1,2,4,5-Tetrachlorobenzene: A Deep Dive
Historical Development
Back in the late 1800s, chemists began exploring how chlorinated benzenes could change both farming and industry. Research papers and patents from the early twentieth century map a trail of experiments where chlorination of benzene led, steadily, to a group of chemicals with varied uses. By the 1940s, demand for compounds that could boost crop yields or serve as feedstock in growing chemical sectors brought 1,2,4,5-tetrachlorobenzene out of laboratory curiosity and into real production lines, especially in Europe and North America. The Cold War era’s industrial expansion only accelerated the need for building block chemicals like this—its role in synthesizing herbicides and as an intermediate in dyestuff production locked in steady demand. Today, regulations and market forces keep pushing for cleaner processes, but those early decades built both the knowledge and environmental burden we deal with now.
Product Overview
1,2,4,5-Tetrachlorobenzene stands out from its cousins thanks to its specific arrangement of chlorine atoms on the benzene ring—this pattern shapes not only its reactivity, but the way industries can use it. Sitting in a crystalline state at room temperature, this aromatic chemical has developed a reputation both as a valuable intermediate and a problematic pollutant. Its primary markets lie in synthesis: companies turn to it when building up more complex molecules for agrochemicals or specialty polymers. Over time, its direct application has narrowed, replaced by other less persistent chemicals, yet its shadow remains in old stockpiles, contaminated sites, and as a breakdown product of heavier industrial chlorination.
Physical & Chemical Properties
Anyone handling 1,2,4,5-tetrachlorobenzene day-to-day notices its pale, sometimes off-white crystals and its barely-there aromatic aroma. It boils at about 280°C and melts near 137°C. The molecule doesn’t dissolve well in water at all, but it mixes much more easily with organic solvents like ethers or chlorinated hydrocarbons, which helps in industrial applications. Its specific molar mass—217.9 g/mol for those keeping score—gives it a density just north of 1.5 g/cm³. This compound resists breaking down, both in environmental and chemical conditions, which makes it durable in pipelines, storage, or waste streams but also notoriously hard to clean up from the environment.
Technical Specifications & Labeling
Regulations tie strict labels to shipments of 1,2,4,5-tetrachlorobenzene. International agreements like GHS color-code its hazards: the compound meets H400 (hazardous to aquatic life) and H351 (suspected carcinogen in humans) criteria. Shipping containers and drums carry UN numbers and risk pictograms, backed by batch-specific purity data—usually greater than 98%, with impurities of other chlorobenzenes or polychlorinated biphenyls closely monitored. Companies may analyze lots using GC-MS and provide safety data sheets in multiple languages, especially for international shipments. These practices reflect more than just red tape—people and ecosystems depend on real-time, reliable information to reduce danger.
Preparation Method
Large-scale production usually involves direct chlorination of benzene under controlled temperatures and pressures, using iron or ferric chloride as a catalyst. Achieving the 1,2,4,5 substitution pattern requires precision over reaction conditions, with staged addition of chlorine and careful quenching to limit side-products. The process generates multiple chlorobenzenes as by-products that companies must separate using distillation or crystallization. Afterward, solidified tetrachlorobenzene gets washed and dried until it meets technical specs suitable for downstream chemistry. Process improvements sometimes include greener solvents or attempts to recover energy from the exothermic reaction, but legacy plants may still use methods dating back to the mid-1900s.
Chemical Reactions & Modifications
On the lab bench, 1,2,4,5-tetrachlorobenzene enables a bunch of transformations thanks to its reactive chlorine substituents. Chemists can swap those chlorines for nitro, amine, or alkoxy groups, paving the way toward dyes, specialized agricultural chemicals, and flame retardants. It also gets used in coupling reactions to build polycyclic aromatics. These transformations demand careful handling and the right reagents—a slip-up brings hazardous by-products or environmental releases. Longstanding experience shows just how versatile this molecule can be, but each use brings trade-offs, especially with waste and toxicity.
Synonyms & Product Names
Over the years, this compound has picked up a collection of names—sometimes a headache in regulatory paperwork. Besides “1,2,4,5-tetrachlorobenzene,” industry specs and global trade lists also call it “TCB,” “tetrachlorobenzene (1,2,4,5-),” or just “aromatic tetrachloride” in older texts. Some suppliers brand their high-purity versions for laboratory use, while technical-grade materials for manufacturing go by broader trade names. Consistent naming protects both suppliers and buyers from costly mix-ups, especially given the presence of related but less toxic isomers.
Safety & Operational Standards
Anyone who has spent time around warehouses or chemical plants knows safety is not just paperwork—it's routine. Workers wear gloves, goggles, and full suits when handling 1,2,4,5-tetrachlorobenzene, based on occupational exposure limits set by agencies like OSHA and the European Chemicals Agency. Facility air gets filtered and monitored for accidental releases, especially since this compound travels in dust particles or vapor. Environmental plans require capturing spills and controlling discharge into waterways. Real-world experience highlights regular drills and medical monitoring as non-negotiable, because historic exposure cases show real risks for skin, respiratory, and long-term cancer outcomes.
Application Area
1,2,4,5-Tetrachlorobenzene filled a niche in the agricultural revolution, appearing in the ingredient lists for both herbicides and pesticide intermediates. It found uses as a starting point in dye manufacturing—the sort of color-fast dyes needed for army surplus or industrial uniforms. In plastics, the compound sometimes acts as a flame retardant additive, boosting safety ratings for building materials or consumer goods. These days, regulations have trimmed back many of those uses, forcing companies to reassess or move toward alternatives, but in parts of the world with loose oversight it still pops up in legacy applications and unregulated supply chains.
Research & Development
Research circles constantly probe ways to reduce the environmental impact of chlorinated benzenes—developers chase improved catalysts, recycled by-product systems, and routes that skip harmful solvents entirely. Universities and industrial labs publish findings on cleanup technologies like advanced filtration or microbial breakdown, driven by persistent soil and water contamination. Ongoing studies examine substitution strategies, where less-chlorinated or non-aromatic compounds recreate essential chemical properties with fewer hazards. Collaborations among regulators, industry, and academia shape new product stewardship programs—partly out of practical necessity, partly out of growing consumer and watchdog pressure. Personal experience from industry meetings shows excitement and skepticism blink side by side—solutions move slowly, sometimes facing old habits and tight margins.
Toxicity Research
Long-term studies on animals link 1,2,4,5-tetrachlorobenzene to liver enzyme changes, organ inflammation, and increased tumor formation—facts that have pushed most developed nations to restrict or ban its emission. Acute exposure cases, rare but well-documented, trigger skin rashes, respiratory discomfort, or nervous system symptoms in unprotected workers. Bioaccumulation and slow breakdown mean this compound lingers in food webs, with wildlife and humans both vulnerable if environmental controls slip. Decades of field research in contaminated areas, especially near legacy production sites, show lasting health impacts, pushing governments to invest in new remediation or health surveillance plans.
Future Prospects
Looking forward, 1,2,4,5-tetrachlorobenzene’s story is at a crossroads. Companies are taking hard looks at the costs—not just in dollars, but in regulatory fines, cleanup obligations, and battered public image. Alternative chemistry is becoming the new normal, and international trade agreements pressure holdout nations to follow stricter rules. Renewed research into green chemistry lights up the conference circuits, with talk of biodegradable intermediates and recyclable catalysts. On cleanup frontlines, technical solutions like thermal desorption or bioremediation grow more affordable as need rises. Transparency and accountability now drive real change in chemical manufacturing. Old habits linger, but those exposed to legacy risks, whether workers or neighbors, are not letting their stories go untold.
What is 1,2,4,5-Tetrachlorobenzene used for?
What Makes 1,2,4,5-Tetrachlorobenzene Stand Out?
1,2,4,5-Tetrachlorobenzene enters conversations mostly in industries working with specialty chemicals. On the periodic table of workplace smells and hazards, you won’t want this one in your lunchbox. It’s tough—persistent, stable, and slow to break down. Many times I’ve heard factory workers talk about the chemical as that “sticky” compound, hard to wash off both hands and equipment.
How Industries Make Use of It
This compound finds its way into the manufacture of other chlorine-based chemicals. Talking to an uncle who works in paint development, I heard about its value as an intermediate, basically a stepping stone, in making herbicides, dyes, and even some rubber products. It’s not the flashy, final ingredient. It’s more like flour in baking—no bread without it, but no one celebrates it. Most notably, it supports the synthesis of pentachloronitrobenzene, used in fungicides.
Some older records mention its past use in making coolants and lubricants for heavy machinery. By the time these oils and greases are pumped through large equipment, the original chemical gets transformed into something less familiar, but that first step started with compounds like this one.
Risks and Impact on Health
The problem starts when you realize this chemical isn’t just hard on pipes—it’s hard on people and wildlife too. It’s one of those compounds that lingers, sometimes ending up where nobody wanted it. My friend working in hazardous waste cleanup says that sites contaminated with this stuff often need careful, costly attention. Prolonged exposure can attack the liver or nervous system. The United States Environmental Protection Agency places it on a list of persistent organic pollutants because it clings to soil, moves slowly through water, and shows up in animal tissues.
Long-term, the impact weighs heavily on agricultural communities. A grandfather living in the Midwest often complained about the “chemical dust” from nearby fields, and Department of Agriculture studies back up those stories. Over time, a buildup of these chemicals in soil can bite back, ruining crops or flowing into water supplies.
Real-World Solutions
If people want factories and fields to stay productive without harming their neighbors, better management of chemicals like 1,2,4,5-Tetrachlorobenzene matters a lot. That can mean switching to less harmful intermediates, or changing the way disposal works. Companies in Europe have already shifted away from using this chemical by tightening rules and finding substitutes in manufacturing. Regulations forced inventors to come up with new compounds that don’t build up so much in the environment.
Community awareness plays a big role, too. Local health agencies now test for these residues, especially near old landfills or chemical plants. It takes teamwork between city officials, farmers, and companies to map out these hotspots and clean them up. On a personal level, talking honestly about industrial dangers and insisting on regular monitoring seems small, but I’ve seen communities turn that vigilance into real change: newer filtration systems, stricter safety training, even policies that keep kids away from certain fields during spraying season.
Final Thoughts
1,2,4,5-Tetrachlorobenzene works behind the scenes in industrial chemistry, but its consequences stretch far. Strong oversight, smarter manufacturing, and regular cleanup make the biggest difference. You don’t need to see this chemical up close to feel its effect—its story lingers in the soil and water, pressing us all to pay closer attention.
Is 1,2,4,5-Tetrachlorobenzene hazardous to health?
Looking Closer at 1,2,4,5-Tetrachlorobenzene
Chemicals like 1,2,4,5-Tetrachlorobenzene show up in industrial spaces, labs, and sometimes in places they don’t belong. This complex name covers a chlorinated substance pulled from benzene—a base many industries use for plastics, dyes, and pesticides. Anyone spending much time in a chemistry lab will spot long lists of chemicals like this, but the tricky part is knowing which ones could hurt you and which ones simply look intimidating.
Health Risks From Real Exposure
Ask anyone who has worked around strong chemicals—many stories start with odd headaches, irritated eyes, sore throats, or feeling off after a short time at work. Reports show 1,2,4,5-Tetrachlorobenzene targets the liver and kidneys. Breathing its vapor or getting it on skin sets people up for harm. I’ve seen coworkers brush off gloves in the rush of the day—skin contact often felt minor at the time, but those exposures can stack up.
The U.S. Environmental Protection Agency warns that chlorinated benzenes aren’t just workplace hazards. If these leak into rivers or soil, neighborhoods could pick up long-term risks as well. Animal tests point to organ trouble, reduced growth, and problems with blood cells. People don’t line up for these tests, so the details on human effects come mostly from accidents and bad workplace habits. Safety data sheets stress limits: keep levels as low as possible, use strong ventilation, and never skip personal protective gear.
Real World Scenarios Show Higher Risk
A few years ago, I toured an old chemical plant being torn down. The workers wore respirators and taped sleeves—exactly the right move. By the dumpsters behind the fence, chunks of soil tested positive for chlorinated benzenes. The local cleanup teams moved the dirt, checked air quality, and set tight protocols. Not every site takes these issues as seriously, but the ones that do keep people safe and avoid big medical bills later.
Skip ahead to the community side. Not everyone near industrial hotspots learns about what lurks on old land. Groundwater can travel farther than most think. In places with looser controls, people sometimes end up growing vegetables or swimming in water that has trace chemical leftovers. Public health campaigns can make all the difference. A trusted source of facts—maybe the local clinic or a skilled journalist—brings real change by letting people know what symptoms to watch for and where to get water tested.
Better Steps for Protection
Hard lessons teach safety best. Chemists, plant workers, and cleanup crews all benefit from stronger training and real talk about health outcomes. Instead of hiding behind technical jargon, open discussion makes dangers clear. Workers should ask for, and get, gloves, masks, and safe work plans built on tested research. Managers who keep safety at the front end up with fewer missed shifts, less turnover, and healthier teams.
Community leaders and healthcare providers can team up to watch for signs of exposure in neighborhoods sitting near old factories or waste sites. Quick response matters—constant testing, honest outreach, and clear reporting all cut the odds of small leaks turning into real health crises.
Looking Ahead
Dealing with chemicals like 1,2,4,5-Tetrachlorobenzene calls for focus, knowledge, and a level head. Left unchecked, the risks are real—but armed with facts, solid gear, and a community tuned in, the dangers shrink fast.
What are the handling and storage recommendations for 1,2,4,5-Tetrachlorobenzene?
Understanding the Risks
Anyone working with industrial chemicals recognizes the hazards hidden behind technical names. 1,2,4,5-Tetrachlorobenzene doesn’t make the headlines often, but anyone dealing with it ought to respect its risks. This substance used to appear in older pesticides and remains in a handful of specialty chemical processes. Its legacy includes toxic effects and stubborn persistence in the environment.
Safe Handling Starts with Awareness
Donning gloves or goggles shouldn’t become a ritual done on autopilot. Contact with 1,2,4,5-Tetrachlorobenzene can irritate your skin and eyes. Breathing in its dust or vapors can lead to headaches and other symptoms. Building a culture where nobody cuts corners matters far more than any checklist pinned to the wall. Good habits keep teams healthy. Always work under a fume hood or in a well-ventilated area, and limit routes for the chemical to escape.
Personal Protective Equipment
Chemical-resistant gloves, long sleeves, safety goggles, and a lab coat make up standard gear. Respirators enter the picture if there’s any risk of inhaling the dust or vapor. Take off any contaminated clothing at once. From my own days working in older industrial facilities, peer checks have stopped more accidents than any sign or memo. Real safety comes from people looking out for each other, speaking up, and staying focused.
Storage Concerns That Can't Wait
Storing this material right isn’t an afterthought. 1,2,4,5-Tetrachlorobenzene needs cool, dry, well-ventilated storage, far away from food, feed, or anything meant for people or animals. Keep it locked up, with warning signs in plain language. Steel or HDPE containers with tight-fitting lids work best to prevent leaks. From hard-earned lessons in older labs, always keep inventory tight—a smaller container or an organized storehouse often makes spills less costly to contain.
Labeling and Segregation
Labels have saved lives. Every bottle or drum should carry the chemical’s full name and a hazard pictogram. Keep incompatible materials apart. Strong oxidizers or acids stored together with chlorinated aromatics can set off reactions that turn bad fast. Color-coding shelves or bins makes those dangers easier to spot even when people change shifts.
Cleanup and Waste Disposal
Spill response kits need to sit within arm’s reach, not somewhere in a supervisor’s office. Absorb spills with sand or vermiculite and use sealed bags for cleanup debris. Dispose wisely—most jurisdictions treat this as hazardous waste and will want a manifest on every shipment out. There’s no shortcut to legal or safe disposal.
Checks and Oversight
Never just trust one inspection, one training session, or one person’s memory. Schedules for routine checks, backed up by real records, uncover leaks and near-misses before trouble turns serious. Regulators don’t accept excuses, and neither should anyone who works with dangerous chemicals. It’s not paperwork for its own sake. It’s the difference between a regular day and a disaster.
Better Systems Mean Fewer Accidents
The best place I ever worked didn’t just put up safety posters; they welcomed questions and expected honest feedback after every incident, large or small. Anyone handling 1,2,4,5-Tetrachlorobenzene should expect as much from their workplace. It only takes a single mistake to remind everyone why these rules exist. Consistent habits, strong safeguards, and teamwork form the only real answer for protecting people and the environment from industrial chemicals like this one.
How should 1,2,4,5-Tetrachlorobenzene be disposed of safely?
Understanding the Chemical’s Risks
1,2,4,5-Tetrachlorobenzene has built up a reputation for being more trouble than it’s worth outside the research lab. Its structure makes it stick around longer in the environment, and once it ends up in water or soil, it resists natural breakdown. Research from the U.S. EPA and European Chemicals Agency shows this chemical can linger in the air and create toxic byproducts when burned the wrong way. My background in environmental monitoring drives home the worry: trace amounts can build up in places you don’t want, especially where food grows or people live.
Why Tossing It Out Won’t Do
Lots of folks don’t realize how easy it is for industrial waste to end up as a long-term headache. Tossing 1,2,4,5-Tetrachlorobenzene in the garbage or pouring it down the drain might seem convenient, but landfill leaks and sewer systems just send the problem somewhere else. Wastewater treatment isn’t designed for molecules like this. In my time volunteering with community cleanup, I’ve seen what happens when shortcuts get taken—it means groundwater risks, contaminated soil, or even health warnings for nearby residents. This stuff doesn’t just wash away with the next rainstorm.
Trusted Methods for Treating Hazardous Chemicals
Working with hazardous materials means sticking to methods backed by years of science. Professionals use high-temperature incineration for chemicals like 1,2,4,5-Tetrachlorobenzene. These specialized incinerators reach temperatures that break chemical bonds, turning stubborn toxins into safer gases with filters that trap the rest. The U.S. Department of Energy lists incineration at over 1,200 °C as reliable for chlorinated organics. Anything lower leaves you with toxic ash and fumes.
Chemical treatment offers another route, but only under tight controls. Highly trained teams use reagents that react with the chemical structure, transforming the substance into something less persistent or toxic. Every batch gets tested carefully–this stage can’t be rushed. Improper handling or incorrect mixing sets up a bigger hazard. Local authorities require hazardous waste permits, ensuring handlers trained in chemical safety supervise the process each step of the way.
Why Certified Facilities Make the Difference
From my experience consulting with universities and industrial labs, the greatest defense against environmental trouble comes from using certified hazardous waste companies. These folks track every drum and batch, record the chemical’s life cycle, and maintain strict chain-of-custody documents.
Most states and countries publish lists of registered disposal firms. Companies with ISO 14001 certification and hazardous substance transport licenses invest in staff training and equipment. They collaborate with regulatory agencies like the EPA, ensuring every stage meets public health and environmental standards. My students used to cringe at the red tape, but those forms and logs make it possible to trace problems quickly if something leaks or goes missing.
Safer Practices for Everyday Users
Even small labs—or home-based chemists—play a role. Never store hazardous chemicals in food containers or regular trash bins and always label everything clearly. Collect small amounts in leak-proof containers, then contact local hazardous waste collection programs for regular drop-off days. Public health officers sometimes organize community events where you can turn in old chemicals without charge.
Real progress comes from a mix of good habits, transparency, and relying on proven disposal options. Safe disposal protects more than the environment; it protects families, workers, and neighbors for years to come.
What is the chemical structure and formula of 1,2,4,5-Tetrachlorobenzene?
Looking Closely at the Structure
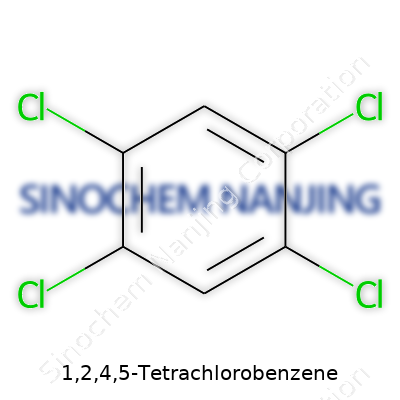
Many folks have run into chlorinated benzene compounds somewhere in their work—maybe in a lab notebook or maybe printed on the back of a chemical drum. 1,2,4,5-Tetrachlorobenzene draws attention in the field of industrial chemistry and environmental science. This compound comes with a benzene ring, which serves as a kind of framework in organic chemistry, and in this version, four chlorine atoms take the spots at positions 1, 2, 4, and 5 on the ring. Plenty of diagrams will show those chlorines as green dots on the edges of a six-sided ring, but at its core, each carbon in the ring attaches to a hydrogen or a chlorine, with four hydrogens swapped out by chlorines.
The chemical formula is simple but tells a lot: C6H2Cl4. That means the molecule carries six carbon atoms, just two hydrogen atoms left (since four have been replaced), and four chlorines tightly bound to the structure. This specific arrangement makes the compound less reactive than some other chlorinated benzenes, which explains its stability when stored or transported. From my experience moving chemicals in a warehouse, compounds with fewer hydrogens tend to resist breakdown, especially compared to more reactive cousins.
Why Structure Affects Use and Safety
Structure tells scientists and workers a lot about risk. Chlorines attached to benzene rings often signal problems for nature and people. They don’t break down easily, so they stick around in soil and water. The Environmental Protection Agency points to how these chemicals can build up in the environment and cause harm even in low concentrations. In real-world clean-up, folks wearing gloves and respirators remember how persistent these molecules can be. Just a simple shift in atom position along the ring makes regulations and safety protocols stricter or more relaxed.
Factories use 1,2,4,5-tetrachlorobenzene as a building block for pesticides, lubricants, or specialty fluids. It doesn’t show up much in consumer products, but it can escape as a byproduct in older manufacturing plants. In one clean-up project I joined, we tracked vapor levels in an old warehouse to trace spilled tetrachlorobenzenes lurking under the concrete slab. Years after closure, traces still showed up during summer tests.
Managing Risks and Seeking Solutions
Simple chemistry lessons shape big decisions in industry and waste management. Safer handling often starts with a better understanding of a compound’s structure. Workers who know the formula C6H2Cl4 can better predict what kind of gloves withstand the chemical and which filters trap its fumes. Effective training and real-time monitoring cut down on the kinds of accidents that lead to costly spills or long-term contamination. Some newer remedial technologies, like advanced oxidation or bioremediation, target these stable molecules, breaking the bonds that make chlorinated benzenes so tough to remove from the environment.
Switching to less persistent chemicals in some applications reduces future risks. From personal experience, updating product recipes or raw material lists makes a difference over time in reducing workplace incidents and environmental headaches. Knowing the specific spots of the chlorine atoms on the benzene ring isn’t just trivia for chemists—it guides decisions that affect worker health and the environment for years to come.
| Names | |
| Preferred IUPAC name | 1,2,4,5-Tetrachlorobenzene |
| Other names |
Tetrachlorobenzene 1,2,4,5-Tetrachlorbenzol |
| Pronunciation | /ˌwʌnˌtuːˌfɔːrˌfaɪv tɛt.rəˌklɔːr.əˈbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 95-94-3 |
| Beilstein Reference | 1740807 |
| ChEBI | CHEBI:34773 |
| ChEMBL | CHEMBL46390 |
| ChemSpider | 10915 |
| DrugBank | DB03270 |
| ECHA InfoCard | 03d238c4-3382-495b-9b50-96d35231ce66 |
| EC Number | 208-406-7 |
| Gmelin Reference | Gmelin 774 |
| KEGG | C14343 |
| MeSH | D014436 |
| PubChem CID | 6846 |
| RTECS number | CZ4495000 |
| UNII | H54VS8O2RD |
| UN number | UN2321 |
| CompTox Dashboard (EPA) | DTXSID2021873 |
| Properties | |
| Chemical formula | C6H2Cl4 |
| Molar mass | 215.88 g/mol |
| Appearance | White crystalline solid |
| Odor | odorless |
| Density | 1.68 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.9 |
| Vapor pressure | 0.013 mmHg (25°C) |
| Acidity (pKa) | Acidity (pKa) of 1,2,4,5-Tetrachlorobenzene is approximately 0.3 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.563 |
| Viscosity | 3.16 mPa·s (25 °C) |
| Dipole moment | 2.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −24.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4416.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AX04 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life with long lasting effects |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H372, H410 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| Flash point | 136°C |
| Autoignition temperature | 430 °C |
| Lethal dose or concentration | LD50 oral rat 759 mg/kg |
| LD50 (median dose) | LD50 (median dose): 7590 mg/kg (oral, rat) |
| NIOSH | SN2975000 |
| PEL (Permissible) | 1 ppm |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Benzene Chlorobenzene 1,2-Dichlorobenzene 1,3-Dichlorobenzene 1,4-Dichlorobenzene 1,2,4-Trichlorobenzene Hexachlorobenzene |

