1,2,5,6-Tetrahydropyridine: From Its Roots to Future Impact
Historical Development
Chemistry’s landscape often changes in subtle, unheralded ways. The story of 1,2,5,6-Tetrahydropyridine goes back to the early 20th century, emerging from efforts to explore the partially hydrogenated variants of pyridine. Early synthetic chemists, chasing new methods to functionalize common heterocycles, found that this compound could be coaxed out by careful hydrogenation of pyridine. Researchers at the time wanted to understand more about how saturation altered chemical properties, and tetrahydropyridine arrived as a busy stop along the pathway toward piperidine. Over decades, interest in this molecule flickered, reigniting as pharmaceutical science started probing heterocyclic rings for neurological and pharmacological significance. In the late 70s and early 80s, studies about Parkinson’s disease swung the spotlight back onto this molecule because of its key role in models used to study dopamine neurodegeneration. I remember working in a lab where young chemists looked up old patents and tried out ways to improve on those classic routes, hoping to drive down costs and raise yields for the research supply market.
Product Overview
1,2,5,6-Tetrahydropyridine remains an important chemical in modern laboratories. Its partially hydrogenated structure gives it a blend of reactivity and stability that makes it more versatile than fully aromatic or completely reduced congeners. In commercial contexts, it serves both as an intermediate for drug synthesis and as a building block for specialty ligands and materials. It rarely sits on a shelf for long, usually jumping into action in a synthesis batch, test tube, or reactor. From neuroscience research, where it supports the study of neurotoxicity, to exploratory synthesis of complex molecules, this compound holds enough value that chemical catalogs still list it alongside more common organic precursors.
Physical & Chemical Properties
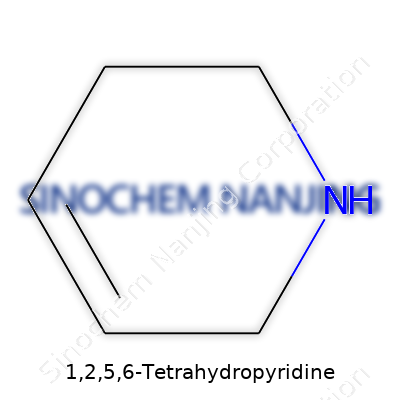
1,2,5,6-Tetrahydropyridine typically appears as a colorless or pale yellow liquid with a distinctive, pungent odor. Its boiling point hovers around 115–120°C, and it has moderate solubility in water but much greater affinity for organic solvents like ether and chloroform. Its structure features a six-membered, nitrogen-containing ring, with four hydrogens added in specific positions, making two double-bonded carbons adjacent to the nitrogen, which imparts both basicity and a readiness to engage in addition or substitution reactions. Thanks to its ring strain and partial saturation, it demonstrates an interesting duality: stable enough for bench handling when protected from air and light, yet reactive enough to participate in reduction, alkylation, and other typical organic transformations. I’ve sometimes struggled with its unpleasant smell during handling, and proper ventilation always stays a top priority in my experience.
Technical Specifications & Labeling
Chemical suppliers label containers of 1,2,5,6-Tetrahydropyridine according to purity, which generally ranges between 95% and 99% for laboratory-grade, and the labeling includes its molecular weight (83.13 g/mol), chemical structure, lot number, and relevant hazard pictograms. Storage instructions clearly state the need for cool, inert atmospheres away from oxidizers and sources of ignition. The Safety Data Sheet (SDS) breaks out important information about flammability, toxicity, and recommended personal protective equipment. In my own ordering and storage process, I always double-check labels for batch dates, since degradation can creep in over time, impacting sensitive synthesis.
Preparation Method
Preparation typically takes place through selective catalytic hydrogenation of pyridine. Commonly, chemists suspend pyridine in a solvent such as ethanol and introduce a catalyst like Raney nickel or palladium on carbon, controlling hydrogen pressure to prevent over-reduction to piperidine. The trick lies in stopping the reaction at the tetrahydro stage, extracting the intermediate before continued hydrogenation. Modern refinements sometimes use high-pressure reactors with automated monitoring for hydrogen uptake, but smaller labs still rely on traditional bench protocols—with a sharp eye on bubbling rates and temperature. After reaction completion, purification involves distillation under reduced pressure, since tetrahydropyridine can degrade at elevated temperatures or upon prolonged air exposure. Getting a good, clean fraction always gives a definite sense of satisfaction after the painstaking carefulness required at each stage.
Chemical Reactions & Modifications
1,2,5,6-Tetrahydropyridine offers an open door to a variety of reactions. Its double bonds, placed within a partially saturated ring, make it an attractive partner for Diels-Alder cycloadditions or Michael-type additions. The nitrogen atom allows for N-alkylation or acylation, letting chemists tweak the molecule for specific activity or solubility. Oxidation can be directed towards pyridine reformation, or further reduction can shift it all the way to piperidine. Its conjugation and electron density make it a flexible synthon for building larger, more complex heterocycles. In medicinal chemistry, attaching side chains or introducing chiral centers often uses tetrahydropyridine as a core, then diversifies from that anchor ring. In my time working with heterocycles, the variety and simplicity of its modifications often impressed me—each new group added creates a new avenue for discovery.
Synonyms & Product Names
Chemists may know this compound by a variety of names, which can confuse sourcing and cataloging. It’s been referred to as 1,2,5,6-Tetrahydropyridine, 1,2,5,6-THP, and Tetrahydropyridine (with the numbers indicating the specific pattern of hydrogen addition). Some references in pharmacology use the abbreviation THP. International chemical suppliers may list it by its systematic IUPAC name or CAS number for clarity, and it sometimes gets grouped under both amines and cyclic olefins in catalog listings.
Safety & Operational Standards
Safety with 1,2,5,6-Tetrahydropyridine goes beyond just wearing gloves. The liquid presents a moderate fire hazard and poses distinct toxicity risks if inhaled, swallowed, or absorbed through the skin. It irritates mucous membranes and can cause headaches, nausea, or more severe neurological symptoms at higher doses. In research environments, good ventilation, fume hoods, and self-contained breathing apparatus in case of spills make up essential measures. Laboratories follow strict protocols for handling, storage, and disposal, including double containment and solvent-resistant gloves. I always kept a spill kit handy, especially since this compound’s volatility can lead to accidental releases during transfer or purification. Chemical fume hoods and eye protection make a real difference here—not just policy but daily habit driven by experience.
Application Area
The application scope of 1,2,5,6-Tetrahydropyridine spans research, industrial, and clinical spheres. In neuroscience, it's famous for anchoring the study of Parkinsonian syndromes—specifically, derived analogs such as MPTP induce symptoms in animal models, clarifying dopamine pathways and neurodegeneration. In chemical synthesis, the molecule provides a scaffold for new drug candidates, catalysis ligands, and agricultural intermediates. For materials chemistry, its reactive ring finds its way into polymers and specialty coatings. Its flexibility appeals especially to pharmaceutical development pipelines hungry for bioactive heterocycles. Over dozens of research projects, I've seen this compound spark sudden pivots in strategy—inspiring both risk and reward across disciplines.
Research & Development
Ongoing research around 1,2,5,6-Tetrahydropyridine continues to surprise. Chemists analyze structure-effect relationships and hunt for safer derivatives or ways to block neurotoxic side effects associated with certain analogs. In medicinal chemistry, modifications aim to produce selective receptor agonists or antagonists, leveraging the molecule’s ability to cross biological membranes and interact with neuronal receptors. Recent work explores asymmetric versions for targeted therapies, using chiral ligands to give improved activity or lower toxicity. Academic collaborations with pharma try out new methodologies, such as microwave or flow chemistry techniques, that speed up the process and economize precious starting materials. Memories of late-night R&D meetings often include this compound’s name scribbled across whiteboards, as teams brainstorm pathways or untangle stubborn side chains from analytic spectra.
Toxicity Research
The toxicology profile of 1,2,5,6-Tetrahydropyridine draws attention, especially after research into MPTP, a derivative famously responsible for rapid-onset Parkinsonism in exposed individuals. Rodent studies repeatedly show neurotoxic effects tied to dopamine neuron degeneration, with analogs serving as vital tools for understanding disease mechanisms. Direct handling can produce acute symptoms, including irritation of eyes and respiratory tract. Long-term or repeated exposure compounds risks, prompting OSHA and REACH frameworks to specify stringent exposure limits for laboratories and production environments. Toxicologists recommend clear labeling, routine air quality checks, and protective procedures to minimize accidental exposure. In my own groups, we always treated this class of compounds with double caution, given past accidents and the molecule’s reputation in the neuroscience community.
Future Prospects
1,2,5,6-Tetrahydropyridine’s outlook ties closely to ongoing medical and chemical innovation. Neuropharmacology continues to use this class for unraveling disease pathways and screening novel therapies, while green chemistry researchers work on more sustainable routes for its synthesis. The quest for less toxic but equally reactive analogs drives inquiry both in the lab and in corporate R&D. Advances in catalyst design, selective hydrogenation, and chiral resolution promise to reduce waste and improve access, paving the way to broader applications and safer handling. I see young scientists using automation, AI-based retrosynthetic planners, and new safety protocols to handle tetrahydropyridine—even as old hands recall its pungent scent and role in foundational breakthroughs. Its journey may be far from over, as every year reveals new uses in unexpected corners of science and industry.
What is 1,2,5,6-Tetrahydropyridine used for?
Understanding 1,2,5,6-Tetrahydropyridine
Folks in chemistry and pharmaceutical labs know 1,2,5,6-Tetrahydropyridine as a valuable building block. This compound features a partially saturated ring, and researchers often turn to it in efforts to develop new medicines or probe the way nerve cells work.
Pharmaceutical Research and Drug Synthesis
1,2,5,6-Tetrahydropyridine isn’t something you’ll stumble upon in your daily routine, but it plays an under-the-radar role in neuroscience research. Scientists use it to study how certain brain disorders develop, especially Parkinson’s disease. In animal studies, one of its byproducts, MPTP, produces damage similar to what’s seen in people with Parkinson’s. This helps researchers better understand the mechanisms behind the disease and gives them ways to test treatments before bringing them to humans.
Beyond Parkinson’s models, this compound pops up as a precursor or intermediate in making medicines that work on the central nervous system. Medicinal chemists appreciate how its chemical structure fits into more complex molecules. With the right changes, it can become a stepping stone toward new drugs that treat mental health problems or movement disorders. I’ve talked to grad students who spend hours tinkering with molecules like 1,2,5,6-Tetrahydropyridine, searching for fresh leads in the hunt for new treatments.
Industrial and Chemical Uses
Chemical companies sometimes employ this compound in syntheses that end up producing fragrances, agrochemicals, or even specialty dyes. Its ring structure lends itself to modifications that let it become part of other useful molecules. While it doesn’t make headlines, its importance comes through in specialized catalogs and research reports. From what I’ve seen in industry publications, suppliers ship small batches of it to labs working on very targeted projects—a far cry from chemicals that fill truckloads.
Health and Safety Factors
Anyone working with 1,2,5,6-Tetrahydropyridine needs to show caution. MPTP, formed from it, is highly toxic to nerve cells. In the 1980s, a tragic story unfolded: a batch of synthetic drugs contaminated with MPTP led to sudden, irreversible symptoms of Parkinson’s disease in young adults. This incident changed how researchers and safety officers view everything connected to this compound. In my old college lab, the supervisor posted warnings and insisted on face shields when even discussing these substances. It wasn’t overkill—as stories like this show, carelessness with chemicals can take a physical toll that no job training prepares you for.
Looking for Safer and Better Alternatives
The dangers linked to 1,2,5,6-Tetrahydropyridine and its byproducts have motivated scientists to look elsewhere. Researchers keep searching for alternatives that capture the scientific value without the potent risks. Some labs now use computer models to mimic its effects, cutting down on lab experiments that generate toxic products. Synthetic chemists also develop routes that avoid dangerous intermediates, showing how safety and innovation can work side by side.
The Bigger Picture
1,2,5,6-Tetrahydropyridine matters because it offers a window into how the brain can break down—and how we might fix it. Immediate applications are limited to specialized spheres, but the stories it brings—of both breakthroughs and hard lessons—matter to anyone who cares about science done right. The work done around this compound nudges the field forward, even as it reminds us why vigilance, knowledge, and responsibility can never be sidelined.
Is 1,2,5,6-Tetrahydropyridine hazardous or toxic?
Unpacking the Risks
1,2,5,6-Tetrahydropyridine does not usually turn up in everyday conversations. Its name sounds like something out of a college chemistry textbook, but this compound matters more than most folks realize. Working in a research lab a decade ago, I remember checking labels twice whenever it appeared on an incoming shipment. It has earned that treatment, and I will explain why.
This compound is a clear liquid used mostly by chemists and pharmaceutical developers. The trouble is, it brings some heavy baggage. 1,2,5,6-Tetrahydropyridine has press around its relationship to Parkinson’s disease. Some research shows it acts as a protoxin. That means the body can turn it into an even nastier substance. Once inside the system, enzymes convert it into MPP+, a well-known neurotoxin that targets brain cells producing dopamine. For anyone not familiar, the loss of these cells is what leads to Parkinson’s symptoms—things like tremors and movement problems.
Almost all the dangerous stories with this chemical come from studies running on animals, especially mice and rats. There are not a lot of spill nightmare tales from manufacturing sites, but strict controls help. Inhaling its vapor or getting it onto skin can cause harm. Lab workers have seen nausea, dizziness, and in severe cases, symptoms that look alarmingly like Parkinson’s. Gloves, fume hoods, and goggles aren’t an afterthought—they’re a lifeline.
Exposure Worries in the Real World
Offices and homes do not usually have to worry. Tetrahydropyridine does not become airborne in normal conditions, and it does not appear in most consumer products. Scientists in controlled labs or at some chemical plants deal with the risks more directly. In those environments, safety checks and proper gear mean everything. Just a few drops on bare skin could eventually turn into a serious health issue, especially if no one acts fast.
Environmental escape is another concern. If a spill hits the ground or drains, it does not just disappear. Toxic metabolites can end up in water sources. I have seen protocols calling for special clean-up teams, which says enough about its potential dangers to the environment.
Can We Do Better?
There are options for making labs and workplaces safer. Substituting this chemical with less hazardous reagents stands out. A scientist may grumble about fiddling with research protocols, but safety outranks convenience. Closing the loop on waste—collecting it properly and sending it to dedicated incineration facilities—cuts environmental risk. Up-to-date safety training for everyone on the floor keeps people sharp about what’s at stake. Sometimes, folks get so used to a compound that they start skipping steps. That’s where accidents happen.
It helps to remember that science only moves forward when people are healthy enough to do the work. There is no prize for cutting corners with a dangerous compound, and 1,2,5,6-tetrahydropyridine demands that respect. Good habits learned from handling tough chemicals can spill over into other parts of life, too—being careful, reading instructions, and watching out for each other.
Staying Ahead of the Dangers
My own experience points to a simple truth: Respect in chemistry labs protects lives. 1,2,5,6-Tetrahydropyridine is not a chemical to fear, but absolutely one to treat with care. Science and industry have every reason to keep looking for safer substitutes and better ways to handle risk. That’s the kind of work that pays off in the long run.
What is the chemical structure of 1,2,5,6-Tetrahydropyridine?
What 1,2,5,6-Tetrahydropyridine Looks Like
Organic chemistry sometimes feels like learning a new language, and 1,2,5,6-Tetrahydropyridine pulls you right into that world. This compound draws attention because of its ring-shaped core, which chemists classify as a partially saturated derivative of pyridine. In plain terms, its skeleton forms a six-membered ring, just like benzene, but it holds one nitrogen atom tucked into the loop with five carbons wrapping around it.
Picturing the structure, the “tetrahydro” part signals that four additional hydrogen atoms have crept in compared to ordinary pyridine. These hydrogens break the double bonds at the 1,2 and 5,6 positions on the ring. That means those two sections act as single bonds now, so the ring's electronic structure changes. The molecule’s formula, C5H9N, tells you it keeps things simple: five carbons, one nitrogen, nine hydrogens. This chemical signature defines how the molecule behaves during reactions and even affects its smell—a musty, slightly fishy odor that people working with amines don’t soon forget.
Why the Shape Matters
Scientists don’t just sketch these structures for fun. The arrangement of atoms decides almost everything about how the compound interacts with living systems or industrial processes. In 1,2,5,6-Tetrahydropyridine, the specific placement of single and double bonds shapes its reactivity. For example, it stands out as a building block in synthesizing compounds linked to neuroscience research. Researchers have used analogs of this molecule to study Parkinson’s disease, since a related structure crops up in the metabolism of certain toxins that damage dopaminergic neurons in the brain. Its resemblance to natural neurotransmitters makes its structure something to look twice at in medicinal chemistry.
On a personal note, my chemistry classes used ball-and-stick models to make these structures real. Arranging the bonds and seeing how the ring flexes taught me that small changes—adding hydrogens or flipping a double bond—can tip the scales from harmless to hazardous. Those hands-on lessons drive home that 1,2,5,6-Tetrahydropyridine isn’t just another chemical formula on a chalkboard.
Making Safe Use of This Compound
The practical side of chemistry always calls for caution. Handling 1,2,5,6-Tetrahydropyridine comes with its own set of precautions, since nitrogen-containing rings often act as irritants or worse. Lab safety gear, good ventilation, and careful storage stand as non-negotiables when working with compounds like this. Real-world stories from research labs point to the risk of inhalation and skin exposure, so those blue gloves and airtight containers matter every day.
Open discussions about risks help more than silence ever did. Teams that share safety information, double-check labeling, and prompt each other to stick to protocols reduce accidents. Using up-to-date material safety data sheets makes a difference, especially as new findings about exposure risks come to light.
Opportunities and Challenges
The structure of 1,2,5,6-Tetrahydropyridine doesn’t just open doors for research, it asks harder questions, too. Chemists look for ways to make and use it without producing extra byproducts or waste, since the nitrogens and reactive bonds can create sticky situations. Green chemistry focuses on producing these compounds with fewer steps, safer reagents, and less energy. Emerging research continues to explore more efficient pathways, including biocatalysts that mimic nature’s careful touch.
Getting familiar with 1,2,5,6-Tetrahydropyridine’s structure isn’t just an academic exercise. It weaves together safety, practicality, and new science—in a way that always circles back to how a change in chemical bonds can ripple through medicine, industry, and everyday life.
How should 1,2,5,6-Tetrahydropyridine be stored?
Understanding the Risks
If you’re dealing with 1,2,5,6-tetrahydropyridine, you’re handling a chemical with some real bite. It smells pretty strong, looks like a clear liquid, and brings some health risks that command respect. Contact with skin or eyes brings immediate discomfort, and the vapors feel just as unfriendly to your lungs. As someone who’s spent years working with chemicals in small labs and big storerooms, I know that the storage approach here makes all the difference between a safe workday and a disaster waiting to happen.
Why Storage Practices Need Attention
The flash point of this compound sits well below room temperature, so even a small heat source can set off a fire. It reacts with oxidizers, so you never want it near bleach, nitrate chemicals, or other fire-loving agents. Even a cracked bottle cap can leak nasty vapors through an entire storeroom. Accidents tend to happen fastest when someone stacks incompatible containers—or when chemical bottles get left out on crowded shelves, away from safety features.
People sometimes overlook the difference that air and humidity make. This chemical absorbs moisture and can degrade if left open, bringing extra hazards. Working in the real world, I’ve seen what happens when someone ignores simple rules: labels peel in the humidity, glass stoppers get stuck, and staff spend more time cleaning up spills than doing real work.
Solutions That Actually Work
Thick glass containers with solid, airtight caps make a huge improvement. Forget plastic jars, because strong solvents often eat right through them, and weak seals let in the moisture that spells trouble. I once trusted a cheap stopper and came back the next morning to a sticky, crusted mess—and a headache to match.
Keep this chemical locked up in a cool, well-ventilated area. I can’t count how many stories I’ve heard about bottles stored close to heaters or sunny windows – every time, it turns into a close call. The best storerooms separate flammable substances from everything else, with clear labels, and their own spill trays. An actual flame-proof cabinet saves a lot more money and grief compared to makeshift shelving.
Ventilation is no small detail. Install a vent system that pulls air away from the storage area and leaves the rest of the building untouched. Simple fans won’t cut it. I’ve watched old storerooms fill with fumes late at night, only for the morning shift to get a nasty surprise. Continuous airflow and frequent checks stop situations like this before they start.
Don’t Skip Regular Checks
Routine inspection keeps things from going downhill. Grab your inventory sheet, open the storage locker, and check for broken seals, sticky residue, and shifted containers. Train everyone—no matter their experience level. Often, the worst chemical spills happen because someone got in a hurry or skipped the safety briefing.
Rely on Experience and Sound Data
The National Fire Protection Association and the American Chemical Society both list 1,2,5,6-tetrahydropyridine as a material with major flammability and health risks. Both recommend storing away from all possible ignition sources and incompatible materials. My own work matches this advice, and mistakes always lead back to ignoring those basics. Stay consistent, use the right equipment, and your storeroom stays safe.
What are the physical properties of 1,2,5,6-Tetrahydropyridine?
The Look and Feel of the Molecule
1,2,5,6-Tetrahydropyridine brings a chemistry lab vibe: it turns up as a colorless to slightly yellowish liquid. The color says a lot about its purity. A perfectly colorless liquid often signals high-grade stock; a yellow tint might mean it’s picked up some impurities along the way. The smell won’t hide in the background. Folks who handle it quickly notice its sharp, ammonia-like odor. This unpleasant scent pops up even at low concentrations, so good ventilation feels less like an option and more like a necessity.
Boiling and Melting: Points of Interest
This molecule doesn’t freeze easily. Its melting point sits at roughly -58°C, so it keeps flowing in below-zero environments. That feature makes it useful in low-temperature processes. On the flip side, its boiling point hovers near 123°C under normal atmospheric pressure. If you’ve ever boiled water on a stove, that number doesn’t sound extreme—so proper equipment remains important. Low to moderate boiling points like this call for well-sealed containers since spills or sudden vapor releases can happen.
Solubility and Polarity
Water and oil rarely mix in life and in labs, but tetrahydropyridine straddles both worlds. It dissolves well in common organic solvents such as ethanol and ether. Its partial solubility in water means it might slip through the cracks during clean-up, making responsible disposal critical. Polarity plays a part here. The presence of nitrogen in the ring bumps up interactions with polar solvents, so it does more than just float on top or sink to the bottom. That N atom encourages hydrogen bonding, setting tetrahydropyridine apart from more stubborn molecules.
Vapor Pressure: Handling with Care
Forget heavy, sluggish liquids. 1,2,5,6-Tetrahydropyridine moves fast, and its vapor pressure clocks in at about 12 mmHg at 25°C. That’s high enough for vapors to fill up a room if given half a chance. My own caution comes from standing too close to open containers of similar compounds—eyes start to sting, and a headache creeps up shortly. This property calls for hood work and airtight storage more than almost anything else on the safety sheet.
Flammability and Risks
There’s a reason chemists treat tetrahydropyridine with respect. Its flash point sits at 25°C. That’s basically room temperature, which means even a warm day could set the stage for a fire risk if vapors meet a spark. Labs that use this molecule take fire suppression and source elimination seriously. One small slip—an unplugged electrical cord or static shock—could lead to a flash fire.
Looking for Solutions in the Lab
Minimizing the risks often means sticking to basics: store it in cool, well-ventilated places and use personal protective equipment. Tight containers keep evaporation in check and cut down inhalation hazards. Water spill protocols don’t always work with organic solvents like tetrahydropyridine. I’ve watched colleagues use sand or inert absorbent materials for spills. Training everyone who handles or stores it lowers the risk much more than any single safety measure.
Why the Details Matter
Each property of 1,2,5,6-tetrahydropyridine writes its own rule in the playbook. From boiling point to vapor pressure, nothing can be overlooked. In the hands of a thoughtful team, its quirks turn into strengths for research and industry. With knowledge, respect grows—and so does safety.
| Names | |
| Preferred IUPAC name | 1,2,5,6-tetrahydropyridine |
| Other names |
1,2,5,6-Tetrahydropyridine 1,2,5,6-Tetrahydropyridine (8CI,9CI) |
| Pronunciation | /ˌwʌnˌtuːˌfaɪvˌsɪksˌtɛtrəˌhaɪdroʊpɪˈrɪdiːn/ |
| Identifiers | |
| CAS Number | 766-43-8 |
| Beilstein Reference | 107174 |
| ChEBI | CHEBI:38948 |
| ChEMBL | CHEMBL194908 |
| ChemSpider | 21508404 |
| DrugBank | DB01684 |
| ECHA InfoCard | 03e21c50-0b18-4221-908f-0ec7e0d37afb |
| EC Number | 206-293-8 |
| Gmelin Reference | 7664 |
| KEGG | C06191 |
| MeSH | D017180 |
| PubChem CID | 15504 |
| RTECS number | UY1050000 |
| UNII | JV6432OI7L |
| UN number | UN3292 |
| CompTox Dashboard (EPA) | DTXSID0052542 |
| Properties | |
| Chemical formula | C5H9N |
| Molar mass | 99.16 g/mol |
| Appearance | Colorless liquid |
| Odor | amine-like |
| Density | 0.926 g/mL at 25 °C(lit.) |
| Solubility in water | Soluble |
| log P | 0.87 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 15.4 |
| Basicity (pKb) | 5.98 |
| Magnetic susceptibility (χ) | -44.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.466 |
| Viscosity | 0.872 cP (25°C) |
| Dipole moment | 1.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 312.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 60.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3683 kJ mol⁻¹ |
| Hazards | |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H370 |
| Precautionary statements | Precautionary statements for 1,2,5,6-Tetrahydropyridine: "P210, P233, P280, P301+P310, P303+P361+P353, P305+P351+P338, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 24 °C (75 °F; 297 K) |
| Autoignition temperature | 340 °C |
| Explosive limits | The explosive limits of 1,2,5,6-Tetrahydropyridine are "1.6-11.7%". |
| Lethal dose or concentration | LD50 (oral, rat): 52 mg/kg |
| LD50 (median dose) | LD50 (median dose): 65 mg/kg (intraperitoneal, mouse) |
| NIOSH | SY7700000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Pyridine Piperidine 2,3,4,5-Tetrahydropyridine 1,4-Dihydropyridine 1,2-Dihydropyridine |

