Looking Closer at 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene: Experience and Direction
Historical Development
Stories about heavily chlorinated cyclodiene compounds always spark debate, and 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene stands out as a telling example of twentieth-century chemical ambition. Scientists first explored chemicals like this in the drive to manage insect pests and fine-tune crop yields. Research ramped up around the post-war years, when breakthroughs in organic chemistry spawned a wave of new insecticides. With these discoveries, hope rose for overcoming food shortages and malaria, and chemists raced to engineer potent solutions. For decades, chlorinated cyclodienes set the pace for crop protection, and their impact bled into both public health and international trade. We can’t talk about this class of compounds without acknowledging the tides of regulation and changing scientific opinion that shifted use over time. Rising concern about environmental persistence led to bans in many countries, so the story here is not just about laboratory success, but about a global rethink of chemical management.
Product Overview
Octachlorohexahydro-methanoindene entered the market as a specialty chemical, with its ancestry rooted in the broader group of cyclodiene insecticides. In practice, this compound once promised improved control of hard-to-treat pests. Its formulation, tied to gains in technical synthesis during the mid-century, played a supporting role in agricultural practice. While it may not match the fame of DDT or Aldrin, this compound shares their chemical fingerprints and a comparable safety debate. The intricate structure allows it to persist in the environment, making its presence felt in soil and water long after intended use. Its original promise in pest control is overshadowed by growing restrictions and attention to environmental health issues.
Physical and Chemical Properties
Chemists admire the architecture of this molecule—chlorine atoms crowding the bicyclic backbone, lending impressive stability and persistence. You don’t get fast breakdown under sunlight or water. Weighing in as a solid under most conditions, it resists most solvents except those chosen by organic chemists. In my experience, working with highly chlorinated compounds like this always calls for proper ventilation because off-gassing at higher temperatures can create toxic vapors. Also, chlorinated species share a tendency to resist bacterial or fungal attack, which unfortunately means residues stick around and bioaccumulate. These properties built the compound’s early reputation for effectiveness and also triggered the eventual alarm bells about environmental and health impacts.
Technical Specifications and Labeling
Chemically precise labeling is non-negotiable in this field. Handling instructions spell out the risks — not just disposal or storage, but guidance on the sort of gloves, respiratory protection, and containment you need. The specs for this compound rely on purity thresholds and impurity limits, acknowledging sensitivity to even trace contamination. Too many stories have circulated among lab workers of headaches or rashes from slip-ups, so manufacturers spell out the need for closed systems and spill kits. Regulatory agencies enforce strict standards for tracking batch numbers and application records, and that has pressed companies to tighten documentation practices across the board, from shipment through end-use.
Preparation Method
Synthesis of heavily chlorinated cyclodiene derivatives like this one relies on multi-step reactions under controlled temperatures, with raw materials that raise risks for everyone involved. Chlorination reactions can run hot, and the hydrogen chloride that comes off as a byproduct demands careful scrubbing and containment. In my hands, scaling up these reactions always increased the stakes — the line between a controlled addition and an overpressure event could be razor thin. Most labs require fume hoods with specialized filtration, and the solvents chosen must tolerate harsh conditions without unwanted side reactions. Special knowledge about reaction exotherms, intermediate stability, and waste handling is what separates safe practice from disaster.
Chemical Reactions and Modifications
People in research often hope for easy tweaks to tailor such compounds for lower risk or greater selectivity, but the chlorinated carbon skeleton resists most efforts at biotransformation or photolytic cleavage. You see minor pathways for dechlorination under strong reducing conditions, but most microbial species can’t touch it without specialized enzymes. Attempts to break down the molecule commonly yield persistent intermediate products, which led to long-lasting problems documented from contaminated sites. In industry, reaction conditions sometimes create byproducts even more persistent than the original, complicating cleanup. Laboratory workers meet frustration trying to dehalogenate selectively, as side-chain or ring cleavage steps can become runaway messes.
Synonyms and Product Names
On the trade front, chemicals like this turn up under a tangled array of names thanks to different regional standards, changes in regulatory status, and shifting naming conventions. This can lead to confusion and risk for anyone tracking chemical inventories or environmental releases. Common synonyms typically reflect historic brand names or trivial names relating to structural features or degree of chlorination. It’s not just a linguistic puzzle; sometimes regulatory checks or international shipments hit snags because registry numbers or legacy trade names don’t match newer labels. Anyone who’s tried filing cross-border paperwork learns the value of keeping a robust chemical synonym database.
Safety and Operational Standards
Working with high-chlorine content organics means following strict operational guidelines, both to protect workers and the public. Personal protective equipment is a baseline, but most real-world incidents stem from poor ventilation, spills, or inadequate decontamination. Older plants sometimes carry residue in pipes or drains, creating a legacy hazard that current operators must clean up. Regulatory audits in this field run deep, demanding not just written plans but demonstration of spill drills and emergency containment. Too many communities face long-term cleanup bills after leaks or illegal dumping, and courts push the burden onto responsible parties. For lab-scale operations, eye washes and backup ventilation earn their place through lived experience — too many labs learned the hard way from exposure incidents that made it into the local news.
Application Area
Use cases for this molecule started with farm pest control, especially for soil-borne insects not tackled by earlier insecticides. Its low solubility meant it stayed put after application, but that same feature laid the groundwork for groundwater contamination in vulnerable areas. Later, some efforts aimed at controlling termites and public health pests followed similar chemical rationale. Over time, laws changed and risk assessments flagged unacceptable residue levels in produce and animal products. Its limited approved uses now focus on closed environments, research, or historic cleanup, not mainstream application. Regions still wrestling with legacy contamination see regulators, farmers, and environmental advocates at odds about responsibility and remediation technique. Nobody wants to inherit a polluted field or catchment, and the debates drag on.
Research and Development
Chemical research has drifted from making new cyclodiene analogs for mainstream use, and now most lab focus pivots on mitigation and detection. Advanced chromatography and mass spectrometry enable labs to chart the spread of these compounds in soil, rivers, and biota. Some teams hunt for microbial strains with novel enzyme pathways that might speed breakdown, and pilot trials test bioremediation in contaminated wetlands or agricultural land. Others build computer models that chart exposure routes to fine-tune public health intervention, and regulatory labs measure residue limits with far more precision than anything possible decades ago. Policy drives funding toward safe alternatives with lower persistence, pushing chemical innovation toward radically different structures, not just tweaking the old ones. Real-world results depend on buy-in from farmers, regulators, and downstream food buyers who all carry scars from earlier oversights and scandals.
Toxicity Research
Early toxicology studies on octachloro-methanoindene and its relatives revealed harmful effects in lab animals at levels achievable through environmental exposure, touching off waves of regulatory action. Chronic exposure can build up in fatty tissues, and some breakdown products turn out even nastier than the parent compound. Evidence ties long-term, low-level exposure to neurological symptoms, developmental delays, and elevated cancer risks in sensitive groups. These research findings didn’t stay academic—the stories of real people dealing with chemical exposure shaped public sentiment in ways chemical companies could not ignore. Mobile testing units, biomonitoring in exposed communities, and legal battles over contaminated food made headlines. Health agencies now track exposure through blood and fat tissue sampling for at-risk populations. The legacy of slow breakdown drives long-term studies, including transgenerational effects, and unfortunately the answers are not always quick or reassuring.
Future Prospects
Current thinking aims to both contain the environmental legacy and push chemistry away from persistent bioaccumulative toxins. Major R&D hubs fund research on chemical substitutes for pest control, especially those breaking down rapidly after serving their purpose. There’s a parallel push for broader adoption of integrated pest management and biological control, so reliance on hard-to-degrade chemicals drops year by year. In my mind, the real challenge won’t come only from new chemistry, but from repairing trust between science, industry, and affected communities. Knowledge about risks travels faster today and calls for transparency in research, open reporting of incidents, and genuine dialogue with the people living downstream from fields or plants. For chemists trained to make molecules, this shift to accountability and dialogue cuts across every level of the field, rippling into how we teach, investigate, and regulate. The future trajectory of this chemical, and its cousins, depends not just on clever synthesis or remediation, but on building systems that put shared health at the center of every step.
What are the main uses of 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene?
A Powerful Legacy in Pest Control
There’s no ignoring the mark that 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene, better known as Chlordane, made in agriculture. Used heavily from the late 1940s up until the 1980s, this compound changed the way people controlled termites and soil-borne insects. Ask anyone involved in post-war homebuilding or farming, and they’ll remember barrels of this stuff being mixed for long-lasting soil treatments. Chlordane stuck around in the environment, which meant one application could last for more than a decade in the right soil. That kind of persistent power sounds impressive on paper, and it led to widespread use beneath homes, around crops, and even in public spaces.
Household Use Shaped Suburbia
Look under a house built before the late 1980s, and there’s a good chance some Chlordane lingers in the soil. In its heyday, it formed a chemical barrier against termites, which can cause structural damage that costs thousands to repair. Many families felt safer knowing their investment was protected by something biting insects couldn’t eat through. The same compound helped prevent crop loss from rootworms and cutworms, giving growers bigger harvests in corn, potatoes, and citrus. I’ve spoken with older extension agents who remember it making the difference between a failed and a bumper year.
Public Health Risks and Global Ban
The downside comes from that same staying power. Chlordane doesn’t break down quickly, and over the years it built up in soil, water, and even in our bodies. Studies linked the compound to nervous system problems, increased cancer risks, damage to the liver, and harm to the immune system. The Environmental Protection Agency finally banned its use in 1988 following mounting evidence of long-term health effects. By then, traces of Chlordane had shown up in wildlife, including fish and birds, far from where it was ever sprayed. Older neighborhoods still see traces of it decades later.
Pushing for Safer Alternatives
The story of Chlordane brought lessons that shaped modern pest management rules. Now, everyone from pest control pros to homeowners faces tighter regulations on what goes into the ground. Alternatives like bait systems and targeted, short-lived chemicals have taken center stage. Government agencies push for integrated pest management, combining smart sanitation, building repairs, and only spot treatments when absolutely needed. There’s also a growing effort to track persistent organic pollutants in soil and water, making sure communities spot trouble before it builds up.
Cleaning Up the Mess
Dealing with Chlordane’s leftovers means regular testing in old homes, especially where kids might play in the dirt. Some people need professional soil removal or sealing crawl spaces to cut off exposure. Today, remediation often involves removing and disposing of contaminated soil at special facilities. Outreach programs try to warn homeowners about the hidden risks in aging neighborhoods and teach safer management around legacy contamination.
Looking Forward
Chlordane taught a hard lesson. Quick fixes in pest control can backfire, affecting generations who never chose to use these products. The focus has shifted to balance—protecting crops and homes, but not at the cost of human health or polluted landscapes. Each innovation in pest management draws on that long, complicated history, trying to keep the gains in food security and safety while avoiding the old mistakes.
Is 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene toxic to humans or animals?
Looking at the Risks Linked to Chlorinated Hydrocarbons
For years, scientists and regulatory bodies have been digging deep into the health and environmental effects of complex compounds like 1,2,4,5,6,7,8,8-octachloro-2,3,3a,4,7,7a-hexahydro-4,7-methanoindene. The name is a mouthful, but the real issue comes down to what this chemical does inside the bodies of animals and people.
What Do We Know So Far?
This chemical often pops up as a byproduct or ingredient in some industrial pesticides, especially those from a group called organochlorines. Decades of research into organochlorines provide a warning: these chemicals tend to stick around in the environment and build up in living tissue. You don’t have to look further than the story of DDT to see the harm—bioaccumulation in fish led to more toxic birds, and local food chains destabilized.
There is limited, direct human toxicity data on 1,2,4,5,6,7,8,8-octachloro-2,3,3a,4,7,7a-hexahydro-4,7-methanoindene, but we know similar chlorinated hydrocarbons cause liver, nervous system, and reproductive problems in animals. Mice and rats exposed to organochlorines show tremors, weight loss, and, at higher doses, death. In people, even exposure to small amounts over a long time can trigger neurological issues or disrupt hormones.
Why Experience Illustrates the Problem
Living near farmland during my teenage years, I saw first-hand how pesticides drifted into water and soil, impacting both the wildlife and the people in the area. Friends who fished brought in stunted or deformed catches some seasons. Conversations with local farmers often circled back to new pesticide formulas and growing cancer rates. Studies from the CDC and WHO confirm this kind of pattern in rural American communities, where organochlorine pollution has correlated with higher risks of non-Hodgkin lymphoma and childhood leukemia.
Tackling the Safety Issue
There’s good reason regulatory agencies demand deep study before allowing such chemicals near crops, water, or communities. Agencies in the US and Europe have banned or tightly restricted many organochlorines. In places where rules run lax, old stockpiles and black-market sales keep these toxins in circulation, sometimes with devastating effect. Animal studies from the EPA reveal birth defects and immune system problems after even short-term exposure.
Disposal of unused chemicals needs more oversight, as hundreds of old storage barrels leak into groundwater every year. Filtering out these persistent chemicals at treatment plants is tough; the molecules stick in fat, resist natural breakdown, and make their way through food webs from plankton to people. Monitoring fish, milk, and water for chlorinated compound residues helps flag risks early, but it demands consistent funding and public attention.
What’s Left Unanswered
Gaps in toxicology make risk assessment tricky. No agency has set firm safe exposure thresholds for this particular chemical. Governments should support new research using sensitive tests—watching for nervous system, thyroid, and fertility changes in exposed populations. Public pressure works: as awareness of DDT grew in the ‘70s, policy shifted fast. The same can happen with any chemical when information is clear and accessible.
There are safer ways to manage pests and protect crops—rotation, biopesticides, and stricter residue checks at borders. Community-driven reporting and open access to local contamination data matter, too. Empowered people ask better questions and push for clean surroundings, whether in big cities or down rural lanes.
What safety precautions should be taken when handling this product?
Respecting the Risks Each Step of the Way
Trust me, there’s no shortcut when you open up a drum or container of chemicals or industrial product. Even if you’ve done it a thousand times, all it takes is one lazy move to turn a routine day upside down. Years ago, I shrugged off the importance of reading the safety sheet. After one close call with a splash that landed way too close to my eye, I started following procedures to the letter.
Personal Protective Gear Is Non-Negotiable
Gloves, goggles, and a decent pair of boots should stay within arm’s reach. That splash I mentioned? If my face shield had been sitting on the cart, things could have gone sideways. Relying on memory for what a product “might” do leaves too much to chance. Labels spell out if fumes could irritate lungs, skin, or worse. More than one worker I know ended a shift in the emergency room because they treated a mask or gloves as optional.
Ventilation Keeps You Breathing Easy
Some products throw off fumes you won’t always smell or see. Over time, even small doses can chip away at your health. The right fan setup or an open window takes a few minutes to arrange, but lungs last a lifetime. Industrial hygiene studies back this up—consistent ventilation in confined spaces slices the risk of accidents drastically. That’s something no one can ignore in good faith.
Don't Wing It—Know the Emergency Plan
Honestly, no manual or label prepares you for real panic. Once, a coworker spilled solvent on her pants. Without hesitation, she sprinted to the safety shower, just as the calendar drill taught us. Protocols exist for every product, from acids to cleaning agents, because minutes count. I stay aware of eye wash locations and exits. Every shop or plant manager should run surprise drills until grabbing the right extinguisher or rinsing a burn feels like muscle memory.
Storage Isn’t About Just Stacking Drums on a Shelf
It’s tempting to set everything down wherever there’s space, especially after a long day. In one summer job, I watched a minor leak run under the break table due to careless stacking. That mistake led to an expensive cleanup and a handful of hospital visits. Incompatible materials spark fires and explosions more often than you’d think. Color-coded bins and locked cabinets do more than look organized—they keep people alive. Regulatory compliance alone isn’t enough; you’ve got to think ahead and keep touch-points clean and obvious.
Training Pays Off in Ways You Might Not Expect
Reading this, you can probably recall a story where someone thought their experience meant they could skip basic steps. Regular, no-nonsense safety training reminds folks to stay sharp. The Occupational Safety and Health Administration (OSHA) found that ongoing instruction drops incident rates by more than 20%. Mistakes shrink when workers share lessons learned from near misses and not just from manuals.
Nothing Beats Paying Attention
No checklist can replace looking, listening, and staying present. Before using any new product, I inspect my gear. I check the area for clutter or spills, and ask if anyone’s working with something volatile nearby. Practicing mindfulness every shift beats relying on luck. In my experience, everyone on the team benefits when vigilance becomes part of the job, not just a reaction after things go wrong.
How should 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene be stored and disposed of?
What Storage Says About Hazard Awareness
Storing a chemical with a name this long—some know it by its trade name, chlordane—has never felt routine. Few things in the lab or field convey greater risk than a jug marked “octachloro...” and a skull-and-crossbones. Tossing containers in any lockable closet isn’t enough. Storage calls for a dedicated, sealed chemical cabinet, best set aside for halogenated pesticides, away from sunlight, water, food, feeds, animal quarters, and human traffic. I keep such compounds double-contained, with clear secondary labeling, inside acid- and corrosion-resistant shelving. The point isn’t just compliance—it’s about making sure nobody touches this stuff without realizing what they’re up against.
Personal Experience in Handling the Risks
Working with persistent organochlorines is a good way to learn what precaution looks like in practice. This chemical clings to gear, skin, and containers. No shortcuts: gloves, goggles, well-fitted respirator if there’s dust or vapor in the air, and thorough washing afterward. Cross-contamination is easy—a single unnoticed smear can linger and spread, so careful decontamination of work areas and tools after each use comes before anything else. Do not eat or drink in the handling area. These substances cause harm through chronic exposure as much as through a single spill, so I enforce zero-tolerance for guessing or skipping a step.
Why This Chemical Raises Red Flags
1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene is no ordinary hazardous waste. It lingers in soil and water, resists breakdown, accumulates in living tissue, and has known links to cancer and nervous system injury. Chlordane built its reputation as a super-effective termite and crop pesticide; it earned its ban in dozens of countries due to health risks and persistence in the environment. I’ve reviewed studies showing how traces show up decades after application, sometimes drifting well beyond the place of use.
Disposal: More Than Just Throwing It Away
Pouring leftovers down the drain or tossing containers in the trash has spelled disaster countless times. Proper disposal takes coordinating with a licensed, certified hazardous waste facility. These places incinerate material at high temperatures—high enough to destroy the persistent molecules. I worked years ago with a crew cleaning up a pesticide warehouse. Only trained hazmat workers wearing full protective suits canvassed the site and packed waste into DOT-approved drums for sealed transport. Documentation tracked every batch from storage shelf to final incineration.
Community and Environmental Responsibility
No point in pretending chlordane’s dangers are someone else’s problem. Regulations exist regardless of scale: household, farm, lab, or industrial. If leftovers are found in old cans or soils, local environmental authorities should be contacted before any action. People living near dumpsites or farm fields should demand clear information about ongoing monitoring, cleanups, or remediation efforts. Local governments and environmental groups sometimes offer “take-back” days that collect hazardous waste free of charge, which provides an accessible solution for individuals not equipped to handle disposal solo.
Supporting Safe Practices
Culture matters as much as compliance. Training, clear signage, and routine checks foster vigilance. Supervisors and peers set the example—there’s no room for sloppiness. Reviewing chemical inventories regularly helps catch forgotten stocks before leaks or legal problems emerge. Responsible storage and disposal save lives, time, and money in the long run. Each person with access to hazardous materials plays a part in keeping poisons out of air, soil, water, and our collective future.
What are the environmental impacts of 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene?
Understanding a Persistent Organic Pollutant
Pesticides always bring a hard choice—manage pests or risk health and environment. This oddly named compound, 1,2,4,5,6,7,8,8-Octachloro-2,3,3A,4,7,7A-Hexahydro-4,7-Methanoindene, steps far beyond most garden chemicals. Also known in some circles as Chlordane, it’s one of those chemicals that clings to soil and sneaks through food chains. Its chemistry makes it tough, sticking around for years, sometimes decades, once it gets released.
How It Behaves in Nature
Soils exposed to this chemical lock it in, but wind, water, or even earthworms can help it move. I’ve seen research showing that even after fields get abandoned or regulated, the stuff lingers deep in the earth. Plants draw up traces. Rain can wash residues into streams. Small animals eat contaminated bugs or plants, and then birds or mammals go after them. Each step, concentrations build. You can read government reports listing Chlordane in rivers, in fish from backyard ponds, and in food that travels halfway across the world.
Impact on Wildlife and People
Toxicity brings no mercy. Insects targeted by old-school pest controls containing this chemical die quick, but the disaster doesn’t end there. Birds snatch those dead bugs and can struggle with reproduction over time. Frogs and fish stumble into polluted waters, and populations falter. Those toxins stick in tissues, meaning predators higher up the chain get the worst dose.
People get exposed from many directions—food grown in contaminated soil, dust stirred up from treated home foundations, old stockpiles gone unnoticed. There’s scientific evidence pointing to nervous system effects, potential cancer risks, and liver problems after long-term exposure. For communities near former application sites, it becomes a real worry, especially for families growing vegetables or catching fish locally.
Why This Problem Sticks Around
Government bans and restrictions hit Chlordane hard in the late 1980s in places like the United States and much of Europe. Still, a ban doesn’t mean the story ends. Once released, persistent organochlorines don’t vanish. Some countries continued limited uses for years, or improper disposal has left stockpiles leaking into the environment. International trade sometimes moves food or goods with trace residues.
Facing Up to the Solution
Cleaning up chlorinated pollutants like this one takes muscle, both technical and political. Soil removal sometimes becomes the only option, especially near playgrounds or water sources. More often, people rely on covering old sites or strict land-use controls, trying to keep exposure low. A smarter move starts with prevention: some countries have adopted mandatory monitoring, catchment programs for old stocks, and educational campaigns in rural communities.
Better farming practices, less reliant on persistent chemicals, need more support and funding. Farmers swapping to integrated pest management find healthier soils and fewer headaches with persistent toxins. On the consumer side, washing produce and checking fish advisories helps cut the risk, but long-term safety only comes from cleaning land and strengthening bans on import or manufacture.
Nobody should underestimate the reach of a persistent chemical. Chlordane makes a strong case for careful regulation, honest public health messaging, and investment in safer alternatives for today and tomorrow. Only then can communities breathe easier—literally and figuratively.
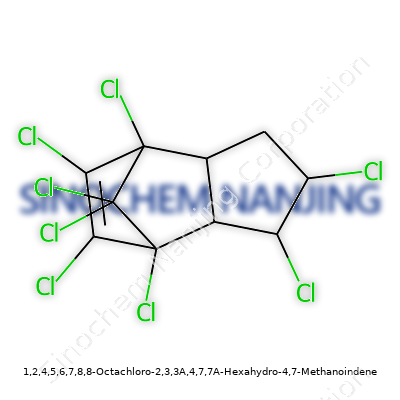
| Names | |
| Preferred IUPAC name | 1,2,4,5,6,7,8,8-octachloro-3a,4,7,7a-tetrahydro-4,7-methano-1H-indene |
| Other names |
Chlordane Octachlor CAS 57-74-9 Velsicol 1068 Chlordan Corodan Kepone Octachlorethane |
| Pronunciation | /ˌoʊ.təˈklɔːr.oʊ ˌhɛk.səˈhaɪ.droʊ ˌmɛθ.ə.noʊˈɪnˌdiːn/ |
| Identifiers | |
| CAS Number | 2385-85-5 |
| 3D model (JSmol) | `3D model (JSmol): C1(C2C(C3C(C1Cl)Cl)(C(C2Cl)Cl)C(=C(C3Cl)Cl)Cl)Cl` |
| Beilstein Reference | 1462301 |
| ChEBI | CHEBI:34623 |
| ChEMBL | CHEMBL2192103 |
| ChemSpider | 14118 |
| DrugBank | DB11185 |
| ECHA InfoCard | 100.004.921 |
| EC Number | 207-439-0 |
| Gmelin Reference | 84338 |
| KEGG | C19615 |
| MeSH | D002922 |
| PubChem CID | 5364980 |
| RTECS number | GV9810000 |
| UNII | 9L1HB15Z4O |
| UN number | UN2761 |
| CompTox Dashboard (EPA) | DTXSID5020729 |
| Properties | |
| Chemical formula | C10H6Cl8 |
| Molar mass | 408.828 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.99 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.88 |
| Vapor pressure | 1.5E-7 mm Hg at 25 °C |
| Acidity (pKa) | 12.11 |
| Magnetic susceptibility (χ) | -0.000085 |
| Refractive index (nD) | 1.629 |
| Viscosity | Viscosity (dynamic, 25 °C): 4.24 mPa·s |
| Dipole moment | 2.67 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 377.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -290.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4063 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Pesticides |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. Suspected of causing cancer. Very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H301, H331, H373, H410 |
| Precautionary statements | P261, P273, P280, P301+P312, P305+P351+P338, P308+P313 |
| NFPA 704 (fire diamond) | 2-4-0 |
| Flash point | Flash point: > 110°C (closed cup) |
| Lethal dose or concentration | LD50 oral rat 100mg/kg |
| LD50 (median dose) | Oral-rat LD50: 25 mg/kg |
| NIOSH | GN1400000 |
| PEL (Permissible) | 0.5 mg/m³ |
| REL (Recommended) | 0.1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 2 mg/m3 |
| Related compounds | |
| Related compounds |
Chlordene Heptachlor Aldrin Dieldrin Endrin Chlordecone Lindane Methoxychlor |

