1,2,4,5-Tetramethylbenzene: A Ground-Level Look at an Unassuming Chemical
Historical Development
Chemists back in the nineteenth century stumbled on many aromatic hydrocarbons during their hungry search for new dyes, fuels, and industrial heavy-lifters. 1,2,4,5-Tetramethylbenzene emerged out of this bustling era when researchers began breaking down coal tar, poking around its labyrinth of compounds. The molecule's four methyl groups clinging to a single benzene ring invited closer scrutiny, since folks noticed it popped up as a byproduct when processing coal tar and other aromatics, and its regular structure gave it some curious technical properties compared to its o-xylene or mesitylene cousins. Over the past several decades, manufacturers shifted focus to petrochemical sources, rooting production in modern refineries and not in the messy coal operations of old. Innovation in separation technology and chemical catalysis has led to far cleaner, more reliable methods to gather this compound, which pushed it out of a chemical curiosity and into daily use in labs and factories.
Product Overview
1,2,4,5-Tetramethylbenzene—a symmetrical, stable-looking hydrocarbon—shows up as a colorless crystal or, if the day’s warm, a viscous liquid. Looking at it, you'd never guess how many containers, solvents, and specialty projects depend on it. It stands as a raw material in organic synthesis, often overlooked but always needed, acting both as a solvent to coax along chemical reactions and as a building block for the high-performance compounds that drive the rubber, plastics, and electronic industries. Formulators in the laboratory favor tetramethylbenzene for its reliable behavior and modest, manageable hazards. It handles both heat and chemical stress better than lighter benzene derivatives, making it a preferred ingredient in reactions that get warm or need strong chemical punches.
Physical & Chemical Properties
Solid at lower temperatures and melting just above 78°C, 1,2,4,5-Tetramethylbenzene forms fluffy white crystals that don't catch the eye. It gives off a faint aromatic smell, something between oil and a dry cleaning shop. The molecule packs four methyl groups around a six-carbon ring. That extra bulk pushes its boiling point up to 199°C and hands it a molecular weight of 134.22 g/mol. Tetramethylbenzene shakes off water but dissolves well in ether, benzene, and many of the stronger organic solvents. Its structure gives it a low dipole moment and a habit of resisting oxidation—that confers stability in the bottle and reliability under fire in the lab. Chemists appreciate its ability to behave the same way, batch after batch.
Technical Specifications & Labeling
Any manufacturer ready to ship tetramethylbenzene must pin down its purity and physical constants. Specifications usually demand assay levels above 98%, measured using gas chromatography. Impurities—especially related isomers like pseudocumene—can't rise above a few tenths of a percent. Sensitive equipment, especially flask reactors or chromatographs, show just how much out-of-spec tetramethylbenzene can foul a process. Labels always list the Chemical Abstract Service (CAS) number, 95-93-2, and keep hazard information front and center. Containers, whatever their size, carry information about flammability, melting point, storage recommendations, and the key emergency instructions dictated by local safety codes.
Preparation Method
Modern pathways to 1,2,4,5-tetramethylbenzene start with toluene or other methyl-substituted benzenes. Using Friedel-Crafts alkylation, chemists bolt extra methyl groups onto a benzene ring, controlling temperature and acid catalysts to keep all four methyls lining up on alternating carbons. AlCl3 and methyl chloride still rule the process. To keep side products to a minimum, operations depend on carefully tuned feeds and separation columns stacked in refinery units. Sometimes, by shifting conditions, operators pick up tetramethylbenzene straight out of the xylene isomerization streams found in large petrochemical outfits. Each supplier tries to edge out a purer, less expensive stream, refining both process and output with each production run.
Chemical Reactions & Modifications
This molecule doesn't just sit on the shelf. Tetramethylbenzene holds its methyls with pride but can shuffle hydrogen atoms when faced with strong oxidizers, cough up new derivatives under nitration, and hand off its methyls to form phosphonium or ammonium salts that serve as intermediates for dyes and drugs. The methyl groups can jump into halogenation reactions, giving tetramethyl chlorides and bromides, or fuel ring-opening steps in more robust, high-energy processes. Chemists often modify it to get the right functional groups for specialty polymers, OLED compounds, or performance-enhancing resins. Each tweak changes melting points, solubility, or electrical properties.
Synonyms & Product Names
You won’t hear one name everywhere. In catalogs and safety sheets, you might spot durene, sym-tetramethylbenzene, or 1,2,4,5-tetramethylbenzol. German and Japanese reference books often favor tetramethylbenzol, while English-speaking manufacturers stick with durene and 1,2,4,5-tetramethylbenzene. No matter the label, that same molecular arrangement sits inside each container—a reality buyers learn to navigate, especially when importing material from overseas suppliers using different naming conventions.
Safety & Operational Standards
Nobody who handles chemicals every day takes shortcuts with aromatic hydrocarbons. Durene is no acutely toxic substance, but its fine dust can spark fires if carelessly handled. Storage rules call for cool, dry, ventilated spaces, far from ignition sources. Workers wear gloves and safety glasses, especially during weighing, pouring, or when using hot plates. MSDS sheets underline the need for good ventilation since even low vapor exposure causes headaches or temporary lightheadedness for some technicians. All transport regulations stick to the flammable solid class, with UN numbers hanging on drummed shipments. Regular emergency drills keep warehouses and lab floors prepared for spills or fires—even though outright accidents rarely occur with careful, trained handlers.
Application Area
Pure durene may sound niche, but several sectors rely on it. It acts as a starting material in the world of organic electronics, particularly for synthesizing modern OLED and semiconducting compounds. Traditional uses include resin and dye manufacture, where the symmetrical placement of methyl groups helps products achieve thermal and light stability no other aromatic hydrocarbon matches. In the plastics world, it forms backbones for polyimide films or acts as a phase transfer agent in specialty coatings. Pharmaceutical intermediates sourced from tetramethylbenzene find their way into drugs where high structural symmetry improves reactivity and purity during scale-up. Even fuel researchers eye it as a model compound for combustion studies because it reflects real-world engine behavior when burned.
Research & Development
Labs worldwide hunt for molecules that push materials and processes further. Research on tetramethylbenzene rarely gets splashy headlines, yet it moves steady in analytical chemistry journals and engineering papers. One trend involves using durene derivatives to build organic electronic components—think flexible transistors and light-emitting polymers. Other efforts focus on tweaking its methyl groups to turn out high-temperature resins that withstand harsh processing, targeting devices in aerospace and automotive sectors. Analytical chemists lean on it as a standard in calibration and as a matrix for mass spectrometry. Every few years, journals document a new catalysis pathway or synthesis trick that tightens up yields or slashes waste in tetramethylbenzene preparation, proving that even workhorse chemicals refuse to stand still under an inquisitive eye.
Toxicity Research
Most toxicology studies show that durene does not threaten adult health in small, controlled exposures. Inhalation of dust can irritate lungs or eyes, often flagged as an occupational nuisance rather than a life-shortening danger. Acute oral toxicity studies in rodents put LD50 values above 2,000 mg/kg, making it less risky than many industrial solvents. Long-term studies typically find no evidence that regular, low-level exposure produces cancers or organ damage, but environmental agencies regulate disposal to keep run-off and air emissions low. Because it's a hydrocarbon, spilled tetramethylbenzene can linger on surfaces or in groundwater—a fact that shapes local rules for handling and cleanup, especially around streams and sensitive zones.
Future Prospects
The business of specialty aromatics faces growing pressures: tighter environmental standards, demands for cleaner production, and the scramble for higher-value chemical synthesis. Tetramethylbenzene stands on solid ground. It already has a track record supporting advanced electronics, high-performance plastics, and safer solvents. Researchers continue hunting for eco-friendly ways to make it from renewable feedstocks—improving catalysts, turning to biomass, and designing closed-loop processes that marry efficiency with sustainability. If upcoming generations of flexible displays, lightweight devices, or greener polymer systems need chemical scaffolding, durene derivatives will likely factor in. Even now, a growing list of patent filings suggests industry watchers expect the compound’s circle to widen as rising demand for high-purity, thermally stable intermediates grows.
What is 1,2,4,5-Tetramethylbenzene used for?
Understanding Its Place in Chemical Manufacturing
Walk into any conversation about chemical feedstocks, and names like 1,2,4,5-Tetramethylbenzene—sometimes called durene—might not come up much. This compound fills a surprisingly important slot in the production lines of industries that often stay out of the public eye. It takes a key role as a building block in organic synthesis, serving as an intermediate that helps chemists design and build more complex molecules.
Real Uses, Real Impact
One area where durene shines is in the manufacture of pyromellitic dianhydride (PMDA). That name sounds intimidating, but PMDA actually forms the backbone of some useful plastics, particularly polyimide resins. You run into polyimides if you've ever handled flexible printed circuit boards, high-temperature-resistant films, or certain specialty adhesives. Electronics makers count on these materials when they need insulation that won’t fail under heat. One can forget the role small molecules like this play in keeping our devices running safely.
There’s also a link between 1,2,4,5-Tetramethylbenzene and explosives, though not in the headline-making way people might imagine. Chemical engineers use it to help make explosives that demand reliability and precision, rather than brute power. Its predictable chemical structure allows for consistency batch after batch, reducing accidents. These practical details sometimes get overlooked until a disaster brings up questions about supply chains and safety.
Why Purity and Process Matter
Dealing with aromatic chemicals like this one means purity isn’t just a best practice; it’s a necessity. When producing PMDA or other derivatives, even small impurities can cause performance problems in the end product. Poorly manufactured durene can bring headaches down the line, leading to defective insulation or circuit failures in electronics. Here, process control and attention to detail show the skill of the producer, not just the efficiency of a factory.
Honestly, most folks won’t hear about 1,2,4,5-Tetramethylbenzene unless there’s a shortage or an accident. Yet sourcing reliable material underpins plenty of high-tech manufacturing. In my time talking with materials engineers and chemists, many stress how much trouble sub-par raw materials can cause. One small supplier error can ripple all the way to product recalls and customer complaints. This chain of responsibility often stays hidden from the public, overshadowed by more visible parts of the tech industry.
Looking at Safer Handling and Environmental Challenges
Handling aromatic hydrocarbons always brings up questions about worker safety and environmental impact. Facilities that deal with 1,2,4,5-Tetramethylbenzene face the same scrutiny found across the chemical sector. Solvents, aromatic vapors, and contaminated water mean companies have to prove their commitment to safety, not just for employees but for surrounding communities. Regulations keep tightening, and for good reason. Incidents involving leaks or improper handling might seem rare, but each one erodes public trust. Industry experts continue to invest in better extraction and recycling methods to cut waste and reduce risk.
Potential Paths Forward
Innovation rarely starts at the consumer end with these materials. Instead, research labs dig into better catalysts or new processes to synthesize durene more efficiently. Efforts to reclaim it from chemical waste streams also point toward a future less dependent on new fossil resources. Companies see value in this not just because of tighter rules, but because raw material prices can swing fast.
It’s easy to forget how much depends on a few grams of a substance like 1,2,4,5-Tetramethylbenzene. Dig a little deeper, and this mundane-sounding aromatic ring looks a lot more important than its unglamorous name might suggest.
What is the chemical formula and structure of 1,2,4,5-Tetramethylbenzene?
Understanding 1,2,4,5-Tetramethylbenzene
For years, working in chemistry labs, certain molecules stick with you—not just because they're common, but because their structure tells a story. 1,2,4,5-Tetramethylbenzene is one of those. This compound belongs in the family of aromatic hydrocarbons known as methylbenzenes. Its chemical formula, C10H14, looks simple on paper, but the way the atoms fit together defines its character, uses, and even its hazards.
The Real-World Structure
Imagine a benzene ring, itself a symbol of aromatic chemistry. Instead of six identical hydrogens around the ring, here you swap four for methyl groups. These groups land on the 1, 2, 4, and 5 positions of the benzene ring. The result is a molecule with added bulk and a slightly shifted set of chemical properties compared to plain benzene or toluene. The IUPAC name, 1,2,4,5-tetramethylbenzene, points straight at the arrangement: each of those four carbons carries a -CH3 group. Chemists often draw it as a hexagon with alternating double bonds, dots at positions 1, 2, 4, and 5, and short lines for methyls branching off those spots.
Beyond the Formula: Why It Matters
I’ve had my share of confusion with isomers—molecules with the same formula but different layouts. 1,2,4,5-Tetramethylbenzene stands out from its isomers such as durene, which carries methyls at the 1,2,3,5 positions. This difference changes physical traits: melting point, solubility, and reactivity. For instance, 1,2,4,5-Tetramethylbenzene melts at a much higher temperature than some isomers. I remember using it as a solid reference in a melting-point experiment. It also has a specific use as an intermediate in making other chemicals—think dyes and perfumes. The dense arrangement of methyl groups changes how it reacts in electrophilic substitution reactions, compared to plain benzene or toluene, guiding chemists toward specific synthetic pathways.
Hazards and Handling
In many labs I’ve worked, safety data hangs by every fume hood for a reason. Compounds like 1,2,4,5-Tetramethylbenzene deserve respect. Despite a pleasant-sounding use in fragrances, this is no ordinary "aroma" molecule. At room temperature, it stays solid, but it’s still volatile enough for inhalation risks to pop up. Eyes and skin can get irritated; breathing its dust or vapors can irritate airways. Storage calls for well-ventilated, cool spaces, away from open flames or oxidizers. These facts matter for health and legal compliance. Too many labs ignore risk because a substance seems benign.
How Do We Approach These Chemicals?
It's important to stay aware, use up-to-date safety resources, and never rely on old habits. Digital safety sheets now offer nearly instant updates—useful for anyone handling methylbenzenes. For disposal, don’t pour it down the drain. Instead, follow hazardous waste protocols to prevent soil and water contamination. The key is embedding safety and environmental consciousness into daily work—whether you’re in research, manufacturing, or teaching.
Future Pathways
As more labs turn to green chemistry, the impact of aromatics like 1,2,4,5-Tetramethylbenzene draws greater scrutiny. More sustainable synthesis routes, better recycling practices, and smarter substitutions will likely shape how we use molecules like this. The formula and structure give us a starting point, but responsibility goes deeper—it’s about understanding the molecules, their risks, and their role in the bigger chemical picture.
Is 1,2,4,5-Tetramethylbenzene hazardous or toxic?
Understanding an Everyday Chemical
Folks who spend time around industrial chemicals know the long names often seem scarier than the substances themselves. Still, any chemical can pose risks if handled the wrong way. 1,2,4,5-Tetramethylbenzene pops up in lab settings, petrochemical processes, and even as a byproduct in refineries. It’s a four-methyl cousin of benzene, and methyl attachments always change the story compared to the original molecule.
Health Concerns: Fact Over Fear
Speak with any health and safety pro, and you’ll usually hear one rule: Don’t eat, drink, or breathe chemicals unless you know exactly what they are and what they do. The research on 1,2,4,5-tetramethylbenzene suggests that it isn’t as notorious as benzene itself, which we know causes cancer over long-term exposure. Still, methylbenzene derivatives like this one can irritate eyes, skin, and the respiratory tract. That’s from direct reports and animal studies, not just guesswork.
There’s a reason the Material Safety Data Sheets for these compounds recommend gloves, goggles, and keeping a tight lid on ventilation. Got a headache or started coughing after a spill? That’s your body reacting to the fumes. Eye redness or itchy skin can crop up after direct contact. Long story short, acute exposure usually leads to local irritation before anything else—serious, but not in the same league as chronic issues tied to benzene over years.
Environmental Impact Shouldn’t Be Ignored
Go upstream or downstream from a refinery, and the subject of chemical leaks always comes up. 1,2,4,5-Tetramethylbenzene survives a while in the environment because of those tightly bound methyl rings. Break down takes place, but it’s not a quick process, especially if the chemical spills into a water source or soil. Toxicity for fish and other aquatic life tends to ramp up when these substances build up. Regulators require paperwork and containment systems for a reason. Leaks can hurt local ecosystems if left unchecked, and the aftermath of a spill can take years to recover from.
The Right Way to Handle the Stuff
By now, most labs, factories, and refineries lean on standard procedures: Storage in airtight containers, use of chemical fume hoods, personal protective equipment for all workers. Good habits matter more than flashy new gear. The old veterans on the job always say that proper housekeeping and clear labels keep folks safer than the fanciest alarms. Forgetting the safety basics brings trouble fast, whether it's burns, breathing trouble, or environmental messes.
Back in the day, mistakes happened because chemicals weren't taken seriously. Now, training covers more real-world stories—3 a.m. spill drills, regular reviews, updates when research finds out something new about the risks. Sharing that knowledge saves headaches and lawsuits.
Pushing for Safer Chemistry
Modern industry keeps searching for substitutes that get the job done without bringing health or environmental baggage. Regulations also pressure companies to track their emissions and waste, making it harder to ignore the downsides of chemicals like 1,2,4,5-tetramethylbenzene. Community advocates and worker unions keep asking tough questions about what’s in the air and water. These hard conversations have pushed safer handling and, where possible, safer alternatives.
Experience on the ground proves this: Respect the risks, trust the data, and nobody gets hurt. Ignore the warnings, and no amount of paperwork fixes the mess.
How should 1,2,4,5-Tetramethylbenzene be stored and handled?
Recognizing the Real Risks
Many folks working in labs or chemical plants can spot a benzene derivative by smell or structure, but 1,2,4,5-Tetramethylbenzene brings challenges some don’t expect. This compound shows up as a crystalline solid with a slight, sweet odor, easy to overlook in a busy storeroom. Yet ignoring care compounds the dangers. I’ve watched more than one rookie surprised when improper storage led to unexpected leaks or contamination. All it takes is one rushed day to turn a small hazard into a larger problem.
Storage: More Than Just a Place on the Shelf
This isn’t a compound to toss next to anything else. Each time a drum arrives, trained eyes should check for signs of moisture or heat exposure. Damp or hot environments speed up decomposition. Solid organic compounds sometimes seem less intimidating, but ignoring temperature control risks purity. In buildings using open shelving, the odds of cross-contamination rise fast. A clean, cool, well-ventilated room serves better than a mixed storage closet gathering dust.
Start by isolating this compound from strong oxidizers. One mistake—putting it next to nitric acid—can land a whole team in trouble. Years back, a reckless storage system in one workshop led to a close call. Everyone remembers lessons from that day. Now, sealed, labeled containers get their own shelves, with regular checks to spot leaks or bulges. Some companies use explosion-proof refrigerators calibrated below 30°C, cutting risk and keeping insurance happy.
Personal Protection Means Respecting the Details
I remember a young intern trying to handle samples with bare hands—bad idea. Gloves do more than prevent skin irritation; they guard against accidental absorption. Nitrile works best. Eye protection and lab coats, already standard in many labs, become crucial here. Vapors hang around in enclosed areas, and repeated exposure over time raises serious health risks.
If a spill escapes containment, proper cleanup makes all the difference. I once saw an experienced colleague immediately grab absorbent pads, then open every nearby window. Quick, thorough action prevented vapor buildup and permanent damage. Disposing of waste through the right channels—not the sink—keeps toxic compounds out of the water supply, a point people sometimes forget under stress.
Investing in Training and Regular Audits
The best-written protocols fall short if nobody understands them. Ongoing safety training helps everyone spot weak points in handling routines. Mixing up safety drills with real-life stories from the floor sticks with people—most remember losing their lunch break to a spill more than any theory class. Management has a role too: regular audits pick up on careless stacking or unlabeled jars before trouble starts.
Solutions Rooted in Experience
Some teams go beyond printed checklists by marking floors and shelves specifically for different hazard levels. That small step makes it much harder to stash incompatible chemicals together. Investing in spill kits and ventilation doesn’t cost much compared to an emergency room visit or a shutdown. Keeping a clear emergency plan—one known by every worker on the shift—has bailed out more than one crew from a bad spot.
In my time on the floor and at the bench, respect for compounds like 1,2,4,5-Tetramethylbenzene grows from hard lessons and shared responsibility. Proper handling starts long before an incident—and keeps people safe long after.
What are the physical properties of 1,2,4,5-Tetramethylbenzene (melting point, boiling point, solubility)?
Why Study 1,2,4,5-Tetramethylbenzene?
1,2,4,5-Tetramethylbenzene, also called durene, holds an interesting spot among aromatic hydrocarbons. Its structure—four methyl groups crowding a benzene ring—transforms how it behaves. Industrial folks and researchers care about this compound, not just for curiosity’s sake, but due to practical reasons like safety in labs, handling, and its use in dye, plastic, and pharmaceutical work.
Melting Point: Solid at Room Temperature
Durene’s melting point sits around 79°C (174°F). That’s higher than many other substituted benzenes, and the reason sits in its symmetry. The four methyl groups keep the molecule balanced, pushing its molecules to pack tightly in a crystal network. That tight fit builds up stronger intermolecular force, so more heat is needed to shake things loose. If you’ve ever tried scooping out durene, you’ll notice its waxy, crystalline texture—a reminder of that firm molecular grip.
Boiling Point: Volatility and Stability
Set a flask over a bunson burner and durene starts boiling at about 196°C (385°F). That’s no lightweight number and means it hangs together as a solid through a broad temperature range. In the lab, this lets chemists purify or distill durene without rushing, since it won’t evaporate at room temperature like lighter aromatic relatives, such as toluene. The high boiling point also cuts down risks of inhalation exposure during standard handling.
Solubility: Loves Organics, Shuns Water
Durene falls in line with most aromatics—water barely makes a dent. Drop some in a beaker and you’ll see it float or sink, refusing to mix. Water molecules want to bond with each other, not the oily durene, so the two will always separate. Try dissolving it into a bit of ether, chloroform, or hot acetone, and it slides in easily, showing off its preference for organic solvents. That shapes how it’s handled, from extraction to cleanup; standard industrial solvents deal with durene, not water.
Health and Practical Concerns
With a melting point above room temp, durene usually remains solid, cutting down opportunities for accidental spills or inhalation. High boiling means less vapor, but folks should still work in ventilated areas—organic solvents used for durene can carry health risks. Glove use protects against contact since methylated aromatics sometimes irritate skin.
The Bigger Picture and Safer Practices
Durene’s physical traits shine brightest when you look at industry routines. Purification depends on its solid state; workers heat it above the melting point and then cool it down for crystallization. Companies drawing out durene from coal tar or petroleum must use safety gear, especially since organic vapor from solvents becomes the real exposure risk. Investing in enclosed systems or vent hoods keeps the workspace cleaner. Institutions train staff on solvent use. A strong eye for solvent choice and temperature control keeps processes tight and safe.
Closing Thoughts on Durene’s Role
Getting familiar with the unique behavior of 1,2,4,5-tetramethylbenzene isn’t just textbook science—it’s the kind of detail that saves time, money, and helps people work smarter. By looking close at its melting and boiling points and seeing how it shuns water for organic solvents, chemists and manufacturers steer clear of trouble, keep labs safe, and turn a simple aromatic into a tool for progress.
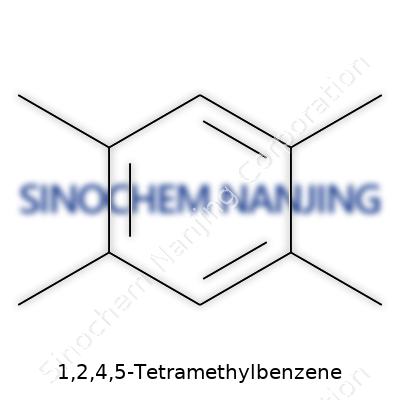
| Names | |
| Preferred IUPAC name | 1,2,4,5-Tetramethylbenzene |
| Other names |
Durene Prehnitene |
| Pronunciation | /ˈwʌn ˈtuː fɔːr faɪv ˌtɛtrəˈmɛθ.əlˌbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 95-93-2 |
| Beilstein Reference | 1209240 |
| ChEBI | CHEBI:28856 |
| ChEMBL | CHEMBL157485 |
| ChemSpider | 6810 |
| DrugBank | DB01856 |
| ECHA InfoCard | 100.010.888 |
| EC Number | 201-852-7 |
| Gmelin Reference | 56648 |
| KEGG | C01746 |
| MeSH | D015555 |
| PubChem CID | 6977 |
| RTECS number | CG3850000 |
| UNII | 6V9V1H64K |
| UN number | UN2268 |
| CompTox Dashboard (EPA) | DTXSID9020715 |
| Properties | |
| Chemical formula | C10H14 |
| Molar mass | 134.22 g/mol |
| Appearance | Colorless crystalline solid |
| Odor | Aromatic odor |
| Density | 0.867 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.9 |
| Vapor pressure | 0.13 mmHg (25°C) |
| Acidity (pKa) | 14.0 |
| Magnetic susceptibility (χ) | -74.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.495 |
| Viscosity | 3.9 mPa·s (20 °C) |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 17.51 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2871.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D02BB03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P261, P273, P280, P301+P312, P304+P340, P312, P403+P235 |
| NFPA 704 (fire diamond) | 1,2,4,5-Tetramethylbenzene: "1-2-0 |
| Flash point | 52 °C (126 °F; 325 K) |
| Autoignition temperature | 530 °C |
| Explosive limits | 1.1–6.4% |
| Lethal dose or concentration | Lethal dose or concentration: LD50 oral (rat): 3280 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3280 mg/kg (rat, oral) |
| NIOSH | GR0700000 |
| PEL (Permissible) | 25 ppm |
| REL (Recommended) | 125 mg/m³ |
| IDLH (Immediate danger) | IDLH: 900 ppm |
| Related compounds | |
| Related compounds |
Benzene 1,2,3,4-Tetramethylbenzene Durene 1,2,3-Trimethylbenzene 1,3,5-Trimethylbenzene 1,2,4-Trimethylbenzene Toluene Xylene Mesitylene Pentamethylbenzene |

