didn't just appear out of necessity; it arrived as chemists sought new intermediates for building more complex molecules. Early laboratories recognized this molecule’s value as a reactive precursor, and as detection methods improved, so did interest in its controlled manipulation. What stands out is not just that scientists learned to make and use this compound, but also the way its discovery exemplified the push for more versatile halogenated intermediates. This pattern reveals something bigger: chemistry always reflects the state of knowledge and tools of its time, and every compound with a bromine on its backbone holds countless stories about how research pushes boundaries.
Product Overview: Nature of 1,2-Dibromo-3-Butanone
1,2-dibromo-3-butanone exists as a colorless to pale yellow liquid, distinct in smell and instantly recognizable to those familiar with halogenated organics. Researchers handle it in small vials in modern labs, often examining its sharp aroma and high density. Unlike everyday chemicals seen in home kitchens or workshops, this compound rarely leaves specialized environments. Its value comes from high reactivity, especially thanks to bromine atoms on adjacent carbons—a structure rarely found in simple environmental samples, but common in the chemist’s arsenal for nucleophilic substitution. Techs and researchers handling this molecule often understand, intuitively, that it isn’t just another ketone. It carries both promise and risk, demanding precision and respect in use.
Physical & Chemical Properties in Practical Settings
With a molecular formula of C4H6Br2O, 1,2-dibromo-3-butanone behaves as one might expect from a bromo-ketone: dense, moderately soluble in organic solvents, and reactive with water in some conditions. Its boiling point comes in significantly higher than that of unhalogenated analogs, making it less volatile yet still prone to vaporization with enough heat. Bromine’s presence boosts its molecular weight and gives rise to noticeable reactivity, especially toward nucleophiles. The distinct sharpness in its odor and the way it clings to glassware mark it as a compound that lingers—chemists remember working with it by smell alone. Its density, heavier than water, and tendency to stain surfaces reminds users to handle it with care, since a careless spill won’t just disappear with a little rinse.
Technical Specifications & Labeling Observations
In labs, 1,2-dibromo-3-butanone carries hazard labels with symbols for toxicity and environmental impact. The specifics often arrive in the form of small amber bottles, complete with clear and bold warnings because of its potential to cause harm on contact or through inhalation. Real-world labeling focuses on visible risk symbols, lot numbers, and clear chemical names rather than convoluted codes or regulatory jargon. Researchers—especially undergrads getting their first exposure to halogenated organics—don’t just look at purity but also for batch date or potential decomposition. Improper storage creates headaches, as this molecule reacts with moisture or exposure to light, meaning tight caps and refrigerated cabinets always stay part of its protocol.
Preparation Method: Craft and Caution
Preparation of 1,2-dibromo-3-butanone often involves controlled halogenation of 3-butanone or a similar suitable precursor. Adding bromine to a ketone under buffered, cool, and dry conditions transforms the plain carbon backbone into a reactive platform. Those running this synthesis work in fume hoods and keep sodium thiosulfate handy for neutralizing spills of unreacted bromine. The exothermic nature of halogenation stays etched in the minds of graduate students—one slip on temperature control or errant addition speed, and the risk of uncontrolled outgassing becomes all too real. Every step feels consequential, from reagent addition to careful separation of the product from side reactions. Watching the orange tinge of excess bromine disappear tells experienced hands that the process nears completion.
Chemical Reactions & Modifications: More Than a Building Block
1,2-dibromo-3-butanone opens the door to countless synthetic manipulations. Its adjacent bromines make it particularly responsive to nucleophilic substitution, allowing researchers to swap these groups for amines, alcohols, or thiols. Each new substitution can push a project toward pharmaceuticals, agrochemicals, or even specialty polymers. Real breakthroughs come from creative minds who spot ways to turn vulnerabilities—reactivity, instability in air—into assets for making otherwise tough-to-synthesize molecules. In educational labs, it often serves as a teaching example for carbonyl reactivity and the interplay of electron-withdrawing halogen atoms. The molecule’s willingness to undergo further transformation draws out a certain respect, turning each reaction into a hands-on lesson in organic synthesis—balancing risk, potential, and the joy of discovery.
Synonyms & Product Names: Navigating the Nomenclature Maze
Ask ten chemists and you may hear ten names: 1,2-dibromo-3-butanone, alpha, beta-dibromobutanone, and sometimes even 3-butanone, 1,2-dibromo. Nomenclature varies by region, supplier, or publication, which makes inventory checks or literature searches more challenging than expected. For anyone troubleshooting a reaction or pulling references for a protocol, recognizing all these synonyms means catching details others might miss. This tangle of names also highlights a persistent issue: chemistry still grapples with the legacy of naming conventions that don’t always cross borders cleanly, so researchers stay alert to synonyms in every procurement or literature search.
Safety & Operational Standards in the Real World
1,2-dibromo-3-butanone rarely enters a lab without a checklist of precautions. Gloves, splash goggles, and fume hoods always come out for weighing or transfer. Accidental dermal or eye contact leads to immediate symptoms, demanding swift first aid responses. Unlike some benign lab reagents, this one forces everyone to take chemical hygiene seriously. Spills prompt not just mopping up, but may require evacuation and specialized cleanup routines. Disposal routes always route through hazardous organic waste, never down the drain, to prevent environmental damage. Training sessions feature it as an example when teaching new staff about the downside of chemical innovation: with opportunity comes obligation to manage risk at every turn.
Application Area: Small Niche, Big Impact
Applications for 1,2-dibromo-3-butanone stay squarely in the field of synthesis. Medicinal chemists may use it to decorate molecules with harder-to-install functionalities, while polymer scientists occasionally pursue customized backbones for niche applications. Few industrial users depend on it in high volume, but the compound’s strategic reactivity makes it a gem in academic settings focused on reaction mechanisms or organic methodology development. In my experience, every lab that keeps a bottle tucked away does so because their route to a certain compound would be downright impossible without it. Innovation in small-batch, customized molecules keeps this compound relevant far beyond what one might expect from a rarely seen chemical.
Research & Development: Expanding Understanding and Utility
R&D for 1,2-dibromo-3-butanone often lives at the intersection of ambition and caution. Researchers continue hunting for new reaction pathways, especially those that minimize hazardous waste or improve yields. Projects focus on tuning its reactivity, sometimes swapping bromine for less dangerous moieties post-reaction, to leave a safer residual product behind. Development teams spend time seeking catalysts or conditions to make syntheses more sustainable, or investing in analytical tools for rapid purity checks. The compound’s sharp reactivity profile means even small advances in process control can translate to safer workflows and greener chemistry. Bench chemists know that any reduction in waste or boost in selectivity means not only a safer workplace but also lower impact on the environment—a lesson that seeps into every new project charter involving halogenated intermediates.
Toxicity Research: The Balance of Risk and Reward
Data points to acute toxicity on skin and through inhalation; animal studies highlight damage to tissues and potential for mutagenicity at even low doses. This information gets taken seriously by all operators, steering work toward closed systems and minimum exposure techniques. Toxicologists dig deeper into metabolic byproducts, looking at whether breakdown in natural systems poses risks to aquatic organisms—a real concern given the molecule’s potential to persist in water and soil. For many, the data flows from animal studies to regulatory discussions and practical advice, shaping lab safety culture and influencing whether or how much to order for a given project. The fine print on every MSDS gained from this research isn’t just legal protection—it’s the product of countless hours extracting, analyzing, and validating risk, day after day.
Future Prospects: Charting a Path Forward
Up ahead, the world faces tough decisions on halogenated organics, and 1,2-dibromo-3-butanone finds itself in the crosshairs of greener chemistry trends. While new synthetic methods continually emerge, demand remains tied to ease of halogenation and the unique possibilities this backbone unlocks. Green chemistry advocates push for milder reagents and less hazardous intermediates, and this often means innovations in catalysis or process engineering take center stage. Researchers eye every new substitution method that could replace bromine or cut down on hazardous byproducts. As synthetic goals shift toward environmental responsibility, the old ways of handling such molecules have to evolve too. Chemistry as a field has always grappled with risk, benefit, and the realities of large-scale waste. That’s where real change will, and must, happen—at the bench, not just on the page.
What is the chemical structure of 1,2-Dibromo-3-Butanone?
Looking Closer at the Chemical Puzzle
1,2-Dibromo-3-butanone sounds intimidating at first. You can crack open that name, though, and make sense of what the molecule looks like. I remember the first time I tried drawing out organic structures, the key was to break the words down and figure out the backbone, then fill in the details.
The word “butanone” gives it away. You’re working with a four-carbon chain—a butane skeleton. “One” at the end points straight to a ketone group, which sits on the third carbon, making it a 3-butanone. Add “1,2-dibromo” at the front, and things get more specific: you’ve got bromine atoms attached to carbons one and two. Put it all together, and it looks like this: BrCH2-CHBr-CO-CH3.
None of these structures float around in a vacuum. A molecule like 1,2-dibromo-3-butanone sits at a crossroads of chemistry and practical use. Back in university, my lab partner joked that once you know how to name these things, you can reverse-engineer them from the label alone. This isn’t just a classroom exercise, either. The placement of those bromine atoms transforms the way this compound reacts—more than a simple feature.
Why the Structure Matters for Health and Environment
Chemists and toxicologists have their eyes on brominated compounds because of their behavior in the environment and in living systems. The carbon-bromine bond stands out as far less stable than its chlorine cousin. Many brominated molecules break down faster in sunlight or when exposed to heat. Add the ketone into the chain, and reactivity jumps up. Now you’ve got a substance that could act as a powerful alkylating agent—a molecule that can stick onto proteins or DNA, often with unpredictable results.
Health studies on similar molecules point to several areas of concern. Many brominated byproducts crop up as impurities when disinfecting water with bromide present. Compounds with a ketone right in the backbone can act as intermediates in making pharmaceuticals or pesticides. My background in laboratory safety keeps reminding me that each functional group increases the risk profile—volatile, potentially harmful if inhaled or if it comes into contact with skin. The Environmental Protection Agency keeps close track of chemicals like this for a reason.
The Drive for Safer Handling and Transparency
Many companies aim to stay ahead of regulation by replacing risky brominated chemicals in their processes. Sometimes that means swapping for alternatives improved on both the yield and environmental impact. It often starts with understanding a structure down to the finest detail—seeing which part of the molecule gives it its properties, and which features invite trouble. Open data, detailed reporting, and clear labeling give researchers, workers, and regulators the information they need to step in early.
Substitution, improved worker protections, and more thorough environmental monitoring make a difference. Chemists design molecules with fewer halogen atoms wherever possible. Employers invest in air filters, gloves, and emergency protocols. On the policy side, pushback against “regrettable substitutions”—where one harmful chemical replaces another—reminds us not to lose sight of the broader consequences.
Solutions Have Their Roots in Structure
The arrangement of atoms in 1,2-dibromo-3-butanone packs a big punch. The structure opens the door to countless uses and risks. Clear insight into that pattern—beyond rote memorization—can push science in a more responsible, useful direction. My years with organic chemistry textbooks taught me plenty, but it’s the implications for health, the planet, and real-world use that drive the calls for change.
What are the primary uses of 1,2-Dibromo-3-Butanone?
1,2-Dibromo-3-butanone doesn’t make headlines like plastics or fuels, but it plays a quiet yet crucial role in making sure some things in our daily life work right. Folks in both academia and industry handle this chemical with care, thanks to its unique chemical structure and reactive bromine atoms. These properties give the compound specific jobs, useful in settings that rely on strong chemical reactivity.
A Tool in Laboratory Research
Scientists in organic chemistry value 1,2-dibromo-3-butanone for the way it can tweak molecular structures. Its bromine atoms give chemists a tool for halogenation reactions—a trick that lets them introduce or swap out atoms in complex molecules. Talking with chemists, I’ve seen how valuable these reactions become in drug discovery. The ability to precisely add certain groups onto a molecule opens doors for fine-tuning pharmaceuticals or testing new reaction pathways. Without accessible chemicals like 1,2-dibromo-3-butanone, the road to new medicines or specialty materials gets a lot steeper.
Precursors and Specialty Synthesis
Another key job involves acting as a “building block” for other chemicals. The unique structure, combining bromine and a reactive ketone group, means the compound helps create all sorts of ingredients for more advanced work. In my work with colleagues from the specialty chemicals sector, I’ve seen this material used to develop compounds that clean up pollutants or act as steps toward higher-value products. These intermediates don’t land on store shelves, but they’re the reason advanced products—from water treatment agents to agricultural chemicals—come together the right way.
Pesticide-Related Applications
Some research traces the compound’s roots back to pesticide science. In older literature, you’ll find the molecule mentioned as a useful part of forming certain brominated pesticides or biocides. These applications took advantage of its toxicity and reactivity against living cells and pests. Folks in agriculture and pest control sometimes still rely on legacy chemicals built this way, though regulatory changes in places like the U.S. and Europe have squeezed out several related compounds over health and environmental worries. Responsible handling and clear labeling have become more pressing, especially as people demand safer food and cleaner water.
Handling Hazards and Environmental Impacts
That last point hits close to home for anyone tracking chemical safety. My old mentor drilled into me the importance of understanding both the upsides and the risks. 1,2-Dibromo-3-butanone, with its reactive bromine atoms, can cause serious harm if spilled or inhaled. Its breakdown can lead to toxic byproducts, a concern echoed by environmental monitoring groups. Accidental release or improper disposal risks soil and water contamination. Companies invest in training staff, upgrading facilities, and tightening rules to meet safety checks—but cases keep coming up. The EPA and similar agencies invest in surveillance and encourage green alternatives, but they face challenges keeping up with industry pace.
Looking at Safer Alternatives
One way forward involves designing chemicals that do the same jobs but break down into safer stuff. Green chemistry advocates point to “benign by design” thinking—planning for the chemical’s fate in the environment before production ramps up. Companies have started working with academic partners to test less toxic ketone and bromine substitutes, especially where older compounds have links to persistent pollution or bioaccumulation. If these projects scale up, the next generation of chemicals might shed some baggage tied to health and environmental risk.
1,2-Dibromo-3-butanone might seem like just another formula in a long list, but for anyone in industrial chemistry or synthetic labs, its future will depend on striking the right balance between utility and stewardship. Until newer options show up that get the job done without as many hazards, places that manage this chemical owe a duty both to their workers and to the communities downstream.
What safety precautions should be taken when handling 1,2-Dibromo-3-Butanone?
Understanding the Real Risks
I remember helping out in a chemistry lab during my university days. The responsibility that came with every new chemical on the list never felt small. 1,2-Dibromo-3-Butanone brings its own set of hazards. This is not just another compound you pour down a drain after the experiment finishes. Every bottle sits with a purpose, and this one requires respect. The structure means it's reactive. Bromine atoms aren't known for playing nice with skin and lungs. The faintest spill can burn or irritate exposed skin, and just a little vapor can upset the respiratory system. I learned early on that ignoring the material safety data sheet (MSDS) isn’t just careless; it’s dangerous.
Getting Practical: What Works in Real Life
Gloves should never be an afterthought. Nitrile gloves provide a physical shield, but they wear out. I always check for holes or thinning before use. Poly-coated aprons aren’t just for show. When I once worked with a reactive compound and skipped my apron before lunch, a drop left a mark on my jeans that never came out.
Respiratory hazards sneak up on you. Fans and open windows help a little, but a fume hood changes the game. Breathing in the vapor from this compound even once can leave your lungs feeling raw. I never run a reaction or open a bottle outside of a certified chemical hood – that’s my non-negotiable rule. If that option isn’t available, at minimum, an N95 mask offers a layer of defense, though it can’t match the protection of proper ventilation.
Spills and Storage: Lessons from Experience
Tight-fitting containers make a real difference. I had a teacher who would label every single bottle with the open date, usually with a thick piece of tape and marker. This habit sticks with me. Chemicals degrade. Storing 1,2-Dibromo-3-Butanone away from sunlight and heat sources stops breakdown, which can make the substance more dangerous over time.
Spills are a moment of truth. Keeping a spill kit ready, stocked with absorbent pads and neutralizing agents, settles panic before it starts. Each spill gets cleaned right away, with gloves swapped out if there’s contact. Any contaminated material, from paper towels to clothing, ends up in a clearly marked bag for hazardous disposal. Never skimp on this process — local environmental laws take waste disposal very seriously, and ignoring them puts others at risk.
Training and Communication Make the Difference
Protocols mean little if nobody follows them. I always check in with the newest members of the lab and go over the risks. We run through quick drills so everyone knows where to find eyewash stations, fire blankets, and emergency exits. Fixing a mistake is harder than preventing it in the first place. Consistent reminders, written instructions at every workstation, and easy-to-read hazard signs speak louder than any policy manual.
Emergency showers and eyewash stations should work every time. Testing these regularly — even monthly — heads off the kind of surprise nobody wants during an accident. Keeping a direct line open to medical help, whether through a posted phone number or a buddy system, makes sure nobody faces a crisis alone.
Safe Science Builds Trust
Safety isn’t about making the lab look strict or slowing down projects. It’s about making sure everyone gets home at the end of the day. Complacency doesn’t just impact an individual; it’s a problem that spreads. The best labs I’ve seen encourage questions about every single step and treat every chemical, including 1,2-Dibromo-3-Butanone, as something that teaches — not just something that reacts.
How should 1,2-Dibromo-3-Butanone be stored?
Understanding the Substance
Anyone working in a lab or industrial setting knows that some chemicals come with more baggage than others. 1,2-Dibromo-3-butanone falls into this category. This liquid looks harmless enough, but the presence of two bromine atoms makes it reactive and potentially hazardous. If you’ve spent time around halogenated ketones, you know mishandling can bring more problems than anyone wants to deal with—ranging from environmental headaches to health risks.
Why Storage Choices Make a Difference
Chemicals like this take no prisoners when it comes to improper storage. Direct sunlight, fluctuating temperatures, and the wrong kind of container can all turn a manageable reagent into something no one wants spilled in their workspace. Based on years mixing, transferring, and shelving countless bottles with similar profiles, I’ve seen overlooked details spark major issues—including inhalation hazards and shelf fires.
Better Safe Than Sorry: Practical Steps
A cool, dry, well-ventilated spot stands as the bare minimum. Most modern labs use dedicated chemical cabinets made of strong, corrosion-resistant plastic or powder-coated metal to reduce reaction risk between the chemical and its surroundings. Storing the bottle behind a lock adds another layer of protection, not just against accidents but also unauthorized access.
Glass containers with tight-fitting, chemical-resistant caps stand up well to 1,2-Dibromo-3-butanone. Polyethylene or polypropylene might work, but glass eliminates doubts about reactions with plasticizers or slow leaching. Labels matter just as much as the bottle—large, clear, and up to date, including hazard information and usage history if possible.
Personal protective equipment has its place, too. I keep gloves and goggles handy at the point of storage because taking shortcuts after a long day can tempt anyone. Chemical-resistant gloves—nitrile or neoprene—prevent day-ruining skin contact. Quality goggles do more than just fog up; they protect against accidental splashes when lifting heavy or awkward bottles.
Controlling the Air—Not Just the Space
Fumes from 1,2-Dibromo-3-butanone present a hidden risk. Even if the bottle looks fine, slow leaks from ill-fitting caps can fill a cabinet with enough vapors to cause nausea or long-term health effects. Vented storage cabinets, or even a ducted fume hood for bulk containers, cut down on bad surprises. In labs that have skipped ventilation upgrades, I’ve seen more than one person regret working next door to a “just in case” storage area.
Handling Spills Before They Happen
A good plan always tackles emergencies before they show up. Spill kits close to storage, with absorbent pads rated for halogenated organics, save time and cut down panic during a leak. Clear guidance, reviewed in safety meetings, equips staff to act quickly rather than freeze. Calling the safety team before shifting anything reminds everyone how dangerous complacency can get.
Disposal: Closing the Circle
No chemical stays in storage forever. Trusted waste contractors, regular checks, and strict logs ensure 1,2-Dibromo-3-butanone never becomes a forgotten bottle in a back corner—risking both safety and compliance with regulations. From experience, the cost of doing things right pales next to the price of investigations or clean-up after an accident.
What are the common methods of synthesizing 1,2-Dibromo-3-Butanone?
Why It’s a Big Deal in Organic Synthesis
1,2-Dibromo-3-butanone looks simple on paper, but getting this molecule right often challenges chemists who need it for research or industrial use. This compound shows up in flavor chemistry, material science, and drug synthesis. For anyone spending time in a lab, making it efficiently matters for both time and budget.
Go-To Methods That Chemists Actually Use
Bromination dominates here. The most straightforward method starts with 3-buten-2-one, also called methyl vinyl ketone. Most labs choose bromine in the presence of a solvent like dichloromethane. Stirring this setup at low temperature doesn’t just give a bunch of byproducts; it consistently lands you the 1,2-dibromo-3-butanone. This process taps into the high reactivity of the double bond and the carbonyl, so chemists don’t waste hours trying to separate a mess of products.
Some labs go with N-bromosuccinimide (NBS) for bromination, especially if they want to avoid handling elemental bromine. NBS drops bromine at the double bond but does this with less toxic fumes drifting around. It makes sense if you’ve spent time in a cramped lab where even a little bromine spill causes mayhem. With methyl vinyl ketone and a catalytic amount of light or a radical initiator, NBS reliably forms the dibrominated product. Reports show solid yields with minimal side reactions using this approach, and it keeps cleanup straightforward.
Alternative Routes: Not Always Worth the Trouble
Some folks try making 1,2-dibromo-3-butanone by the bromination of 3-butanone, hoping the process will halogenate at both α-positions. That rarely goes clean unless you precisely control conditions, because over-bromination and competing side reactions wreck the yield and complicate purification. If you’ve fought through chromatography to salvage small amounts, you know the frustration.
Keeping Safety and Sustainability in Mind
Green chemistry keeps getting louder in the lab, and bromine’s hazards demand respect. More chemists, especially in teaching settings, prefer using NBS or even trying phase-transfer catalysis with aqueous bromine, which cuts down on direct bromine handling. Dropping the amount of toxic waste should be the goal, not just for the environment but for anyone who’s ever cleaned up after a spill wearing protective gear that’s not quite thick enough.
Thinking About the Real Risks and Solutions
1,2-Dibromo-3-butanone hasn’t just caught attention for its uses. These synthetic setups create brominated waste, and labs can’t ignore disposal. While the methods above work, there’s a strong push for greener bromination agents and even electrochemical routes that don’t create so much hazardous byproduct. One promising idea involves using hydrogen peroxide and hydrobromic acid—releasing bromine in situ—instead of pure bromine. This lessens the safety issues, though these approaches still need refining to reach the yield and selectivity that organic chemists expect.
Reliable routes to 1,2-dibromo-3-butanone exist, but every chemist choosing a method has to balance safety, yield, and environmental cost. At a bench or in an industrial plant, those choices affect more than just the bottom line—they change lab culture, risk, and what the next generation of chemists will see as “normal.”
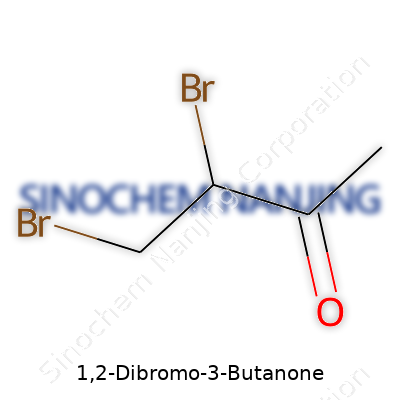
| Names | |
| Preferred IUPAC name | 1,2-dibromobutan-3-one |
| Other names |
1,2-Dibromobutan-3-one 1,2-Dibromo-3-oxobutane Ethyl bromomethyl ketone |
| Pronunciation | /ˈwaɪ,tuː daɪˈbroʊmoʊ θriː bjuːˈteɪnoʊn/ |
| Identifiers | |
| CAS Number | 4314-29-6 |
| Beilstein Reference | 1922388 |
| ChEBI | CHEBI:52220 |
| ChEMBL | CHEMBL222731 |
| ChemSpider | 546403 |
| DrugBank | DB01842 |
| ECHA InfoCard | 03b7de78-776d-4815-9941-9eb2b7b29ea5 |
| EC Number | 214-952-0 |
| Gmelin Reference | 140492 |
| KEGG | C19100 |
| MeSH | D006964 |
| PubChem CID | 2734435 |
| RTECS number | EK9275000 |
| UNII | X85DKT5GQM |
| UN number | UN2664 |
| Properties | |
| Chemical formula | C4H6Br2O |
| Molar mass | 201.90 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | pungent |
| Solubility in water | Slightly soluble |
| log P | 0.8 |
| Vapor pressure | 0.125 mmHg (25°C) |
| Acidity (pKa) | 6.23 |
| Magnetic susceptibility (χ) | -7.38 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.553 |
| Viscosity | 2.314 cP (25°C) |
| Dipole moment | 2.70 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 367.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -146.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2330 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H302, H315, H319, H332 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | Flash point: 91°C |
| Autoignition temperature | 240 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2520 mg/kg (rat, oral) |
| NIOSH | WT1887000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 300 ppm |

