1,2,4-Trichlorobenzene: Walking Through its Journey and Future
Historical Development
Chemistry doesn’t spend too much time in the spotlight, but every now and then, someone stumbles onto a compound that manages to stand out. 1,2,4-Trichlorobenzene has roots digging deep into the evolution of synthetic organics. The search for better pesticides and intermediates in dye manufacturing drove its first industrial-scale production, tracing back to the early 20th century when the chemical industry rolled out chlorinated benzenes for everything from agriculture to textiles. The decision to keep pushing forward with chlorinated aromatics had a lot to do with growing industrial demand and the need to keep up with innovations in materials and pest control. Stepping into the 1970s and 1980s, regulations tightened, particularly as worries about persistent organic pollutants and occupational safety ballooned. Companies started refining their processes, aiming for higher yields and less pollution, though many challenges remain in keeping byproducts and exposures low. In labs, students and industrial chemists alike still find themselves working with trichlorinated benzenes, revisiting lessons from older scientific discoveries while casting a skeptical eye on the safety legacy left behind.
Product Overview
You see 1,2,4-Trichlorobenzene pop up across chemistry texts and product lists, always marked by its sharp, sweetish odor. Commercial drums and smaller bottles crowd warehouse shelves, mostly destined for roles as herbicide intermediates, solvents, or calibration standards in labs. Each use seems to highlight both the value and the risk baked into halogenated aromatics. While you don’t find it lining hardware store shelves for consumers, industrial sites and research organizations keep it handy for the promise it offers in selective extractions and synthesis pathways. Lab techs and chemists learn to respect both its practical strengths and the responsibility of handling it safely, a tension familiar to anyone who’s ever juggled utility against risk in chemical work.
Physical and Chemical Properties
Measuring out 1,2,4-Trichlorobenzene, you meet a white crystalline solid with a melting point that sits above the room temperature in most climates. Its volatility lets it slip into the air, especially when heated. That means anyone working near open containers quickly understands the need for ventilation. The density gives it weight in solution, so it doesn’t float in water or disappear in the blink of an eye. Solvents like ethanol and ether welcome it easily, while water shrugs it off—classic behavior for a chlorinated benzene. Stability serves as both friend and enemy: it holds together well for storage or analytical use, yet that same persistence means it doesn’t rust away in the environment, making cleanup and exposure more challenging.
Technical Specifications and Labeling
Every drum and flask arrives stamped with hazard markers common to chlorinated aromatics. Labels identify it as a flammable solid, worthy of respect in any warehouse. Lab chemists keep the Material Safety Data Sheet close, pointing out exposure risks for anyone handling powder or vapors. Transport regulations update every few years, so logistics teams check packing guides carefully. Routine sampling usually includes melting point and purity assays, especially when high-spec chromatography or synthesis depends on chemical consistency. Customers in different regions often face additional labeling layers as local authorities tighten standards or introduce new environmental watch lists.
Preparation Method
Over the decades, the typical route hasn’t changed much: direct chlorination of benzene or monochlorobenzene, controlled to nudge the substitution pattern into yielding the 1,2,4-isomer. Tinkering with catalyst choices and temperature can swing byproduct profiles, something process chemists learn quickly as they wrestle with yields and unwanted dichlorinated or tetrachlorinated compounds. In the hunt for efficiency and fewer emissions, some facilities have tried closed-loop systems or integrated purification streams, though balancing pressure, temperature, and chlorination conditions still leaves plenty of room for improvement. Waste minimization remains an ongoing headache, given the stubborn and sometimes hazardous byproducts that come with large-scale chlorination.
Chemical Reactions and Modifications
The story of 1,2,4-Trichlorobenzene in the lab doesn’t end at synthesis. It becomes a building block for nucleophilic aromatic substitution reactions, making it handy for crafting complex molecules in agrochemicals and dyes. Its chlorine atoms invite different nucleophiles, depending on which positions need to hold up under chemical attack. In the right conditions, those chlorines can be swapped for other functional groups—amines, nitro groups, or even more exotic tags—offering a platform for medicinal chemists and materials researchers. Heating or burning it in open air spells trouble, kicking out some nasty byproducts, but under controlled circumstances, you can direct its reactivity toward useful targets. The stubborn structure hinders easy degradation, placing pressure on anyone who might generate waste to plan disposal routes with care.
Synonyms and Product Names
Catalogs bristle with alternate names—“1,2,4-TCB” or “Sym-TCB” show up alongside the more formal title. Overseas suppliers put their own spin on product branding, but in the end, the structure stays the same: three chlorines perched around the benzene core in a 1,2,4-pattern. Researchers navigating older journals or import papers often find the names swirling together, making vigilance necessary to avoid mix-ups with similar chemicals. The world of halogenated aromatics isn’t short on overlaps, and keeping synonyms straight guards against both scientific errors and safety problems.
Safety and Operational Standards
Nobody gets far handling chlorinated solvents and intermediates without a clear safety culture. Vapors sting the nose, and environmental headlines about groundwater contamination put everyone on edge about poor containment or accident risks. OSHA and similar authorities require proper storage, spill control kits, and tight restrictions on direct skin contact. Respiratory and dermal exposures call for gloves, goggles, and in confined rooms, fume hoods or exhaust fans. Once this compound hits waste bins, firms have little choice but to send it down hazardous disposal paths rather than ordinary drains or landfill routes. Training programs pound home safe practices, driven home by case reports from sites where shortcuts led to illness or contamination.
Application Area
Industry leans on 1,2,4-Trichlorobenzene for specialized solvent needs and as a key stepping-stone in herbicide and dye manufacture. These uses tap into the chemical’s ability to solvate tough-to-dissolve resins or push substitutions forward in complex syntheses. Some analytical chemists rely on it as a reference compound for calibrating heat transfer or thermal analysis equipment. Yet, its broader use shrank in the face of stricter regulations, both because of environmental toxicity and pressure to reel in chlorinated compound emissions. Where it does remain, it often shows up in sectors investing heavily in closed processes and rigorous environmental controls.
Research and Development
Research groups investigate fresh ways to repurpose or detoxify 1,2,4-Trichlorobenzene, knowing that legacy waste and strict new targets shape the direction of modern chemistry. On one side, some researchers want greener chlorination methods, using benign catalysts that stress less over accidental pollution or high-temperature requirements. Others focus on developing more robust filtration and carbon absorption systems capable of scrubbing emissions and spilled waste from manufacturing and lab settings. Analytical chemists pick apart degradation products in soil and water, hoping to understand the compound’s journey and how to arrest its spread, while policymakers weigh up data on persistence and toxicity, looking for evidence to support lower emission ceilings and stronger occupational exposure controls.
Toxicity Research
Concerns about long-term exposure won’t fade anytime soon. Toxicology teams highlight how 1,2,4-Trichlorobenzene can upset respiratory and liver systems in animal studies, sparking calls for stricter workplace exposure limits. The EPA and similar agencies keep an eye on its metabolites, many of which can linger and magnify harm as they pass up food chains or find a way into groundwater. Human case studies remain relatively rare, but occupational data underline the importance of avoiding chronic vapor inhalation and direct skin contact during handling. Calls for regular air monitoring, industrial hygiene reviews, and enhanced PPE resonate through every article and training session tied to chlorinated aromatics.
Future Prospects
Industry and researchers feel the push towards fewer hazardous intermediates and safer alternatives. As green chemistry gathers steam, demand for new processes threatens the status of traditional chlorination-based products. Regulators and public interest groups make it hard to ignore the cleanup and hazard reduction priorities, especially for persistent organic pollutants. Some innovations in catalysis or process containment promise real reductions in emissions, giving trichlorobenzenes a shot at cleaner production for those applications where substitutes don’t cut it. At the same time, chemical engineers and waste management teams face growing pressure to prevent leaching and reduce stockpiles of this compound. The vision for tomorrow favors less risky materials and smarter safeguards, forcing everyone from plant managers to regulatory authors to reimagine the role of halogenated aromatics in an era demanding responsibility.
What is 1,2,4-Trichlorobenzene used for?
Taking a Closer Look at Industrial Chemistry
1,2,4-Trichlorobenzene shows up a lot more often than most people expect, tucked away in factories and labs, rather than in consumer products. This chemical shows its worth mostly in the world of manufacturing, specialty chemistry, and research. Most 1,2,4-trichlorobenzene gets produced on purpose, not as a random byproduct, which tells you manufacturers find good reasons to keep it on hand.
Solvent Power in Plastics and Dye Production
Factories working with plastics often lean on 1,2,4-trichlorobenzene to dissolve stubborn polymers, like those in polyesters and PVC. Some resins need heat and a powerful solvent to get them going—regular solvents can’t always do the job. Take polyester testing, for example. This compound provides an effective medium for the analysis of intrinsic viscosity in polyethylene terephthalate (PET), which is essential for controlling the quality of everything from PET bottles to synthetic clothing fibers.
Dye chemists pick this compound for its ability to break down and carry other reactive chemicals in the production of complex dye formulations. The textile industry, where synthetic dyes rule, still relies on methods developed in the twentieth century. A solvent like 1,2,4-trichlorobenzene lets dye and pigment particles mix smoothly during manufacturing, which leads to brighter and longer-lasting colors.
Role in Pesticide and Herbicide Formulation
Modern agriculture leans hard on chemistry to boost yields and protect crops. 1,2,4-trichlorobenzene acts as a starting material for creating herbicides and pesticides. Its chlorine atoms set up good positions for building more complex chemical shapes. Several well-known herbicides trace their roots to trichlorobenzene chemistry. By allowing precise tweaking at the atomic level, chemists can build molecules that control weeds or pests while trying to limit toxicity to crops or people. Agriculture companies monitor these steps to meet international safety and residue standards, but the underlying chemistry often starts here.
Research Settings and Specialty Uses
Academic and industrial labs favor this compound for its stability and predictable boiling point. Chemists who run experiments that need high heat without breaking down their solvent turn to 1,2,4-trichlorobenzene. Its chemical resilience lets researchers work in tough conditions, especially during synthesis of specialty chemicals or in controlled high-temperature studies. The compound has earned a key spot in the calibration of instruments or in thermal testing, thanks to that reliability.
Potential Issues and Safer Practices
Handling 1,2,4-trichlorobenzene comes with real responsibility. Exposure to the chemical, even through its vapor, can harm people’s health—causing skin and eye irritation, potentially damaging liver or kidney function with repeated exposure. I’ve seen workplaces improve safety records by investing in good ventilation, enforced glove use, and monitored storage areas. Factories must meet legal requirements set by agencies like the EPA or OSHA, which involve air monitoring and stringent handling protocols.
Cleaner alternatives sometimes exist, but swaps aren’t always straightforward. Some solvents don’t provide the same dissolving power. Ongoing research focuses on safer, less persistent chemicals, but regulatory changes, worker advocacy, and industry leadership drive real progress. Companies with strong training, clear spill procedures, and up-to-date chemical management keep risks lower.
Understanding Importance
People outside of science and industry might never hear about 1,2,4-trichlorobenzene, yet the plastic bottles, clothes, and safe food supplies in daily life all link back to what happens in labs and factories. Keeping focus on responsible use—backed by strong science, solid rules, and plain common sense—makes sure this compound supports progress without sacrificing health or the environment.
Is 1,2,4-Trichlorobenzene hazardous to health?
The Risks Under the Microscope
1,2,4-Trichlorobenzene doesn’t land in the top ten of everyday conversations, but this chemical pops up in certain workplaces and can drift into the environment from manufacturing operations. Not everyone has heard of it, but folks who have worked around heavy industry or chemical plants likely recognize it. Some common uses include solvents, dyes, and pesticides, so it’s no stranger in the world of industrial chemistry.
What Happens in Real Life Exposure
I spent a bit of time near an old chemical facility. Neighbors often shared stories about odd-smelling air and headaches that came on strong during the workweek. Health concerns hung in the air as clearly as those lingering odors. When a chemical like 1,2,4-trichlorobenzene leaks or lingers where it shouldn’t, most people notice physical symptoms. Eye and throat irritation, dizziness, and nausea can hit hard after breathing in fumes. Those with asthma or chronic lung issues sometimes end up at the clinic more often than usual.
Animal studies draw a stronger connection to danger. Scientists report liver and kidney problems, changes in blood chemistry, and reduced immunity in subjects exposed over extended periods. The Environmental Protection Agency classifies it as a possible human carcinogen, pointing to evidence that it causes tumors in lab animals. Studies linking trichlorobenzenes to cancer in people are less clear, partly because most people only face exposure in short bursts or at very low concentrations. Still, the warning lights are flashing.
Why This Matters—More Than Numbers
Issues pop up when people live near factories or dump sites. I’ve seen property values dip when chemical names like trichlorobenzene turn up in water or soil tests. It doesn’t take a large spill—decades of low-level leaks can leave a mark. Parents talk about rashes or breathing issues in kids who spend every summer running through the same backyards. Real experiences give a sharper edge to the data the experts gather.
Not every chemical needs to scare us, but letting our guard down when experts flag danger doesn’t help. Around the world, regulations only work as well as the folks who enforce them. Sometimes, plants let minor leaks slide to save money or time. Community members grow frustrated answering to higher sickness rates or surprise water bills tied to clean-up costs.
Taking Steps to Cut the Risks
Tighter rules on emissions, regular soil and groundwater checks, and modern safety gear can shift the balance. Public data reporting gives neighbors a fighting chance to spot problems early. I’ve seen grassroots groups push for cleaner practices after news stories brought hidden chemicals to light. People grow more cautious, calling in city inspectors or pressing for fence-line monitoring. The more neighbors learn, the harder industry leaders find it to cut corners.
Safe handling begins in workplaces. Training workers on chemical dangers and supplying proper masks, gloves, or ventilation can prevent hospital trips. At home, fixing leaks and safely disposing of old paint and solvents protects families from unnecessary exposure. Living with chemicals sometimes comes with modern life, but people shouldn’t settle for risks that regulators and scientists already warn about.
Every city or town deserves to know what flows in the air and water nearby. Transparency, regular health surveys, and tough enforcement often bring more trust to communities that have long carried the weight of chemical exposure.
What is the chemical formula of 1,2,4-Trichlorobenzene?
The Formula and Its Roots
1,2,4-Trichlorobenzene lands you with a formula: C6H3Cl3. Benzene, the backbone here, sports a six-carbon ring. Swapping three hydrogens for chlorine atoms, you get this particular configuration. The numbers—1,2,4—just tell you where those chlorines sit around the ring. It sounds straightforward, but placing those chlorines rewires the molecule in ways that matter to industry, workers, and the environment.
Real Industrial Uses
Factories use 1,2,4-Trichlorobenzene as a solvent, especially for cleaning and degreasing. You’ll run into its name in dye production, electrical insulation, and even as an intermediate for pesticides and herbicides. I’ve talked to engineers who remember its strong, characteristic smell in old workshops where it saw heavy use. These workers often mention the product stripping their skin’s oils or lingering in the air, pushing companies to rethink old habits. Past decades watched this compound come off the shelves in consumer settings, sticking mostly to industry now.
Safety and Environmental Impact
Data tells us that 1,2,4-Trichlorobenzene does not belong in the “innocuous” pile. Inhaling vapors or direct skin contact can irritate or even damage tissues. Prolonged exposure can trigger headaches, dizziness, or worse. Back in my university days, safety videos drove home why gloves, goggles, and proper fume hoods count for every handling. The chemical’s persistence shows up in groundwater studies too—spills or leaks stick around, disrupting organisms in soil and water.
Long-term, these persistent substances raise flags among environmental protection agencies worldwide. The US EPA and the European Chemicals Agency have both flagged trichlorobenzenes for extra scrutiny. Their slow breakdown rate means even small releases begin to stack up in ecosystems. Evidence from monitoring programs has pushed manufacturers and governments toward stricter regulations and better containment processes.
Finding a Smarter Path Forward
Chemistry students first learning about trichlorobenzene formulas often ask why we keep producing substances with a clear record of risks. The truth: no easy substitute for its specific solvency profile, especially in niche manufacturing. Still, changes look practical at ground level. Companies now use safer blends for cleaning when possible, and engineers experiment with alternative reagents or less toxic intermediates. My time in research labs taught me that even small tweaks to process—such as sealed systems and better recycling—pay off both in worker health and waste reduction.
Teaching chemical handling now, I point out how changes in law, technology, and culture shape what ends up on benches and in pipes. Bringing everyone together—frontline workers, policy makers, chemists—makes it more likely that smart practices spread. Even with a simple-seeming compound like C6H3Cl3, the conversation around its use reminds us: chemical formulas unlock real-world choices, for better or worse.
How should 1,2,4-Trichlorobenzene be stored?
Why Chemical Storage Matters
People working in labs or warehouses know chemicals aren’t just boxes on a shelf. They demand caution and respect, not just because of regulations, but to protect lives. 1,2,4-Trichlorobenzene isn’t some household cleaner or garden weedkiller. It's often part of chemical research, industrial applications, and even pesticide manufacture. Mishandling brings real health risks—skin irritation, serious fumes, and long-term exposure problems. If a container fails or vapors spread, everyone in the building and neighborhood feels the consequences.
The Right Conditions for Safe Storage
Too many skip reading the label or the safety data sheet, believing experience outweighs paperwork. That’s a shortcut nobody needs. This compound stays liquid at room temperature, but it doesn’t mean just any shelf will do. The right approach starts with a cool, well-ventilated storage room. Heat might not send it up in flames at the drop of a hat, but a higher temperature means more fumes, and these are both toxic and hard to detect. Exposure to sunlight or direct heat just raises the odds of trouble. A shaded, ventilated corner—away from other chemicals, especially oxidizers—prevents unwanted reactions.
Container Choices Aren’t Optional
Metal drums sometimes corrode when holding chlorinated organics. From personal experience, nothing beats thick-walled glass or specialty plastic containers designed for strong chemicals—polyethylene or fluorinated polymer bottles outlast almost anything in a lab. Always shut the lid tightly, label the vessel with the full chemical name and hazard information, and avoid worn or makeshift closures. I’ve seen too many near-misses from rubber stoppers eaten away by leaking fumes or from forgetting to thread a screw cap securely. A single fumble results in costly cleanups. Double-check, always.
Keep Distance from Incompatibles
This isn’t a compound for grab-and-go use. Keep it away from strong acids, alkalis, or oxidizers. Mixing with these invites explosions or hazardous vapors. Arrange chemical shelves with clear sections, and don’t save time by stacking dissimilar bottles side by side. Simple dividers or distance between incompatible groups stop small spills from turning into major incidents.
Ventilation Means Health
Airflow saves. Whether you use a mechanical fan or just let fresh air through vents, don’t close off storerooms in the name of cleanliness or security. In my old lab, a simple exhaust fan made the difference. The smell, sometimes sweet but always warning of toxicity, clears quickly. Workers breathe easier. If anyone feels dizzy, coughs, or has itchy eyes, don’t brush it off—address the airflow and check seals instantly. No chemical is worth risking a colleague’s well-being.
An Emergency, Not a Knock
Every facility storing 1,2,4-Trichlorobenzene should show easy-to-read emergency instructions and keep protective gear—gloves made for corrosives, face shields, chemical-resistant coats—close at hand. Spill kits, not just a roll of paper towels, prepare for leaks. Training newcomers regularly avoids complacency. Real-world drills, not just memos, matter most. Safety grows from habit, not rules alone.
Better Practices Get Better Results
Treating storage as more than a checklist, relying on strong containers, steady ventilation, careful separation, and ready emergency plans, keeps people and business safe. Each detail supports the next. Good storage builds trust among everyone sharing the workspace—and earns the confidence of the community outside those walls.
What precautions should be taken when handling 1,2,4-Trichlorobenzene?
Understanding the Chemical’s Reach
Anyone who’s ever worked in a lab or plant gets quickly familiar with the warnings printed on chemical drums. 1,2,4-Trichlorobenzene, with its sharp chemical smell and oily feel, falls in the group of hazardous solvents that demand real respect. Once opened, its vapor starts filling the room. Anyone breathing that air for a couple hours will start to feel the effects—headaches, dizziness, sometimes an irritation that doesn’t quit. The potential to cause long-term harm like liver or kidney issues adds another layer of concern.
Personal Protection and Awareness
Gloves and goggles seem basic, but skipping them can turn into a ticket to the emergency room. Thick nitrile or neoprene gloves block the substance from soaking through to your skin—a trick I learned after watching a colleague lose a work week over a careless splash. Eye protection matters just as much. All it takes is a careless hand gesture or a knocked-over beaker for the chemical to hit sensitive tissue.
A respirator steps into play if the area doesn’t vent out fumes fast enough. 1,2,4-Trichlorobenzene quickly reaches concentrations above recommended limits in small or closed rooms. Air-purifying respirators with organic vapor cartridges cut the risk sharply. Nobody wants to wear a mask all day, but losing clear thinking mid-shift puts everyone on site at risk.
Handling and Storage Practices
Spill control makes a difference between a minor scare and a full-on emergency. Proper containers with secure seals and tight-fitting lids stop leaks before they start. Storing this chemical away from any open flames or sparks drops the risk of a sudden fire—no one forgets how combustible vapors can change a calm day in an instant. Posting clear signage means visitors and new hires can spot the danger, whether or not they know the smell.
Ventilation keeps fumes at manageable levels. Negative pressure hoods and well-placed fans do the heavy lifting. I’ve seen old warehouses rely on open doors and windows, but those measures feel risky during windy days when vapors could blow right back inside. The best lab managers run their exhaust systems at all times.
Waste Disposal and Clean-Up
Tossing leftover solvent down the drain or into regular trash sounds simple, yet those shortcuts spread the hazard from workplace to water supply. Hazardous waste containers, labeled and dated, keep the clean-up team safe and give disposal companies a fighting chance. In one plant, storing spent solvent in metal drums instead of old paint cans made all the difference after a spill knocked over a row of containers.
Absorbent pads, sand, and special neutralizers deal with spills fast. A designated emergency kit, stocked and checked regularly, spares workers the scramble for paper towels or scrap cloth. A documented response plan makes confusion less likely when nerves run high.
Ongoing Training and Improvement
Education can fall behind in busy shops. Refresher drills and updated signs matter as staff rotate or new interns join in. Sharing real stories—both mistakes and close calls—helps everyone remember these chemicals don’t forgive shortcuts. Regulatory inspections from OSHA or the EPA function as another layer of accountability. Recognizing that rules serve to protect health, not just slow us down, sets a safer tone for all.
1,2,4-Trichlorobenzene won’t cause trouble if people show it care and keep their habits sharp. In the end, personal responsibility and practical steps save jobs, protect families, and keep the community out of harm’s way.
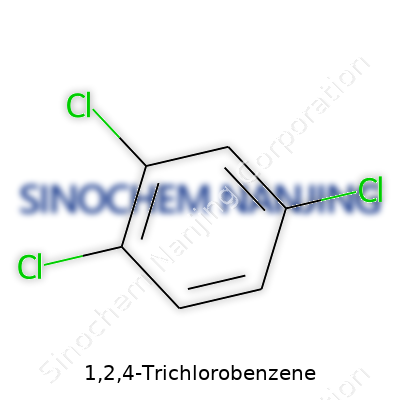
| Names | |
| Preferred IUPAC name | 1,2,4-Trichlorobenzene |
| Other names |
1,2,4-Trichlorobenzol Unsym-Trichlorobenzene |
| Pronunciation | /waɪ.tuː.fɔːr.traɪˌklɔːr.əˈbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 120-82-1 |
| Beilstein Reference | 1209222 |
| ChEBI | CHEBI:35710 |
| ChEMBL | CHEMBL22339 |
| ChemSpider | 7753 |
| DrugBank | DB14096 |
| ECHA InfoCard | 03d3d6a8-7ab5-474d-9262-e4fdfcaa5f15 |
| EC Number | 203-400-5 |
| Gmelin Reference | Gmelin Reference: 81847 |
| KEGG | C01744 |
| MeSH | D014287 |
| PubChem CID | 6950 |
| RTECS number | DC3325000 |
| UNII | QRI3C4XP19 |
| UN number | UN2321 |
| CompTox Dashboard (EPA) | DTXSID4020223 |
| Properties | |
| Chemical formula | C6H3Cl3 |
| Molar mass | 181.45 g/mol |
| Appearance | Colorless crystals or liquid |
| Odor | Aromatic odor |
| Density | 1.450 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.8 |
| Vapor pressure | 0.397 mmHg (25°C) |
| Acidity (pKa) | pKa = 30 |
| Basicity (pKb) | 5.19 |
| Magnetic susceptibility (χ) | '-83.0e-6 cgs' |
| Refractive index (nD) | 1.552 |
| Viscosity | 0.813 cP (20°C) |
| Dipole moment | 2.5 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 150.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -58.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3621 kJ/mol |
| Pharmacology | |
| ATC code | D08AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS05,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335, H411 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P362+P364, P391, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 113°C |
| Autoignition temperature | 648 °C (1,194 °F) |
| Explosive limits | 0.7–5.0% |
| Lethal dose or concentration | LD50 oral rat 756 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 756 mg/kg |
| NIOSH | YU8225000 |
| PEL (Permissible) | 5 ppm (40 mg/m³) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
1,2,3-Trichlorobenzene 1,3,5-Trichlorobenzene Chlorobenzene Dichlorobenzene Tetrachlorobenzene |

