1,2,3-Trimethylbenzene: An Insightful Look into a Widely Used Aromatic Compound
Historical Development
1,2,3-Trimethylbenzene has played a notable role in the story of synthetic chemistry and industrial progress. Dating back to the earliest days of organic chemistry, aromatic hydrocarbons took center stage as chemists puzzled out molecular structures from coal tar distillates. Over time, the understanding and purposeful synthesis of 1,2,3-trimethylbenzene grew. Centuries ago, isolation depended on the labor of distillation and crystallization. By the twentieth century, refineries started producing this compound in significant quantities, harnessing advanced catalytic reforming of petroleum fractions. Instead of being a mere curiosity, the substance moved firmly into the camp of chemical building blocks for countless products that define daily life, spanning coatings, solvents, and lubricants.
Product Overview
1,2,3-Trimethylbenzene—some may know it as hemimellitene—provides more than just a mouthful for chemists. This is an aromatic hydrocarbon with a distinct three-methyl grouping on a six-membered benzene ring. That arrangement shapes molecular reactivity and solubility, setting it apart from its isomers. 1,2,3-trimethylbenzene frequently arrives as a clear, colorless, oily liquid, rarely calling attention to itself except for its slightly sweet, gasoline-like odor. Its humble outward appearance hides a backbone of chemical stability and the ability to act as a launchpad for making more challenging molecules. Anyone who has worked in an industrial or laboratory setting has probably crossed paths with it, perhaps unknowingly—blended into paint thinners or fuel additives or acting as an intermediate in surprising applications.
Physical & Chemical Properties
Under normal conditions, 1,2,3-trimethylbenzene shows a boiling point around 176 °C and a melting point hovering just above room temperature. It mixes poorly with water but readily with organic solvents, forming azeotropes in some cases, a trait familiar to chemists looking to separate compounds. That tendency to dissolve organic material drives its role as a solvent. Its vapor, much like other hydrocarbons, is heavier than air and can travel along surfaces to ignition sources—a real concern for handling and storage in industrial settings. Its flashpoint, vapor pressure, and reactivity mirror family members like mesitylene but subtle differences in the methyl distribution lead to small tweaks in physical constants, which influence how technicians use and store large volumes.
Technical Specifications & Labeling
In regulated production, strict technical standards govern what ends up in the drum or tank. For example, most industrial suppliers tout purity measured above 98 percent, with documentation on moisture, sulfur content, and trace organics. Packaging varies; steel drums and tank trucks dominate in bulk handling. Labeling speaks the language of hazard communication, marking flammability, environmental risk, and health danger. Storage warnings stress a cool, ventilated space away from oxidants or sources of ignition. These practices stem not only from regulatory compliance but also from lessons hard-earned—spills and vapors can pose environmental and safety headaches. Careful labeling and specification reporting support the flow of information from manufacturer to user, enabling traceability.
Preparation Method
Refining facilities extract 1,2,3-trimethylbenzene using modern catalytic reformers, which boost the aromatic content in naphtha streams. Catalysts like platinum or rhenium on alumina rearrange hydrocarbon skeletons to generate more valuable rings with desired side groups. Fractional distillation methods isolate the isomer from similar compounds. Synthetic routes also start from toluene or xylene by methylation, feeding methyl groups into the benzene ring under controlled heat and pressure. None of these steps happen haphazardly. Chemical engineers and plant operators tune parameters to favor high yields and minimize troublesome residues or byproducts. This focus on efficiency springs from high demand, cost concerns, and a commitment to sustainability.
Chemical Reactions & Modifications
The story doesn’t end with bulk production. The molecule’s three methyl groups and aromatic core make it an inviting starting point for further transformations. Nitration can attach nitro groups, preparing it for subsequent reductions into amines. Chlorination or bromination locate halogens at specific ring positions, unlocking new intermediates used in dyes and pharmaceuticals. Gentle oxidation targets methyl groups, which can generate carboxylic acid derivatives. Through Friedel–Crafts alkylation or acylation, more complex aromatic structures emerge, often with commercial significance. The balance between stability and reactivity fosters its utility in labs and factories. Skilled chemists appreciate how the position of methyl groups affects the resulting product profile, giving them a tool for selectivity.
Synonyms & Product Names
Common lexicons in chemical supply distinguish 1,2,3-trimethylbenzene as hemimellitene, reflecting traditional nomenclature habits. Sometimes catalogues use the descriptor “trimethylbenzene,” specifying "1,2,3-" for clarity, particularly important given presence of similar isomers like pseudocumene (1,2,4-trimethylbenzene) and mesitylene (1,3,5-trimethylbenzene). This proliferation of names can add confusion, particularly for those new to industrial chemistry. Documentation and inventory systems must clarify structures to prevent accidental misapplication, an all-too-common pitfall that can lead to costly delays or wasted materials. Transparent naming supports a culture of safe, precise work in labs and warehouses.
Safety & Operational Standards
Handling 1,2,3-trimethylbenzene requires a steady hand and attention to detail. Flammability stands out among the safety concerns. The vapor can form explosive mixtures with air at relatively low concentrations. Workers in chemical plants quickly learn to respect the risks, favoring closed systems and using proper grounding to prevent static discharge. Prolonged inhalation may lead to headaches, dizziness, and—if limits are exceeded—central nervous system effects. Safety data sheets highlight the value of respiratory protection, localized exhaust ventilation, and protective gloves or goggles. Emergency planners account for spill control and fire suppression, since hydrocarbon blazes bring unique hazards compared to other industrial fires. Regulations set permissible exposure limits, and monitoring technology gives real-time measurements in critical zones. Over the years, a culture of safety-mindedness has kept incidents to a minimum, but complacency can creep in, so ongoing training keeps everyone sharp.
Application Area
1,2,3-Trimethylbenzene finds its way into more products than most people realize. It dissolves resins and pigments in paints, improving flow and finish. Coatings and varnishes depend on it for smooth application, rapid drying, and desired gloss. The compound also serves oil refineries and petrochemical plants as an antiknock agent added to gasoline, supporting cleaner combustion and improved engine efficiency. In lab settings, analytical chemists use it as a calibration standard or as a solvent in chromatographic techniques. Manufacturers of adhesion promoters, specialty lubricants, and plasticizers trust it to deliver consistent chemical behavior batch after batch. The wide reach results in steady demand, and each usage scenario brings its own handling, recovery, and recycling challenges.
Research & Development
Innovation rarely slows down in chemical industries. Scientists investigate greener and more selective production methods, driven by tightening environmental standards and consumer expectations. Cleaner catalysts, improved recycling, and novel extraction techniques all attract research. Computational chemistry and process modeling allow fine-tuning of yields and identification of potential hazards before scaling up. In universities, organic chemists continue to probe aromatic reactivity, uncovering untapped valorization routes for 1,2,3-trimethylbenzene. The continuing challenge is to maximize value from each molecule while keeping waste generation in check and energy consumption low. Many hope that data-driven process control and intensified reaction engineering can push performance even higher in the years ahead.
Toxicity Research
Concerns about environmental and human health effects spark ongoing study of 1,2,3-trimethylbenzene. Animal testing reveals potential for acute and chronic toxicity at high exposures, including possible impacts on kidneys, liver, and central nervous system. Most evidence suggests that normal industrial and consumer exposure remains far below those danger thresholds, but vigilance never wanes. Research tracks atmospheric breakdown, bioaccumulation, and degradability to predict environmental persistence. Workplace measurements guide occupational health professionals, who tailor interventions to match measured risk. Toxicologists keep watch for subtle effects, such as allergenic reactions or long-term low-level impacts, building a clearer picture with each new study. This body of work shapes not just regulatory action, but also guides material substitutions and process redesigns.
Future Prospects
Looking ahead, the fate of 1,2,3-trimethylbenzene reflects larger shifts in energy, environment, and materials science. Demand for coatings, specialty fuels, and advanced materials isn’t going away, so efficient production will remain vital. Yet, expectations for clean air, climate responsibility, and circular economy push innovation to further reduce emissions or develop biodegradable alternatives. In the lab, scientists explore bio-based feedstocks and engineered catalysts with selective reactivity, hoping to lessen the dependence on fossil resources. Regulatory agencies seek ever-better monitoring and exposure control, while industry responds with better personal protective equipment and containment technology. The combined force of chemical engineering, business incentives, and environmental stewardship will continue shaping this compound’s story, reflecting both its utility and responsibilities.
What are the main uses of 1,2,3-Trimethylbenzene?
Industrial Workhorse
Walk into any chemical manufacturing plant, and you’ll spot tanks marked for specialty solvents. 1,2,3-Trimethylbenzene, also called hemimellitene, fills some of those tanks. It helps to dissolve and blend substances that wouldn’t normally mix. Factories use it when they need something a bit more punchy than your average solvent. Chemicals need to break down quickly and evenly, especially in paint and resin manufacturing. Using a reliable aromatic hydrocarbon like this one keeps manufacturing lines running smoothly, saves money, and cuts down on waste.
Lifting Research and Testing
Laboratories value consistency and predictability. 1,2,3-Trimethylbenzene delivers that. Whether researchers are studying fuel performance or testing new lubricants, they need reference materials with known boiling points and chemical resistance. This compound fits the bill. It shows up in analytical chemistry for calibrating equipment, which keeps machines honest during testing. If you’re wondering whether your gasoline mix meets the mark, you can bet this chemical played a part in making sure the answer is accurate.
Link to Fuels and Energy
I’ve spilled more than a few drops of fuel while tinkering with engines. Turns out, 1,2,3-Trimethylbenzene helps improve the performance of gasoline. Refineries add it to help boost octane ratings, squeezing a little more energy out of each drop. That’s good news for drivers looking for cleaner combustion and fewer knocking noises from under the hood. It isn’t thrown in randomly; refiners test every additive to balance performance with safety standards. The Environmental Protection Agency keeps a close eye on it, checking emissions to guard public health.
Role in Coatings and Paints
Paints seem simple until you see how quickly a bad batch can lead to jobs gone wrong or warranty claims. 1,2,3-Trimethylbenzene matters here. Paint companies use it to control flow and drying times. Too fast and the finish gets ruined, too slow and costs go up. In the hands of a skilled formulator, this compound helps make surfaces shine, reduces imperfections, and keeps maintenance crews happy. Quality coatings stay bright and tough, reducing the churn of repainting fences, bridges, or vehicles.
Personal Safety and Environmental Questions
Handling chemicals like 1,2,3-Trimethylbenzene calls for attention. Wearing rubber gloves, goggles, and a mask isn’t just a box to check. I’ve seen colleagues learn the hard way that skin contact or breathing in fumes makes for a bad day. Chronic exposure may cause headaches or worse. It’s one reason regulatory agencies set strict workplace limits and recommend plenty of ventilation. Most companies now work hard to recycle or control emissions, but legacy pollution still needs cleanup. Moving forward, the solution depends on strong monitoring, good training for workers, and investment in greener alternatives whenever possible.
Why These Uses Matter
In daily life, few folks talk about 1,2,3-Trimethylbenzene. Yet, it sits behind better engines, reliable paints, and safer lab results. As long as manufacturing, research, and transportation need robust performance, chemicals like this will stay in demand. Balancing utility with health and environmental concerns—through science, transparent regulation, and honest communication—keeps progress responsible. Every decision along the way shapes how safe and effective these substances remain for the next generation.
What are the health and safety hazards of 1,2,3-Trimethylbenzene?
Understanding Where Exposure Can Happen
Plenty of folks run into 1,2,3-trimethylbenzene not at a science lab, but in day-to-day industries. The compound shows up in paints, solvents, some fuels, and even near busy highways. I’ve seen workers in auto shops or factory settings sneeze and rub their eyes after handling chemicals without realizing a real risk goes beyond simple irritation.
How It Affects the Body
Most people don’t expect their job to make them feel off, let alone sick, but breathing in vapors or getting liquid on your skin can bring on more than a mild headache. Scientific literature links high-level inhalation to dizziness, fatigue, and even confusion — not a great look for anyone operating heavy equipment. Skin contact may lead to redness or rashes. Hospitals sometimes see workers come in with these symptoms, only to trace things back to inadequate protection during handling of degreasers or thinners containing this chemical.
Long-Term Health Considerations
Short-term discomfort causes worry, but people in regular contact, like maintenance crews, end up with repeated, low-level exposures. Over time, this can mess with the nervous system, especially if proper ventilation isn’t part of the work environment. Some research has suggested impacts on the liver and kidneys, though main concern centers on the lungs and the brain. Anyone regularly exposed really ought to think of this as a risk worth addressing, not just a workplace nuisance.
Fire and Environmental Hazards
This chemical catches fire easily. Mixing fumes with a spark—sometimes just a static discharge from clothing—has set off dangerous situations. Firefighters know that industrial fires involving aromatic solvents move fast and create toxic smoke, so no one should feel comfortable storing this stuff near ignition sources. Runoff or spills can damage soil and water. Municipal water supplies sometimes test for solvents that seep in due to accidents. Fish and mammals respond badly, so small community lakes and water tables could end up harmed for years.
Making Workplaces Safer
In my experience, the best way to keep people safe starts before any mixing or spraying begins. Employers ought to train staff on leak detection and provide gloves plus reliable respirators — not flimsy dust masks. Good exhaust fans and open bay doors reduce vapor buildup. OSHA has guidelines for safe air quality, and regular monitoring shows gaps in real protection.
Switching to less hazardous alternatives makes sense sometimes, especially if the work site deals with kids, food production, or sensitive electronics nearby. Proper storage — well-labeled, with spill kits available and fire extinguishers close — brings emergencies under control quickly if things go south.
What People Should Do
If headaches, nausea, or skin issues show up after a shift, no one should shrug it off as “just part of the job.” Healthcare providers need to know what chemicals were in the mix. Reporting symptoms and small leaks prevents big disasters. Community residents should speak up if they notice strong odors near storage tanks or dumping grounds. State health departments run hotlines for these concerns and can bring out inspectors.
Final Thoughts
Living and working around chemicals means staying alert to dangers that don’t announce themselves loudly. Safety isn’t about following rules for the sake of rules; it’s about keeping families and communities from facing risks they never signed up for.
How should 1,2,3-Trimethylbenzene be stored and handled?
An Everyday Chemical Few Have Heard Of
Chances are, most people haven’t come across 1,2,3-Trimethylbenzene outside a chemistry lab. This solvent serves a quiet purpose in making coatings, dyes, and as an intermediate for other chemicals. Its clear, flammable liquid form tells anyone used to working with solvents: treat it with respect. Coming from a background in environmental safety, I’ve seen the difference between close calls and real accidents with substances like this.
Serious Fire Hazards
This chemical catches fire easily. Vapors spread quickly and can ignite from static electricity. Local fire departments almost always stress three things for flammable liquids: keep away from open flames, store in tightly closed containers, and use only in areas with good ventilation. Even a forgotten pilot light counts as a danger point. Fire codes recommend using flame-proof storage cabinets, preferably outside main workspaces, especially if people are nearby. One metal drum in an out-of-the-way room has prevented more headaches and late-night emergencies than any policy memo.
Health Risks You Can’t Ignore
Absorbing 1,2,3-Trimethylbenzene through lungs, nose, or skin can cause dizziness, headaches, and irritation—worse if someone already deals with asthma or allergies. Gloves made from nitrile or neoprene help, and protective goggles cut down on the risk of eye splashes. People who pour, pump, or transfer this chemical should remember a simple trick: get in the habit of checking their gloves before every shift. Too many times, someone grabbed the closest pair, not realizing small tears defeat the whole purpose.
Ventilation and Good Habits
Air circulation ranks at the top of any practical safety list. Fume hoods or local exhaust fans at the spot where chemicals get used stop vapors from building up. Someone in my field once said, “If you can smell anything, your setup needs adjusting.” Air monitoring devices help, but so do old-school methods like opening doors and running fans. Limit the time you spend with open containers by planning out your work and keeping clean-ups quick and simple. Simple routines mean people are less likely to rush or lose focus, even on busy days.
Disposal and Small Spills—No Shortcuts Allowed
No drain or trash bin can handle cleanup for 1,2,3-Trimethylbenzene. Liquid waste goes into labeled metal or plastic containers with screw-on lids. Signage and sturdy secondary containment—solid tubs or trays—catch leaks before anyone tracks the chemical around the shop. One overlooked splash can travel on shoes or gloves, causing risk in break rooms or hallways later.
Training, Labels, and Culture
Regulations from OSHA point out that chemical storage only works well if everyone knows what’s in every bottle and drum. Clear, weatherproof labels and Safety Data Sheets in plain sight are essentials, not extras. Annual safety drills, hands-on fire extinguisher training, and honest conversations about “near-misses” build a culture where everyone takes care of each other. In my experience, a bit of peer pressure—backed by real stories about what went wrong—works better than the thickest manual.
Small Steps Add Up
Treating chemicals like 1,2,3-Trimethylbenzene with common-sense precautions, regular reminders, and teamwork makes safety routine, not just a line on a checklist. Real protection comes from the everyday choices of everyone on the floor, not some hidden policy tucked away in an office.
What is the chemical structure and formula of 1,2,3-Trimethylbenzene?
The Structure That Shapes 1,2,3-Trimethylbenzene
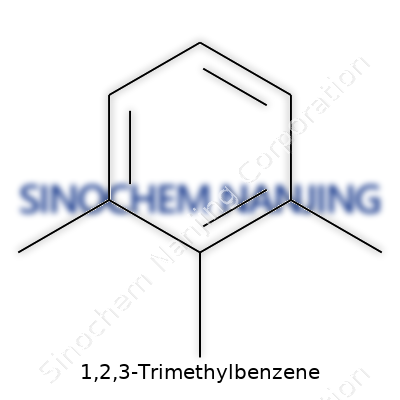
Picture a benzene ring, which chemists recognize as a hexagonal loop of carbon atoms. Three methyl groups branch off this core, claiming the 1, 2, and 3 positions. Draw it and you'll see those methyl (–CH3) groups lined up on the ring, side by side. The rest of the ring holds on to hydrogen atoms, balancing the formula to C9H12.
Why Structure and Formula Matter Here
The way those methyl groups attach themselves matters a lot more than it seems. Three together influences both the shape and the behavior of the molecule. In my first year in the lab, I watched as small changes in structure totally altered smell, boiling point, and even the way these chemicals would dissolve. Chemical structure isn’t just something that fills a textbook. It guides which industries pick up a material and how it’ll react down the line.
1,2,3-Trimethylbenzene claims a spot with the formula C9H12 and is known sometimes as hemimellitene. Physically, the presence of three methyls tightly grouped means this aromatic hydrocarbon doesn’t just act like other benzene derivatives. Its structure gives it a higher boiling point (about 175°C), a sweet odor, and a fuel-like nature that’s tough to miss in any workshop with solvents.
Impact on Industry and Environment
With a formula so simple but a structure that’s tightly tailored, 1,2,3-trimethylbenzene weaves its way into fuel additives, solvents, and chemical syntheses. Engineers look for compounds able to boost octane without gumming up engines. Chemists crave molecules that introduce methyl groups in the right spacing, so 1,2,3-trimethylbenzene lands on the list. Refineries usually make it in the process of reforming petroleum, extracting these aromatic hydrocarbons as useful byproducts.
But there’s another side to this. Too much reliance on aromatic hydrocarbons means air quality can take a hit, since volatiles escape and trigger smog or respiratory problems. Data from air quality monitors link high urban hydrocarbon concentrations with asthma and even greater risks in areas near heavy industrial use. Health agencies and environmental groups have called for stricter controls, not just because of emissions at the refinery but spills and evaporation all the way through the supply chain.
Searching for Smarter Solutions
So where do we go from here? Labs and industry leaders have started to look for safer alternatives. The main goal often revolves around designing better containment, boosting recovery from fumes, and switching up production routes to cut down on raw aromatic output. Some research groups invest in catalysts that selectively synthesize just the right benzene derivatives, reducing waste and unnecessary byproducts.
Safer transportation of chemicals, tighter standards within storage, and regular training for workers can lower risks for spills or leaks. At the same time, there’s real innovation in green chemistry—substituting aromatic solvents with plant-based alternatives that break down more easily and create less air pollution. Each step in refining or discarding these chemicals carries consequences, so creating alternatives with low toxicity and environmental persistence makes sense.
It took real experience—splitting time between benches, industry tours, and reading environmental reports—to see how the simple story of “structure and formula” expands into public health and innovation. Every methyl group influences more than a line on a drawing. The story of 1,2,3-trimethylbenzene shows how basic chemistry links together with bigger challenges, including safety and sustainability.
Is 1,2,3-Trimethylbenzene environmentally hazardous?
What Is 1,2,3-Trimethylbenzene?
1,2,3-Trimethylbenzene pops up in petrochemical industries and in some gasoline blends. Most folks come across it as an ingredient in solvents or as a byproduct from oil refining. The strong smell signals that this chemical doesn’t blend into the background, even in small amounts. Whenever I work on projects near refineries or rail yards, colleagues talk about these aromatic chemicals because of their reputations, not just their industrial value.
How Does It Move in the Environment?
This compound doesn’t stick around in one place. Wind and rain carry vapors away from release sites, especially on hot days, helping them spread through air and water. Once it hits soil or water, it doesn’t hang out forever, but it doesn’t vanish instantly either. Microbes eventually break down trimethylbenzene, but the process drags on for several days. In rivers near chemical plants, researchers have measured trace levels lingering far downstream, showing that cleanup takes time.
Is There a Risk to Wildlife?
Fish and aquatic insects can suffer if exposed to high levels. The U.S. Environmental Protection Agency points to evidence that 1,2,3-trimethylbenzene threatens the health of bluegills, minnows, and water fleas. At certain concentrations, these animals show reduced activity and can even die off after long enough exposure. Birds and mammals avoid tainted water sources, but smaller creatures living in those waters don’t have a choice. From my own time volunteering at river cleanups, I’ve seen how even a brief spill can kill hundreds of minnows along the banks — the ecosystem bounces back, but only after a serious hit.
Health Risks for People
Breathing fumes from solvents or fuels containing trimethylbenzenes can cause headaches, dizziness, and throat irritation. Large amounts can inflate risks for liver and kidney trouble. For folks working with this chemical, safety steps matter. Protective gloves, masks, and proper ventilation cut down on accidental exposure. I’ve seen safety lapses at small shops, mostly from poor storage and no training, which shows that education isn’t filtering down as well as it should.
Waste and Accidental Spills
Spills happen during transport or refueling. Clean-up teams use absorbent booms or foams for large liquid releases. Rainwater can carry smaller leaks into storm drains, sending them quickly toward streams. Every drop counts. It’s worrying how often accidents come from overlooked storage tanks or rusty pipelines. I once saw an emergency team dig up an entire parking lot after discovering chemical-tainted soil below a leaking fuel tank — the soil piles waited for months before removal, each rainstorm bringing concerns about spread.
What Can Reduce the Danger?
Good storage practices keep leaks in check. Regular tank inspections, especially at older sites, make a clear difference. Upgrading infrastructure cuts risk — double-walled tanks and lined catchment basins have stopped spills cold. Emphasizing basic training at every level, from lab techs to truck drivers, saves headaches down the line. Community testing on wells near industrial zones builds trust and finds problems before they grow. Local water districts posting annual test data online opens eyes and forces slow-moving companies to pay attention.
Policy and Monitoring
Many states require reporting releases of trimethylbenzene above certain limits. Public records help track trouble spots and push industries to fix weak points. Friends working at environmental nonprofits dig into these records, press for upgrades, and keep officials honest. It’s easier to prevent harm than fix poisoned soil or waterways afterward. With the growth of chemical transport in the last decade, this won’t fade as an issue. Eyes on the process, hands on the fixes — that’s what keeps people and the landscape safe.
| Names | |
| Preferred IUPAC name | 1,2,3-Trimethylbenzene |
| Other names |
Hemimellitene 1,2,3-Trimethylbenzol |
| Pronunciation | /ˌwaɪˌtuːˌθriː traɪˈmɛθ.əlˌbɛn.ziːn/ |
| Identifiers | |
| CAS Number | 526-73-8 |
| 3D model (JSmol) | `3D model (JSmol)` string for **1,2,3-Trimethylbenzene**: ``` C1=CC(=C(C=C1C)C)C ``` |
| Beilstein Reference | 87394 |
| ChEBI | CHEBI:25655 |
| ChEMBL | CHEMBL15414 |
| ChemSpider | 13278 |
| DrugBank | DB01805 |
| ECHA InfoCard | 03e232e9-bd17-4c77-a5a3-2f508dff7bbf |
| EC Number | 202-442-1 |
| Gmelin Reference | Gmelin 785 |
| KEGG | C01480 |
| MeSH | D015867 |
| PubChem CID | 7419 |
| RTECS number | CY1925000 |
| UNII | N7Q218630L |
| UN number | UN2325 |
| CompTox Dashboard (EPA) | DTXSID7020196 |
| Properties | |
| Chemical formula | C9H12 |
| Molar mass | 120.19 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic odor |
| Density | 0.876 g/mL at 25 °C |
| Solubility in water | Insoluble |
| log P | 3.8 |
| Vapor pressure | 2 mmHg (25°C) |
| Acidity (pKa) | pKa = 20.0 |
| Basicity (pKb) | 12.58 |
| Magnetic susceptibility (χ) | -65.5e-6 cm³/mol |
| Refractive index (nD) | 1.490 |
| Viscosity | 0.817 cP at 25 °C |
| Dipole moment | 0.38 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 174.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 104.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4399.8 kJ/mol |
| Pharmacology | |
| ATC code | V03AB72 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335, H411 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P304+P340, P312, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 86 °C (closed cup) |
| Autoignition temperature | “autoignition temperature of 1,2,3-Trimethylbenzene is 480 °C” |
| Explosive limits | 1-6% (by volume in air) |
| Lethal dose or concentration | LD50 oral rat 5170 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 5000 mg/kg |
| NIOSH | MW3800000 |
| PEL (Permissible) | 25 ppm (OSHA) |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 900 ppm |
| Related compounds | |
| Related compounds |
Benzene Toluene Xylene 1,2,4-Trimethylbenzene (pseudocumene) Mesitylene (1,3,5-Trimethylbenzene) |

