1,2,3,6-Tetrahydrobenzaldehyde: A Comprehensive Commentary
Historical Development
Chemists first characterized 1,2,3,6-Tetrahydrobenzaldehyde in the early decades of the twentieth century, marking an important step in the evolution of cyclic aldehydes. The demand for partially saturated aromatics like this compound grew alongside the rising needs in fragrance, pharmaceutical, and polymer industries. Laboratories sought better substitutes for benzaldehyde derivatives due to the latter’s harsh reactivity and tendency to form byproducts. Researchers soon realized that 1,2,3,6-Tetrahydrobenzaldehyde could slot into processes where stability and modern reactivity were needed. Years of work in both academic literature and industry patent files reveal a steady climb in interest, painting a story of adaptation driven by necessity and practical benefit.
Product Overview
Commonly found as a clear to pale-yellow liquid, 1,2,3,6-Tetrahydrobenzaldehyde attracts attention thanks to its manageable volatility and unique aromatic profile. Laboratories use it as a starting point for fine chemicals, such as specialty polymers and designer fragrances. Its ease of handling, compared with stronger, fully aromatic aldehydes, brings a sense of flexibility and safety. Manufacturers often blend it with other cyclic chemicals in scent compositions or as intermediates for more complex molecules, making it a frequent feature in both research and production inventories.
Physical & Chemical Properties
With a molecular formula of C7H8O and a molecular weight close to 108.14 g/mol, this compound straddles the line between classic aromatics and their aliphatic cousins. The saturated portions of its ring dampen the aggressive reactivity seen with benzaldehyde. It has a boiling point in the range of 205 to 210 degrees Celsius, with a density near 1.03 g/cm³. Its moderate solubility in polar solvents lets chemists design around both hydrophobic and hydrophilic environments. The sharp aldehyde scent, rounded out by a softer undercurrent, reflects its semi-aromatic nature. Technicians working with this molecule report that the vapor isn’t as harsh as that of many aldehydes, so standard ventilation covers most use cases.
Technical Specifications & Labeling
Bottles leave the manufacturer’s facility with labels declaring purity levels, CAS number (16517-70-3), lot numbers, and expiration dates. Typical purities exceed 97%, usually confirmed by gas chromatography. For international transit and industrial acceptance, labels show compliance with REACH, GHS pictograms, and recommended storage conditions—usually below 25 degrees Celsius in amber glass. Transporters who handle this compound use UN numbers relevant to lower toxicity organics, easing regulatory headaches.
Preparation Method
The standard preparation of 1,2,3,6-Tetrahydrobenzaldehyde begins with partial hydrogenation of benzaldehyde under carefully controlled pressure and temperature. Catalysts like palladium or nickel steer the reaction just enough to saturate specific ring positions without attacking the prized aldehyde group. Some labs prefer to start from tetrahydronaphthalene, oxidizing it gently to avoid over-oxidation and ring cleavage. Each route throws up challenges—over-reduction leads to loss of aromaticity, under-reduction means the starting material dominates the mix. Technicians spend years perfecting ratios and run times, learning to trust both their instruments and their noses.
Chemical Reactions & Modifications
This compound supports a suite of reactions. Researchers count on its aldehyde function for condensation reactions, notably with amines and hydrazines, spawning new N-heterocycles and Schiff bases. As a precursor, it morphs under aldol conditions or accepts nucleophilic additions without breaking apart the fragile ring structure. Its partially saturated nature lets it serve as a test case for selective hydrogenation or oxidative tuning. Those working on pharmaceutical actives and next-generation materials appreciate these traits. Checked under routine lab stress, it resists polymerization and oxidation better than many aldehydes, reducing the surprise factor that comes with finicky carbonyl chemistry.
Synonyms & Product Names
Beyond the IUPAC name, this chemical appears as “tetrahydrobenzaldehyde,” or “3,4,5,6-tetrahydrobenzaldehyde” in trade catalogues and material safety sheets. Older literature sometimes describes it as “cyclohex-3-enecarbaldehyde.” You might see proprietary product names, especially in catalogs of fragrance or specialty intermediates suppliers, but the structure remains the same. These naming quirks can create search headaches, especially for students and new professionals.
Safety & Operational Standards
Chemists handling 1,2,3,6-Tetrahydrobenzaldehyde equip themselves with nitrile gloves and goggles since aldehydes act as irritants. Even though this compound causes fewer headaches than its completely aromatic cousin, it still demands care to avoid skin or eye contact. Storage rooms remain cool and dry to prevent slow oxidation and maintain shelf life. Industrial-scale users rely on fume hoods and explosion-proof refrigerators. Regulatory agencies stack this compound in the “harmful but not acutely toxic” class, with safety sheets laying out emergency measures for spills and exposure. Over years in the lab, attention to such details means you rarely see injuries linked to this chemical.
Application Area
You meet 1,2,3,6-Tetrahydrobenzaldehyde most often in the labs shaping the fragrance, flavor, and fine chemicals landscapes. Its ability to form new rings—plus a soft, nuanced scent—makes it valuable for specialty perfumery houses. Polymer scientists and pharmaceutical developers appreciate its balance of reactivity and stability, letting it build everything from heterocyclic building blocks to functionalized intermediates. Materials science teams occasionally slip it into resins or investigate its potential for new elastomer formulations. Its presence in chemical libraries signals versatility more than niche status.
Research & Development
Teams seeking new catalysts, reaction paths, or advanced intermediates revisit this compound every few years. Green chemistry researchers focus on optimizing hydrogenation protocols, seeking less wasteful conditions, recyclable catalysts, and milder reagents. Pharmaceutical teams screen it as a core structure for new bioactive molecules, while computational chemists run studies on its partial aromaticity to predict its suitability in electronic or optical devices. Early-stage investigations explore its ability to act as a masked diene for late-stage functionalization. Bench chemists remember it for projects that balanced challenge with reward, not just for the end product, but for lessons gained about controlled reactivity and process engineering.
Toxicity Research
Safety research into 1,2,3,6-Tetrahydrobenzaldehyde shows moderate eye and skin irritation among animal models, but no acute toxicity at low exposures. Chronic studies remain sparse, because the chemical rarely features at high doses outside industry. Marked similarities between this compound and related aldehydes caution against inhaling vapors or allowing repeated skin contact. Animal data suggest low bioaccumulation, but researchers urge continued monitoring, especially for occupational settings or large-volume scenarios. Environmental scientists keep an eye on its breakdown in wastewater, as partially saturated rings transform differently from standard aromatics; most assessments indicate modest persistence and low biohazard risk if contained and neutralized.
Future Prospects
Future directions for 1,2,3,6-Tetrahydrobenzaldehyde center on improved synthetic access and innovative applications. Process engineers continue tweaking hydrogenation and selective oxidation routines, targeting higher yields and fewer byproducts. Teams in materials science and medicine probe its semi-aromatic ring system, betting on new reactivity that earns a place in sustainable polymers and drug candidates. Fragrance houses place bets on nuanced scent molecules shaped from its backbone, and regulatory bodies probe for any health or environmental downsides as production scales up. Collaborative R&D between academic labs and industry often turns up new uses, showing that even well-studied compounds keep surprising us.
What is 1,2,3,6-Tetrahydrobenzaldehyde used for?
Everyday Chemistry Behind the Scenes
You may never spot a bottle labeled "1,2,3,6-Tetrahydrobenzaldehyde" in a grocery aisle, but this mouthful of a compound quietly shapes modern products. For chemists like me, it represents a strange intersection between the clean, careful world of chemical theory and the messier tide of consumer demand. Years back, walking the aisles of a flavor and fragrance plant, I could swear the subtle, slightly spicy aroma in the air held a secret — often, that secret came down to compounds like this one.
What Does Tetrahydrobenzaldehyde Do?
In practice, 1,2,3,6-Tetrahydrobenzaldehyde works primarily as an intermediate. This means chemists use it as a sort of stepping stone—building bigger, more complex molecules for the flavor and fragrance industries, or pharmaceuticals. Researchers have used it in the creation of musky, woody scents that get blended into perfumes, soaps, and laundry detergents. Fragrance formulators like its stable, long-lasting aroma profile.
Outside perfume labs, it shows up in flavor design as well. Certain artificial flavors aiming for nutty or spiced notes can trace their chemical ancestry back to it. It’s never poured straight into your favorite packaged snack, yet the underlying chemistry supports the taste that consumers expect. Chemically, it’s a benzaldehyde derivative, which links it to the family of compounds providing the almond note in cherry-flavored soda or marzipan.
Industrial Impact and the Need for Responsible Use
Sitting in research meetings, I've noticed that certain chemicals tend to pop up in safety briefings more than others. 1,2,3,6-Tetrahydrobenzaldehyde earns its share of attention. While it’s useful, safety data sheets emphasize caution: skin and eye irritation stand out as risks, and environmental persistence raises tough questions about waste disposal.
Any chemical with a scent profile strong enough to build perfumes will naturally attract attention from regulators and environmentalists. There’s growing pressure to double-check how solvents, byproducts, and excess tetrahydrobenzaldehyde get disposed of before leaving chemical plants. The Responsible Care framework, adopted by major chemical manufacturers, has led to closed-loop production systems and cleaner wastewater — progress I’ve witnessed firsthand.
Why This Matters
In my early career, I thought of specialty chemicals as silent, invisible helpers, never lingering long enough to cause problems. Over time, mounting research showed that even trace amounts can affect water quality and aquatic life. Practical solutions matter: batch tracking, waste recycling, and adherence to regulatory limits really do prevent contamination.
Science moves quickly, and newer green chemistry pathways are in development to minimize reliance on aggressive solvents. Some plants have shifted toward biosynthetic routes that mimic nature’s methods for making similar scent compounds. These innovations cost more and take time, yet protect both workers and downstream communities.
Looking Ahead
Chemistry shapes the comfort and pleasure of modern life in surprising ways. Next time the whiff of fresh laundry stirs a memory, there’s a fair chance some clever formulation built on the sturdy backbone of 1,2,3,6-Tetrahydrobenzaldehyde had a part in it. Keeping those comforts safe for people and the planet means never taking shortcuts on chemical stewardship and investing in cleaner, smarter production from the ground up.
What is the chemical structure of 1,2,3,6-Tetrahydrobenzaldehyde?
The Shape of the Molecule
Most of us are taught early on that chemistry is hard to crack, but I think it gets easier when you put a face to a name. 1,2,3,6-Tetrahydrobenzaldehyde may sound intimidating, but its structure isn’t just a pile of letters and numbers. This molecule has a backbone built on the benzene ring we all used to sketch in class, except its ring gets some hydrogen treatment—just enough to change both look and behavior. In this molecule, the benzene ring picks up extra hydrogens at carbons 1, 2, 3, and 6, so it loses a bit of its aromatic punch and adopts more flexibility, almost like relaxing a tight knot.
At one position, usually carbon 1, the classic benzene’s hydrogen swaps out with an aldehyde group. That means you have a framework that blends a partially saturated aromatic ring with a carbonyl slice sticking out like a handle—one carbon doubling up to an oxygen, capped with a single hydrogen atom. The presence of this aldehyde group transforms the structure from a boring hydrocarbon to a reactive builder block, making it useful well beyond the textbook page.
Significance in the Lab and Beyond
For those who work at a lab bench or ever tried to piece together molecules from simple kits, you know that every tweak gives a new tool for building more complicated substances. 1,2,3,6-Tetrahydrobenzaldehyde steps in as a flexible intermediate in the synthesis of flavors, fragrances, and pharmaceuticals. The aldehyde group turns this compound into a useful springboard because chemists can attach or swap out parts right at that site with a wide range of other chemicals. Once you recognize its structure, you also start to see how slight changes can open new doors—like moving the aldehyde around the ring or adding other groups in, which is what the industry depends on to stay innovative.
Why Structure Matters
Structure isn’t just academic trivia. If you work with chemicals, knowing the difference between a fully aromatic ring and a partially hydrogenated one means predicting how it will behave in a flask or inside machinery. The extra hydrogens on 1,2,3,6-Tetrahydrobenzaldehyde’s ring knock down the stability a plain benzene would have, making the molecule more open to reactions. For professionals working with syntheses, that means more options for bond-making, especially at the ring positions next to the aldehyde.
The structure also matters for safety and handling. Aldehydes, in general, can cause irritation with short exposure. Extra hydrogen saturation always shifts volatility and sometimes lowers the combustion risk, but the carbonyl group ensures careful storage and PPE still come first. It’s one of those molecules you handle with respect—you learn the hard way not to cut corners.
Paths Forward
Chemists and process engineers always look for greener routes to these kinds of building blocks. Reducing hazardous waste and making reactions more efficient keeps the pressure on for cleverer ways to use 1,2,3,6-Tetrahydrobenzaldehyde. Researchers continue seeking milder conditions for synthesizing it, less wasteful reagents, and more straightforward purification methods. The more people understand the structure and its behavior, the better their chances of unlocking safer, cheaper, and more flexible applications.
What safety precautions should be taken when handling 1,2,3,6-Tetrahydrobenzaldehyde?
Why Safety Matters with Chemicals Like This
Anyone who’s spent time in a lab or around industrial solvents knows chemicals like 1,2,3,6-Tetrahydrobenzaldehyde demand respect. Though not as notorious as some, this compound comes with its share of dangers. The sharp, pungent odor gives away just how volatile it can get. Breathing in even a little can irritate your nose and throat, and getting it on your skin often means burns or nasty rashes. Stories about workplace accidents show what goes wrong when corners get cut on precautions. No one wants a trip to the emergency room or a call from a regulatory inspector, so safe handling counts for a lot.
Personal Protective Gear Goes Further Than Lab Coats
Relying on a basic lab coat and gloves won’t cut it if you’re serious about working safely. Thick nitrile or neoprene gloves hold up better against leaks and splashes. Goggles that seal tight not only keep vapors out of your eyes but also stand up to accidental sprays. I always keep an extra pair of gloves and a full-face shield nearby in case work gets messy. For clothes, long sleeves and chemical-resistant aprons help keep skin covered. Years of experience have taught me you never want to rush a cleanup if something splashes—grab the right fix from a safety shower or an eyewash fountain immediately.
Ventilation and Storage: The Big Two
Good ventilation makes a huge difference. Relying on a standard fume hood rather than open-air spaces cuts down the buildup of noxious fumes. Keep containers tightly sealed and labeled, and store them away from any ignition sources or sunlight. I’ve seen colleagues get burned by forgetting heat lamps nearby or stacking containers too close together. Chemical storerooms should stay dry and well-organized. Flammable cabinets save headaches in case something spills or a fire breaks out. Check expiration dates and inventory often—old chemicals break down and get even more unstable.
Spill Control and Waste Disposal
Accidents happen even if you stay careful, and being ready with the right spill kit can stop damage from spreading. Absorbent pads, neutralizing agents, and proper waste containers make it simple to handle drips and splashes before they get worse. I keep the kit close to workspace doors for fast response. Never dump this stuff down the drain—contact trained hazardous waste disposal services who know how to handle aldehyde compounds by the book. Training staff on emergency steps saves injuries and fines from regulators. In my experience, a quick huddle before starting a new process works better than long emails no one reads.
Safety Comes from Culture, Not Just Gear
Following safety checklists means little if no one believes in them. Talk openly with coworkers about why rules exist and encourage everyone to speak up if something feels wrong. Encourage reporting mistakes—fixing small problems early often prevents tragedies. Don’t ignore small leaks or ventilation hitches, and don’t work alone if you don’t have to. Safety isn’t just about following the law; it’s about making sure everyone goes home healthy at the end of the day. The real key? Treat every chemical, familiar or not, with the same respect as the first day you ever stepped into a lab.
How should 1,2,3,6-Tetrahydrobenzaldehyde be stored?
Aldehydes and Everyday Caution
Having spent years in research labs, I've developed a strong respect for chemicals with reactive groups. 1,2,3,6-Tetrahydrobenzaldehyde, a mouthful for most, falls into the aldehyde category. Folks who handle these molecules in the field, from fragrance manufacturing to advanced synthesis, know the risks if you get careless.
Why Storage Matters
Poor storage habits not only ruin your material but send money down the drain and raise the risk of unintended chemical reactions. Aldehydes are known to oxidize and polymerize, especially if air or moisture gets in the bottle. That triggers off-odors and unhappy surprises in your reactions.
What prompted my respect for careful storage? One time, working late on a grant deadline, I grabbed a bottle of an "old" aldehyde—stored poorly after a short run by a colleague. Results? Polymerized gunk. Time lost, resources wasted. That drives the point home: proper storage means reliability and safety.
The Right Environment
Aldehydes generally should be kept in a tightly sealed, clearly labeled container. No cutting corners here—a polyethylene or amber glass bottle with a strong lid keeps light, air, and moisture outside. Oxidation can change the molecule's structure within weeks, so an atmosphere of nitrogen or argon in the headspace extends usability. Personally, I like to squeeze out extra air with a shot of nitrogen before re-capping, especially for anything expensive or rare.
Keep the container in a cool, dry, ventilated cabinet. Ideally, use a flammable storage cabinet, because aldehyde volatility combines badly with sparks or heat sources. It’s not fear-mongering: one misstep with a volatile aldehyde and open flame, and you’re one bad day away from a shut-down. Temperature control slows down any autopolymerization. A fridge works in a pinch, but a dedicated temperature-controlled chemical cabinet sets the gold standard.
Preventing Problems Before They Start
In storage, the smallest oversight snowballs. Humid air sneaks in each time the cap’s left off. One time, I saw a student leave a bottle open for an hour while prepping glassware—guess what, the next user complained about a weird smell and stalling reactions. Never underestimate the power of using a bottle opener and closing it tight after every use.
Regular inspections matter. I run through shelves every couple of months, check for signs of cloudiness, color changes, or that sticky polymerized film around the cap. That’s how you spot trouble before it grows.
Responsibility and Safety in Practice
Keeping Material Safety Data Sheets (MSDS) handy makes sense beyond compliance. They give you exact flash points, vapor pressure figures, and spill response steps. Lab safety isn't some bureaucratic hoop—it's personal protection.
Chemical compatibility also matters. Storing 1,2,3,6-Tetrahydrobenzaldehyde next to strong acids, oxidizers, or bases invites reactions you never asked for. Physical separation saves you from emergency cleanups.
The Solution Starts With Habit
I learned that proper chemical storage isn’t just a technical issue—it’s about forming the right habits. Whether you’re a seasoned chemist or a new technician, treat every bottle as though your next project depends on it. Because, in my experience, it often does.
Is 1,2,3,6-Tetrahydrobenzaldehyde available in different grades or purities?
Getting Straight to the Point
Anyone dealing with 1,2,3,6-tetrahydrobenzaldehyde understands how purity affects results. People outside labs might just see it as a chemical name, but inside research and production facilities, choosing the right grade means everything. I remember an old mentor in my first research gig who laughed about “buying cheap, regretting expensive.” Looking back, his words ring true every time I see failed reactions traced to dodgy raw materials.
How Purity Shapes Performance
This compound pops up in specialty synthesis, fragrance chemistry, and pharmaceutical pathways. Each one demands a certain grade. In the fragrance world, any impurities, especially sulfur or lingering solvents, can wreck a batch. Synthetic chemists talk about 'selectivity.' If you want a product without byproducts, starting with high-purity material gives the best odds. Skimping leads to inconsistent yields and messy separations. In pharma, hard limits on impurities protect patient safety and production lines; batch recalls rarely make front-page news, but they cost millions and damage trust.
Sorting Out the Grades
Manufacturers usually offer a range of qualities—sometimes marked as technical, laboratory, or pharmaceutical. The technical grade often lands cheapest, but can carry byproducts and moisture. That's fine for cleaning applications or bulk intermediates, but not for fine-tuned synthesis. Laboratory grade raises the bar, keeping most other chemicals out. Drug makers and fragrance formulators chase the highest purities, often >98%, confirmed by gas chromatography or NMR so they don't have to second-guess what ends up in their reactors or bottles.
This split in quality isn’t marketing fluff. I’ve seen labs push for discounts on solvents only to pay later, re-running tests after contamination appeared. It shows up every time someone cuts corners in sourcing. On the flip side, insisting on certified purity keeps the paperwork heavy, but also builds reliability. Industry standards, like Good Manufacturing Practices (GMP), push suppliers to document every batch. Trust gets built slowly: you check certificates of analysis, trace raw materials—anything less invites disaster.
The Push for Even Cleaner Grades
Fresh challenges appear as regulations push out older, dirtier processes. There's a growing need for cleaner products, especially as analytical chemistry makes it easier to spot even trace impurities. A decade ago, only a few labs could check parts-per-million accuracy. Now almost anyone with the right equipment can flag contamination. That creates pressure: producers can't rely on technical grade when clients expect higher thresholds for safety or regulatory compliance.
What Can Be Done?
More transparency in sourcing builds faith in the supply chain. Buyers should demand detailed test records beyond generic grade labels. Stronger feedback from end-users can push suppliers to publish batch-to-batch differences, so nobody gets surprised by a missed impurity. As chemistry keeps evolving, better collaboration between suppliers and buyers set quality targets that match real-world needs.
If you ever struggle with inconsistent outputs, step back and look at the basics. Grade and purity aren’t just checkboxes. They’re the backbone of reproducible science and safe, effective products.
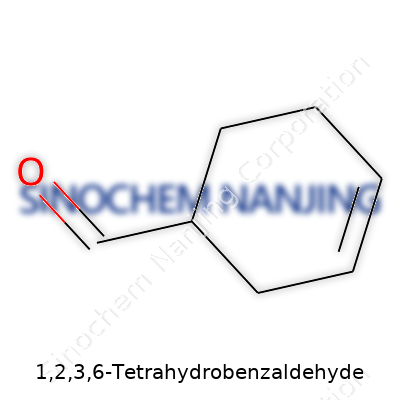
| Names | |
| Preferred IUPAC name | 3,4-Dihydro-2H-1-benzopyran-5-carbaldehyde |
| Other names |
Cyclohex-3-ene-1-carbaldehyde 3,6-Dihydrobenzaldehyde |
| Pronunciation | /ˈwʌn.tuː.ˈθriː.sɪksˈtɛ.trə.haɪ.droʊ.bɛnˈzæl.də.haɪd/ |
| Identifiers | |
| CAS Number | 626-19-7 |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:36639 |
| ChEMBL | CHEMBL227636 |
| ChemSpider | 202349 |
| DrugBank | DB08396 |
| ECHA InfoCard | 03d7bcf7-7137-4e99-91e4-9d3bdf99d218 |
| EC Number | 202-615-1 |
| Gmelin Reference | 107640 |
| KEGG | C02449 |
| MeSH | D007281 |
| PubChem CID | 10717 |
| RTECS number | CY8575000 |
| UNII | EM5L3RI9A2 |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID9064007 |
| Properties | |
| Chemical formula | C7H8O |
| Molar mass | 134.18 g/mol |
| Appearance | Colorless liquid |
| Odor | aromatic |
| Density | 0.994 g/mL at 25 °C (lit.) |
| Solubility in water | Slightly soluble |
| log P | 1.92 |
| Vapor pressure | 0.64 mmHg (25°C) |
| Acidity (pKa) | 12.11 |
| Basicity (pKb) | 13.42 |
| Magnetic susceptibility (χ) | -6.68 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.512 |
| Viscosity | 1.029 cP (25°C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 312.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -112.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3897 kJ/mol |
| Pharmacology | |
| ATC code | This substance does not have an ATC code. |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P264, P273, P280, P301+P312, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1,2,3,6-Tetrahydrobenzaldehyde: "2-3-0 |
| Flash point | 82 °C (180 °F; 355 K) |
| Autoignition temperature | 215 °C |
| Explosive limits | Lower explosive limit: 1.1%, Upper explosive limit: 6.8% |
| Lethal dose or concentration | LD50 (oral, rat): 2170 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1780 mg/kg (rat, oral) |
| NIOSH | DT3325000 |
| PEL (Permissible) | PEL: 2 ppm (skin) |
| REL (Recommended) | 5 mg |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Benzaldehyde Cyclohexanecarboxaldehyde Hydrocinnamaldehyde 1,2,3,4-Tetrahydronaphthaldehyde |

