Seeing 1,2,3-Trichlorobenzene in New Light
Looking Back: The Progression of 1,2,3-Trichlorobenzene
Chemistry often reveals stories of persistent tinkering, and 1,2,3-Trichlorobenzene stands out as a product that shifted from curiosity to critical industrial feedstock over decades. Its early identification arrived as synthetic chemistry matured in the late nineteenth century. Early researchers often landed on compounds like this during attempts to extract dyes, pharmaceuticals, or solvents from coal tar or aromatic hydrocarbons. Chemists pieced together the puzzle of how three chlorine atoms attached to a benzene ring could steer reactivity and open doors to different reaction sequences. It took time, trial, and error before anyone realized how much potential could rest within a simple structure that looks like little more than a chlorine-spiked version of an aromatic ring. Over time, it found space in both academic curiosity and practical industrial flows, woven into specialty intermediates and used as a benchmark for exploring halogenation processes.
Introducing the Compound: What 1,2,3-Trichlorobenzene Brings to the Table
1,2,3-Trichlorobenzene, often abbreviated as TCB, isn’t flashy. It's a colorless liquid—sometimes faintly yellow if not pure—and the chemical formula, C6H3Cl3, hints at its symmetry and practical use. Factories that run large reactors rely on it because of the right blend of stability and reactive potential. It goes by names like meta-trichlorobenzene or TCB, showing up wherever selective chlorination of benzene rings is on a process sheet. People working with agrochemicals, dyes, and even heat transfer fluids know it as a dependable intermediate. Across the research labs and fields, names for the same molecule vary, but the core identity sticks: it’s a trichlorinated aromatic that’s both persistent and functional.
Peeling Back the Science: Physical & Chemical Properties
Having worked with organic solvents, you learn to appreciate density, boiling points, and solubility for reasons beyond numbers in tables. 1,2,3-Trichlorobenzene carries a density higher than water, giving it a weighty, almost oily pour. At room temperature, it carries a faintly sweet odor—a reminder of volatile aromatics in the lab. Its boiling point usually hovers above 215°C, which lets it stick around in processes that strip out lighter solvents. Water hardly dissolves it, but organic solvents take it in readily. The molecule sits somewhere between stability and reactivity, meaning you can store it well outside fire-prone environments, yet it can also tackle a range of chemical modifications if handed the right reagents and catalysts.
Technical Specs & Labeling: Decoding Practical Details
Labeling in industrial contexts matters for safety and clarity. Companies describe 1,2,3-Trichlorobenzene by its CAS number, molecular weight, and key standards based on region. Purity targets typically exceed 98%, demanded by downstream processes that refuse to tolerate many impurities. Bottles and drums show clear warning labels for toxicity and environmental hazards, pointing out risks associated with inhalation, skin contact, or improper disposal. Over time, regulatory agencies modified labeling requirements, aiming to give truck drivers, plant operators, and lab techs the best shot at handling the compound without mishaps.
How It’s Made: Method of Synthesis
Looking at large-scale production, manufacturers go for direct chlorination of benzene. The process runs with careful control of temperature and chlorine flow rates so the right isomer is favored—this isn’t just a matter of adding chlorine gas and hoping for the best. Catalysts and process conditions determine whether the reaction yields mono-, di-, or trichlorinated products, with separation steps taking over from there. The goal remains sharp selectivity, producing the desired 1,2,3 arrangement without drowning in byproducts. Waste treatment and recycling often keep up with the process, since the halogenated byproducts can’t fall by the wayside.
Chemical Reactions & Ingredient Tweaks
1,2,3-Trichlorobenzene answers well to reduction, nucleophilic substitution, and even further functionalization if the right pressure or reagent steps in. The trio of chlorines opens up well-defined, sometimes unpredictable, reactivity with metals and nucleophiles. Its structure encourages chemists to try cross-couplings, hoping to insert new groups and test out catalysts. Industrial users look at these options when targeting new herbicide precursors or specialty chemicals. From experience, reactions can swing from smooth to troublesome depending on temperature, catalyst choice, and even the glassware in use—a reminder that textbook chemistry rarely matches reality in factory settings.
What’s In a Name: Synonyms & Use Cases
Those who spend their days with chemicals rarely call things by full names. In catalogs, you run into meta-trichlorobenzene, TCB, sometimes even “trichlorobenzene” with a position noted only in the fine print. Each label points to a single core product, though the context—lab, field, or factory—can flavor which name takes hold. Specialized applications came up as demand for dye precursors and agrochemical intermediates grew in the 1970s and 80s, tying a simple molecule back to evolving routes in the chemical economy.
Staying Safe: Standards for Handling & Storage
Listening to folks in chemical plants tells you why intense focus on safety makes sense. Technicians rely on gloves, goggles, and tightly ventilated workspaces because 1,2,3-Trichlorobenzene can irritate skin and lungs. Regulatory frameworks, such as OSHA in the U.S. or REACH in Europe, call for detailed material safety data. Safe containment counts as much as personal equipment: drums are sealed tight, warnings sit front and center, and spill prevention plans roll out well before trucks even leave the storage yards. Management systems stress regular training, since one slip can result in exposures that create health issues or trigger environmental fines.
Where It Fits: Exploring Application Areas
In real-world industry, the reach of this compound surprised even me: its uses stretch from seed formulations in agriculture to precursors for specialized colorants and resins. Dyes owe some of their intense shades to trichlorobenzene intermediates. In some thermal fluids, its chemical and physical profile aligns neatly with what engineers want—stability at heat, resistance to oxidation, and ease of handling. Armed with its chlorinated backbone, researchers keep pulling new reactions and applications from the TCB playbook, each iteration trying to match shifting demands for purer, more sustainable intermediates.
On the Frontier: Research and Development Efforts
Research labs focus on greener and more efficient synthesis. Much of this effort aims to cut hazardous waste and improve atom economy. Catalysis teams chase selective chlorination routes that minimize byproducts. Analytical chemists refine detection methods, mostly to address contamination in environmental samples. Scientists chase new modifications, such as building more complex rings or integrating functional groups that serve biocidal or pharmaceutical targets. Several labs have reported new routes using ionic liquids or novel catalysts, proving that even mature chemical processes can yield to modern techniques, reducing both waste and energy consumption.
Toxicity: Addressing the Risks
The dark side of chlorinated aromatics appears in their persistence and health consequences. Studies show that inhalation or repeated exposure stirs up symptoms from headaches to long-term organ damage. Environmental research links TCB and its cousins to soil and water contamination that lingers far longer than most regulators would like. Animal testing unveiled risks to reproduction and development, and agencies class trichlorobenzenes as potential environmental hazards that require scrutiny at every stage—from manufacture to disposal. My experience with solvent cleanups supports the need for robust monitoring and personal vigilance, especially since invisible vapors can escape even what seems like meticulous care. Ongoing studies investigate chronic low-level exposures and their links to cancer, urging regulators and industry to tread carefully and invest in harm reduction.
Looking Ahead: Prospects for 1,2,3-Trichlorobenzene
Environmental pressure and regulatory tightening spur changes in both how TCB is made and where it finds use. The chemical industry keeps exploring ways to minimize chlorine waste, and substitutes for the dirtiest applications move up the agenda in response to stricter governance. Downstream users hunt more targeted reactions to reduce unnecessary emissions. Academic groups push out new sensor technologies and trace analysis systems to track the chemical in air, water, and sediments. Interest builds around safer alternatives, but for many applications, 1,2,3-Trichlorobenzene keeps a strong foothold due to its unique balance of reactivity and stability. As the field advances, only a blend of innovation, vigilance, and ethical priorities will shape the story of this persistent aromatic in a world facing complex chemical risks and opportunities.
What is 1,2,3-Trichlorobenzene used for?
What Sets 1,2,3-Trichlorobenzene Apart
In the chemical world, a few names pop up that play a big role behind the scenes. 1,2,3-Trichlorobenzene is one of those names. Folks working in manufacturing or chemical supply see the benefits and risks that come with it up close. This compound turns up in a few key processes and products, many far removed from the average household. Sitting on a shelf, it looks like a clear liquid. In the real world, it makes waves across plastics, dyes, and more.
In the Plastics and Polymers Factory
On the production line, 1,2,3-Trichlorobenzene acts as a solvent. During some plastic and polymer processes, certain raw materials refuse to mix without a strong nudge. That’s where this compound comes in. Take polyphenylene sulfide as an example; it takes real effort to dissolve and shape, and 1,2,3-Trichlorobenzene handles that job. The result: plastics tough enough for parts used in electronics and the auto world. Modern manufacturing asks for components to resist heat, and this compound quietly supports that demand.
A Tool for Chemical Synthesis
Some chemists use 1,2,3-Trichlorobenzene to create other compounds. Paint pigment producers lean on it to break down raw materials and pull out specific colors. Laboratories study new chemical reactions by adding a bit to the mix, watching how it changes the outcome. It's true, handling such chemicals means needing solid training and tough safety rules, but without these building blocks, many products wouldn’t come to life.
Pesticides, Dyes, and Beyond
Not all applications take place in factories. The dye and pesticide industries have a long history with 1,2,3-Trichlorobenzene. Workers use it as a starting point, building new molecules needed to target specific pests or to color fabrics evenly and deeply. There’s real value to consistency in those businesses—farmers count on effective pest control, and designers expect dye colors to last. Having the right chemical process makes all the difference.
Risks and Realities
Exposure brings up health worries. If you spend time in a lab or manufacturing site, you know why gloves and fume hoods matter. Inhaling or touching this compound can lead to headaches, dizziness, and over long periods, even more serious health impacts. That means enforcing safety measures is not a box-checking exercise—those rules protect real people. In my time working with chemical supply teams, I’ve seen both strong safety cultures and a few near-misses. Safety training and careful storage can prevent most issues before they start.
Seeking Safer Alternatives
As factories explore better ways of running things, talk often turns to finding safer options. Some companies have started testing solvents with lower toxicity, trying to limit workplace risk and reduce the impact on wastewater. Replacing 1,2,3-Trichlorobenzene completely won’t happen overnight, but research into greener processes has real momentum. Smart investment in new chemistry, plus pressure from environmental groups and buyers, helps keep that progress moving.
Final Thoughts
Everyday consumers rarely see or touch 1,2,3-Trichlorobenzene, but it shapes what they use. The jobs it does—helping produce plastics, dyes, and specialty chemicals—matter a great deal. Striking a balance between industrial productivity and worker safety depends on careful use, strong regulation, and ongoing innovation.
Is 1,2,3-Trichlorobenzene hazardous to health?
A Closer Look at 1,2,3-Trichlorobenzene
People rarely talk about 1,2,3-Trichlorobenzene outside laboratories or industrial sites, but its impact deserves attention. This chemical shows up in the manufacture of dyes, pesticides, and as a solvent, places where workers often face the highest risks. With a sharp, chlorine smell, it gets into air and water during industrial use. Breathing even low levels in a factory setting can bring on headaches, dizziness, throat irritation, and sometimes more severe problems.
Worker Safety and Long-term Exposure
Direct experience matters. Anyone spending time near barrels of industrial chemicals will tell you that repeated exposure, day after day, takes a toll. Skin absorbs it quickly, and hands left unwashed burn or itch. After long weeks, lungs don’t feel right, and sleep comes fitful. If companies lack strong safety measures, the effects multiply. Public data echo what workers know: the U.S. National Library of Medicine lists eye and skin irritation, possible liver and kidney damage, and effects on the nervous system with chronic contact. These findings aren’t alarmist—they come from actual cases in factories.
Health Evidence from Reliable Sources
Research provides hard facts. The U.S. Environmental Protection Agency shows that 1,2,3-Trichlorobenzene doesn’t just irritate; it may linger in the body, acting as a possible carcinogen. Studies on animals showed damage to organs after repeated doses, and scientists observed that it often builds up in fatty tissues. Even low doses over months can have subtle, long-term effects not obvious at first. The World Health Organization grouped certain chlorinated benzenes like this one as “of concern” for communities close to industrial zones. I trust reports like these because they rely on years of consistent findings.
Real-World Environmental Concerns
On job sites, I watched how carelessness or leaks sent chemicals into water or soil. 1,2,3-Trichlorobenzene doesn’t break down quickly. Streams near chemical plants showed fish kills, and people living nearby worried about their water. Regulatory agencies stepped in to monitor contamination and demand cleanup. I saw EPA teams sample soil, and while tests aren’t cheap, they soon found the chemical levels above guidelines. In these cases, basic precautions such as sealed storage and proper disposal meant the difference between a spill and a disaster.
What Can Be Done Better?
Real improvement starts at the workplace. Companies benefit from placing effective air ventilation, providing strong gloves, and making clean-up easy. I’ve met people whose bosses skipped these steps who paid the price with health scares. Employers who act on expert advice from groups like OSHA waste less time on sick days and lawsuits. Local governments also have a role—regular inspections and quick response to spills prevent accidents from spreading beyond factory walls.
The Bottom Line
Any chemical that can damage human organs, start headaches, or poison a river asks for respect. Teams leading plants should handle 1,2,3-Trichlorobenzene with real care. Lawmakers, regulators, and everyday workers all play a part in keeping each other safe. The cost of ignoring health risks piles up fast, in both dollars and human stories. We already know enough to do better.
What are the storage and handling guidelines for 1,2,3-Trichlorobenzene?
Understanding 1,2,3-Trichlorobenzene Risks
1,2,3-Trichlorobenzene shows up in a variety of industrial settings, from use as an intermediate for dyes and pesticides to roles in research and formulation. Its chemical structure delivers a certain stubbornness: it doesn’t break down easily, and the vapors hit hard if someone ignores ventilation. People handling it can’t treat it as just another chemical on a list.
Storing 1,2,3-Trichlorobenzene Correctly
The best practice is simple. Keep containers tightly closed. Use steel drums or high-density polyethylene, always looking for signs of rust or wear, since leaks draw trouble. Temperature plays a big role too. Store at room temperature, away from sunlight and direct heat sources. High heat, even for a short while, can bump up vapor levels in a hurry.
It pays to avoid stacking heavy drums and always label containers with clear hazard symbols. Fire departments need to know what sits in that storage room, so tracking inventory and updating SDS sheets keeps everyone safer. From experience, overlooking a single drum at the back can spell a major headache during inspections or emergencies.
Hazards and Real-World Stories
Chlorinated benzenes like this one come with stubborn vapors and can cause irritation or worse if they splash onto skin. I remember seeing a lab worker ignore splash goggles because “it’s a routine process.” The next morning, that small mistake left him facing harsh chemical burns. It’s the ordinary steps, like eye protection and gloves, that often get swept aside during busy shifts—yet those steps mean the difference between a safe shift and a long trip to urgent care.
The fire risk gets overlooked too. 1,2,3-Trichlorobenzene burns with a nasty set of byproducts, including phosgene and other noxious gases. An audit at a chemical plant once revealed forgotten electrical cords passing close to stored drums. It startled the team into a major cleanup. Keeping flammables and ignition sources separated isn’t just a textbook rule—it’s common sense proven by near misses and worse.
Safe Handling Keeps Workers Healthy
A strong ventilation system pulls vapors away from breathing zones. In a cramped stockroom or lab, skip the shortcut of “just a bit of fresh air.” Mechanical exhaust, face shields, thick nitrile gloves, and chemical aprons turn a risky process into something manageable. Spill kits with proper adsorbents—never sawdust or sand—shorten response time if something goes wrong.
Training makes the biggest difference. I’ve seen it firsthand: veterans with years on the job share stories of past incidents, making safety more than just a checklist on a wall. Pairing new workers with mentors helps keep old mistakes from coming back in a new form.
Simple Steps for a Safer Workplace
Metal shelving, spill containment pallets, and clear floor markings cut down on clutter and confusion. Weekly checks—really walking the floor and looking at every label—keep things from sliding toward chaos. Emergency showers and eyewash stations need to sit a few steps from the work area, with regular water tests showing they’ll work if someone ever needs them.
Waste needs careful attention. Label each waste drum and schedule regular pickups, following hazardous waste rules. Accidents often come from mixing incompatible chemicals in waste streams, so keep logs and train every crew on disposal procedures.
Safe storage and steady routines turn an industrial hazard into a chemical that does its job without hurting anyone involved. It’s about respect for the material, built through small daily habits and a steady focus on common sense.
How should 1,2,3-Trichlorobenzene spills or exposures be managed?
Why 1,2,3-Trichlorobenzene Gets So Much Attention
People seldom hear about 1,2,3-Trichlorobenzene unless something gets spilled or a worker calls in sick after handling chemicals. But experts and those on the front lines know the stakes. This chemical, used in research labs, pesticide manufacturing, and dye production, carries real risks. The Centers for Disease Control and Prevention (CDC) highlights its potential for harming living tissue, from burning eyes and skin to impacting internal organs with enough exposure.
Spills Happen: What Makes Response So Critical?
One thing learned from years in industry is that most chemical accidents catch people off guard. It may be as simple as a dropped drum or a leaking gasket. But with 1,2,3-Trichlorobenzene, a slow response can turn a bad day into a health emergency. This chlorinated compound evaporates at room temperature, so vapors can fill a space before anyone sees a puddle. Acute inhalation causes nausea, headaches, or worse: lasting liver or kidney damage. All it takes is a small slip and a lack of protective gear to be in trouble fast.
Real Steps to Take When Spills Happen
If a container tips or leaks, ventilation becomes a top priority. Opening windows isn’t enough. Industrial fans clear contaminated air and pull vapors away from workers. Anyone without a certified respirator and chemical gloves should move out of the spill area fast. Workers who stay behind for cleanup gear up in full splash-resistant suits and goggles. People rely on these barriers because skin absorption is as much of a risk as breathing in the vapors.
Absorbent pads and neutralizers help soak up and contain the liquid. Those products see a lot of action in real-world cleanups, not just on paper. Afterward, the waste goes to hazardous material drums—never down a drain or into landfill. Proper labeling keeps anyone from making a costly mistake later in the process. Reports to local health and fire departments show accountability and keep neighbors out of harm’s way.
Let’s Not Pretend: Exposure Happens
Old habits die hard. Cuts in training or protective equipment lead to sloppy shortcuts. Some facilities focus so much on productivity that people forget these habits can cost lives. Workers exposed to even small amounts feel the effects quickly. If someone breathes the vapors or gets chemical on their skin, showers and fresh air can help—but only after reaching safety, and usually only after removing contaminated clothing right away. Medical help shouldn’t wait.
Education and Strict Policy: Not Just Bureaucracy
Every year, news stories surface about chemical spills or illegal dumping. That tells us company policies don’t always get real support on the floor. Change comes in the form of regular drills, posted action plans, and empowering everyone—especially those down on the floor—to call out unsafe practices. That’s real E-E-A-T: putting expert know-how, genuine care, and clear communication into action.
Plenty of organizations run on tight budgets, but safety must rank above convenience. Investing in ongoing training, regularly refreshed PPE stock, and easy access to spill kits makes a difference. State environmental hotlines exist for a reason: so employees and communities can stay informed and protected. Honest, plain-language communication turns a harrowing cleanup into a manageable incident—and gives workers the confidence to focus on their jobs in a safer environment.
What is the chemical structure and molecular formula of 1,2,3-Trichlorobenzene?
What Makes 1,2,3-Trichlorobenzene Unique
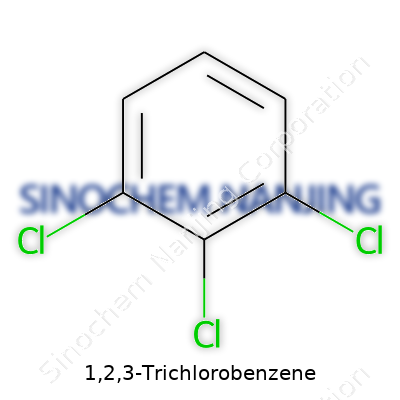
Chemistry shapes so much of daily life, and 1,2,3-Trichlorobenzene reminds us how small molecular changes shift the direction of entire industries. It’s a chlorinated aromatic hydrocarbon, meaning the core is a benzene ring dressed with chlorine atoms. That benzene ring, a familiar hexagon in textbooks, picks up three chlorine atoms each hanging onto the 1st, 2nd, and 3rd carbons. The official molecular formula reads as C6H3Cl3.
Structure: Where Atoms Meet Intention
The arrangement isn’t random. Chlorines parked side-by-side impact properties like melting point, solubility, and reactivity. This "ortho" disposition on the ring sounds like jargon, but visualizing those chlorines as three neighbors in a row gives clarity. Groups like these change how the molecule interacts with other chemicals. I’ve seen how just shifting a single chlorine position changes solvent applications or toxicity levels. Structurally, the remaining hydrogen atoms occupy the 4th, 5th, and 6th spots, rounding off the six-ringed shape. The chemical structure shows alternating single and double bonds, giving the ring its hallmark stability.
Why the Formula and Structure Matter
Anyone mixing chemicals or designing a process cares about details here. The presence of three chlorines causes 1,2,3-Trichlorobenzene to behave differently from its siblings like 1,3,5-Trichlorobenzene. Properties such as boiling point (218°C) and melting point (53°C) stem from this specific arrangement.
In manufacturing, a formula is more than ink on a page. Safety data, environmental profiles, and process rules all flow from it. Industrial workers keep the formula handy, because inhalation or contact guidelines depend on it. Lab personnel use the structure to predict solvent compatibility, wasting less time on failed mixtures.
Health, Environment, and Challenges
Many of the risks appear right on the safety sheets. As a persistent organic chemical, 1,2,3-Trichlorobenzene can linger in the environment. With its resistance to breakdown, waterways and soil can bear traces long after a spill. Personal experience in environmental monitoring showed cleanup is never simple; solvents tend to dissolve such residues, but the process spreads chlorinated byproducts. Regulations limit its industrial discharge, nudging manufacturers to look for biodegradable alternatives.
On a practical level, those working with this compound use the formula as a snapshot of potential hazards. The more chlorines anchored to benzene, the higher the risk of bioaccumulation and toxicity in ecosystems. Even with protective gear and handling protocols, small mistakes create real danger. Communities near production sites bear the burden if storage or transport goes wrong.
Practical Solutions
Industries can use closed-loop systems and more advanced filtration to prevent leaks and contamination. I’ve seen improvement using activated carbon and specialized resin filters—they trap chlorinated molecules before reaching natural water sources. Engineers and scientists keep exploring catalysts that break down trichlorinated compounds safely, converting lingering chemicals into harmless substances. More transparent reporting from companies and wider use of alternatives lower the risk over time. Watching regulations drive better practices reminds me that, at its core, responsible chemistry values people and places.
Final Thoughts on Why This Knowledge Matters
Chemical structure and formula underpin responsible management and innovation. For 1,2,3-Trichlorobenzene, every detail shapes regulations, safety, and environmental impact. The right decisions start with trusting the science behind its molecular form. People making, using, or living near these chemicals depend on informed choices grounded in real structure—not just words in a textbook.
| Names | |
| Preferred IUPAC name | 1,2,3-Trichlorobenzene |
| Other names |
1,2,3-TCB Sym-Trichlorobenzene Trichlorobenzene Benzene, 1,2,3-trichloro- m-Trichlorobenzene |
| Pronunciation | /ˌwʌn tuː θriː traɪˌklɔːrəˈbɛnzɪn/ |
| Identifiers | |
| CAS Number | 87-61-6 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:34208 |
| ChEMBL | CHEMBL16287 |
| ChemSpider | 14104 |
| DrugBank | DB13815 |
| ECHA InfoCard | 100.003.111 |
| EC Number | 204-428-0 |
| Gmelin Reference | Gmelin 774 |
| KEGG | C01837 |
| MeSH | D014264 |
| PubChem CID | 6978 |
| RTECS number | DC3325000 |
| UNII | A4R1FV59WM |
| UN number | UN2321 |
| CompTox Dashboard (EPA) | DTXSID2020327 |
| Properties | |
| Chemical formula | C6H3Cl3 |
| Molar mass | 181.45 g/mol |
| Appearance | Colorless crystalline solid |
| Odor | Aromatic odor |
| Density | 1.450 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 4.02 |
| Vapor pressure | 0.39 mmHg (25°C) |
| Acidity (pKa) | 2.70 |
| Magnetic susceptibility (χ) | -71.0e-6 cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 1.14 mPa·s (25 °C) |
| Dipole moment | 2.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 310.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 78.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4052.3 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ57 |
| Hazards | |
| GHS labelling | **GHS02, GHS07** |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H373, H410 |
| Precautionary statements | P261, P273, P280, P301+P310, P305+P351+P338, P308+P313 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 110°C (230°F) |
| Autoignition temperature | 648 °C |
| Lethal dose or concentration | Lethal dose (oral, rat): 756 mg/kg |
| LD50 (median dose) | LD50 (median dose): 756 mg/kg (oral, rat) |
| NIOSH | PC8225000 |
| PEL (Permissible) | 75 ppm |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | 50 ppm |
| Related compounds | |
| Related compounds |
Chlorobenzene 1,2-Dichlorobenzene 1,3-Dichlorobenzene 1,4-Dichlorobenzene 1,2,4-Trichlorobenzene 1,3,5-Trichlorobenzene Hexachlorobenzene |

