Understanding 1,2,3,5-Tetrachlorobenzene: Its Journey and Impact
Historical Development
Chemists have worked with chlorinated benzenes for well over a century. 1,2,3,5-Tetrachlorobenzene started drawing attention during the mid-1900s as synthetic chemistry ramped up, especially after large-scale industrialization made aromatic compounds such a practical backbone for innovation. This compound, sitting among the highly chlorinated benzenes, came about through research into intermediates that could boost herbicide and pesticide production, given agriculture’s growing role. Since then, scientists have mapped its properties, and industries have shaped their handling protocols according to both its promise and its risks. Key scientific milestones include the documentation of its physical constants and the regulatory landscape that followed—driven not only by questions of productivity, but rising awareness of potential toxicity and persistence in the environment.
Product Overview
1,2,3,5-Tetrachlorobenzene looks like a white crystalline solid in pure form. In practice, manufacturers pay close attention to purity, as any impurities can alter downstream reactions or lead to regulatory issues. In laboratories and factories, it often ends up either as a starting material or as an intermediate product, rather than a key consumer product. Its main value comes from its function within the chemical process industry, especially where high chlorination is a sought characteristic.
Physical & Chemical Properties
This compound crystallizes easily, offering a melting point of about 136°C and a boiling point near 273°C under atmospheric pressure. Its density hovers around 1.6 g/cm³, notably heavier than water, reflecting the high chlorine content. Low solubility in water and strong solubility in organic solvents like ethanol or acetone means it often migrates in industrial effluent if spills occur. Its reactivity stays manageable under cold and dry storage, but both temperature and UV light can coax it into unwanted side reactions. These physical constants shape how plants store, handle, and dispose of waste streams, trying to keep stray emissions from reaching soil or waterways.
Technical Specifications & Labeling
Bulk shipments carry standardized labels, usually including the CAS number 634-66-2 and UN classification. Regulatory standards such as GHS or OSHA guidelines make it clear that handlers need clear hazard pictograms about both health and environmental risks. Specs cover both chemical purity and limits on unwanted isomers, as small differences there can change the product’s fate in a synthesis. High-quality product documentation lists melting and boiling points, molecular weight (215.9 g/mol), and recommended storage guidelines, calling out the dangers of exposure, volatility, and persistence.
Preparation Method
Factories often manufacture this chemical by direct chlorination of benzene or lower chlorinated benzenes under controlled conditions—usually with iron(III) chloride or aluminum chloride as catalysts and careful attention given to temperature and chlorine gas flow. Over-chlorination risks creating pentachlorobenzenes and hexachlorobenzenes, so process operators balance conversion efficiency and selectivity. Purification generally involves fractional distillation, crystallization, and sometimes column techniques, all aiming to separate isomers and remnant catalysts.
Chemical Reactions & Modifications
Industrial chemists value 1,2,3,5-Tetrachlorobenzene as a stable lattice for further functionalization. Its four chlorine atoms lend themselves to nucleophilic substitution, displacement reactions, and as anchor points for coupling with other organic molecules. In practice, the reactivity of the ring allows for the synthesis of specialty compounds, some useful as pesticides, others studied as potential flame retardants or intermediates for specialty materials. Reactions must be monitored closely, as excess energy or poor solvent choice can trigger polychlorinated byproduct formation, tough to deal with from both environmental and economic perspectives.
Synonyms & Product Names
Chemical suppliers often list 1,2,3,5-Tetrachlorobenzene under several synonyms, including Tetrachlorobenzene or 1,2,3,5-TCB. In technical catalogs, it sometimes appears as TCB-1235, highlighting the placement of the chlorines. For import/export documents and regulatory filings, all names trace back to the CAS registry number, sidestepping confusion in global trade.
Safety & Operational Standards
Exposure to 1,2,3,5-Tetrachlorobenzene poses both occupational health and environmental dangers. Workers need personal protective gear when handling the solid or dust—goggles, gloves, and filtered respirators. Facilities implement closed-system transfer and dust control to prevent inhalation and skin contact. Storage away from acids, oxidants, and sources of ignition or strong sunlight prevents breakdown and accidents. Environmental risk drives the adoption of scrubbers or carbon bed filters at vent stacks, while regulatory permits dictate containment measures, spill response, and wastewater treatment. Inspections and training for all staff remain essential, as even small lapses can trigger incidents or releases that attract both public scrutiny and regulatory penalties.
Application Area
The main buyers of this compound typically come from the agrochemical, specialty chemical, and dye industries. In these sectors, 1,2,3,5-Tetrachlorobenzene acts as an intermediate for high-value organochlorine derivatives or functions as a stepping-stone toward more elaborate molecules with specialized uses. Research teams sometimes test it as a reference standard in environmental monitoring or as a component in analytical studies, especially in tracking persistent organic pollutants. Less commonly, it appears in pilot projects connected to flame retardants or specialty coatings, but these uses have attracted extra regulatory reviews.
Research & Development
Academic and corporate laboratories continue to investigate alternatives to chlorinated benzenes, but this one often serves both as a model compound for environmental fate studies and as a benchmark in the search for more biodegradable options. Scientists have built up databases about its environmental persistence, breakdown pathways, and the mechanisms behind its toxicity. Efforts to design more selective synthesis routes also tie in closely with broader industry moves toward green chemistry, aiming for safer reagents, less waste, and milder reaction conditions. Research on encapsulation, targeted delivery, and rapid detection reflects a push to both maximize safety and reduce environmental footprint, aligning with both company goals and public demand for accountability.
Toxicity Research
Over the past few decades, toxicologists have highlighted risks tied to accumulation in fatty tissues, potential for long-term health impacts, and the danger it poses to aquatic life. Rodent studies show liver and kidney changes at high exposure doses, sparking limits on workplace air concentrations and emissions. Ecological studies point out the compound’s resistance to breakdown, raising alarms about bioaccumulation in the food chain and transformation into even more persistent or toxic compounds under certain environmental conditions. This evidence now filters straight into policy, setting rigid controls on both routine emissions and cleanup standards.
Future Prospects
Demand for safer, sustainable chemicals is pushing industries to rethink the role of chlorinated benzenes. Many funded projects are looking at alternative synthetic routes, biodegradable replacements, and advanced remediation for sites with historical contamination. Better real-time sensing for leaks and emissions means problems get caught earlier, reducing downstream harm. At the same time, regulatory frameworks continue to tighten, so R&D managers keep scanning for strict compliance and more efficient, closed-loop production technologies. If companies can leverage lessons from past incidents and channel investment into both cleaner production and robust safety standards, the chemical’s risks might trend downward, even as its essential niche in synthesis endures a little longer.
What is 1,2,3,5-Tetrachlorobenzene used for?
What Does 1,2,3,5-Tetrachlorobenzene Do?
1,2,3,5-Tetrachlorobenzene rings a bell mostly for those who’ve spent any time near industrial chemistry. Factories and labs produce it as a white-to-off-white crystalline solid. Its sharp, chemical smell is hard to ignore. While it’s not something people talk about over coffee, this compound holds a small but significant spot in chemical manufacturing.
Where Industry Puts It to Use
Through my years in science reporting, I’ve rubbed elbows with industrial chemists who see 1,2,3,5-tetrachlorobenzene mostly as an intermediate. They use it as a building block for more complex chemicals. It doesn’t usually land in finished products on its own. Instead, it steps in along production lines to help shape things like certain herbicides and pesticides. These end products keep farm fields clear and food growing strong, but that comes at a price: chemical residues and environmental impact.
This compound also helps in making dyes and pigments. Manufacturers rely on its molecular stability to keep colors brilliant and long-lasting. From textiles to plastics, the reach of colorants based on tetrachlorobenzene is easy to see in everyday life. I remember touring an old dye facility, where vats bubbled with potential, each batch using just a pinch of this chlorinated base for consistency in color.
Balancing Industrial Benefits and Health Risks
Companies value 1,2,3,5-tetrachlorobenzene for its consistent reactions. The flip side shows up in environmental safety reports. Over the past decade, scientists and health professionals noted its stubborn persistence in soil and water. It doesn’t break down easily. Studies from the EPA show levels of chlorinated benzenes—this one included—lingering in groundwater for years. The health risks include liver and kidney problems, and possible links to cancer after long exposure. I’ve read warnings urging strict storage and disposal. My own local water utility held community meetings to discuss trace contaminants, which underscores the public health piece here.
Reducing Harm: Practical Steps Forward
Safer handling has moved out of the scientific journals and onto the factory floor. Workers wear extra layers of gloves and suits now, and spill protocols look like military drills. Large-scale users recycle solvents and capture fumes both to cut waste and to guard against exposure. The EPA, along with European agencies, has pressed for newer alternatives that break down faster and threaten less environmental buildup.
Switching away from chemicals like this one won’t happen overnight. But in my reporting, I saw more companies investing in green chemistry—designing new production lines with fewer hazardous byproducts. Engineers and scientists join forces to create compounds that do the same job but clock out with less ecological debt. It’s a slow shift, though progress shows up most in updated regulations and transparency reports. Public pressure and consumer awareness push companies to look for replacements and keep community health in mind.
Final Thoughts
1,2,3,5-Tetrachlorobenzene isn’t a household staple, but it plays its part in bringing color and food security to daily life. As with many industrial chemicals, its benefits ride alongside environmental and human health concerns. Fact-based decision-making, improved safety measures, and smarter chemistry—these are helping keep risks contained while nudging industry toward safer futures.
What are the safety precautions when handling 1,2,3,5-Tetrachlorobenzene?
Understanding the Chemical: Risks at a Glance
1,2,3,5-Tetrachlorobenzene belongs to a group of chlorinated aromatic compounds used in research, specialty manufacturing, or sometimes as intermediates for other processes. Its history shows that it can harm both people and the environment. This stuff doesn't evaporate quickly, and it loves to stick around in soil and water. From what researchers and industrial workers have seen, direct contact with chlorinated benzenes can irritate skin, eyes, and lungs. It may cause vomiting, dizziness, or trouble breathing if inhaled or swallowed in large amounts. Chlorinated aromatic chemicals like this one have drawn plenty of attention from health and safety experts because their harmful effects often don’t show up right away.
No Shortcut: Personal Protective Equipment
Working with 1,2,3,5-Tetrachlorobenzene, no one can afford to skip safety gear. Gloves tested against organic solvents matter even if your job only lasts a few minutes. Nitrile or neoprene gloves block liquid from touching your skin. Splash goggles protect your eyes from splatter. Inhaling vapors causes problems for anyone with asthma or allergies—respiratory protection, especially a cartridge respirator with organic vapor filters, helps in poorly ventilated spaces.
Ventilation and Spills: Real-World Stories
In smaller labs, someone always thinks cracking a window counts as good ventilation. That mistake fuels emergency calls when fumes lead to nausea or headaches. Fume hoods or local exhaust systems make a lot more sense for toxic substances. I remember a colleague who knocked over a bottle of chlorinated solvent. Instead of panic, we had absorbent pads and a chemical spill kit nearby, so nobody rushed around searching for supplies. Instead of wet mops or paper towels, which only spread the danger, these kits soak up the liquid and keep contamination in check.
Storage: Don’t Just Stick It Anywhere
1,2,3,5-Tetrachlorobenzene calls for a specific spot. Store it away from acids, alkalis, open flames, and sunlight. Use containers with tight-fitting lids, preferably made from materials that don’t corrode. Every bottle deserves a clear label—the last thing anyone needs is a mix-up between this and something less toxic. Most facilities involve lockable cabinets and logging each withdrawal to track what moves where. If you're tempted to keep it next to a less hazardous solvent, think twice. Cross-contamination can turn a minor leak into a major hazard in seconds.
Disposal and Training
Waste tied to chlorinated benzenes lands in labeled, sealed containers set aside for hazardous pickup. Pouring leftovers down the drain sends poisons straight to the water table. I’ve seen far too many new staff act surprised by this rule. Regular training solidifies safe habits and raises flags when procedures seem vague or outdated. Instruction should cover cleanup, emergency washing stations, and symptoms of exposure that demand medical attention. Only with thorough, honest training do people take the risks seriously enough.
Community and Honesty
Chemical safety works best in places where people watch out for each other. Cataloging every bottle matters a lot less if someone hides near-misses or small splashes out of fear or embarrassment. Companies with strong safety teams and open reporting tracks have fewer injuries. Everyone shares the burden, so communication about procedures and exposure history stays clear. Honesty helps keep not just the work area safe, but also the neighbors and the local environment.
What is the chemical formula and molecular weight of 1,2,3,5-Tetrachlorobenzene?
A Closer Look at the Formula
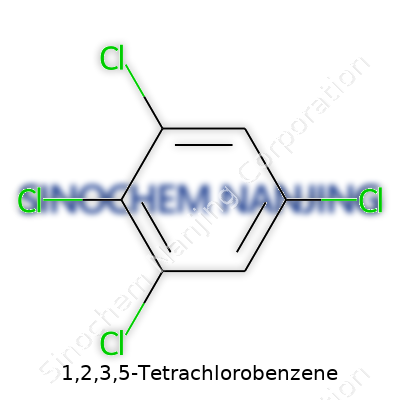
1,2,3,5-Tetrachlorobenzene isn’t some overcomplicated lab mystery. Its formula stands as C6H2Cl4. Pretty straightforward, just a benzene core dotted with four chlorine atoms. The trick lies in the name—those numbers tag the positions of each chlorine around the ring, shaping both its reactivity and place in the chemical world.
The story behind that formula is worth a pause. In a world full of similar-sounding compounds, knowing where each atom lands on the ring marks the difference between a persistent pollutant and an industrial building block. It’s not about nitpicking. One tweak in that structure, suddenly you get a molecule with different safety issues or a new set of environmental challenges.
Calculating the Molecular Weight
Molecular weight might look boring on paper, but in the lab, it means a lot. C6H2Cl4 adds up: carbon weighs in at about 12 grams per mole, hydrogen at roughly 1 gram, and chlorine packs on about 35.5 grams per mole. Do the math and you land right around 233.89 g/mol for 1,2,3,5-Tetrachlorobenzene.
Weight matters in every reaction. Misjudge it and your measurements tip off, your product quality drops, hazards increase. Too many graham crackers on your scales won’t bake a good cake. Chemicals work the same way—precise weights keep results real.
Uses and Concerns in the Real World
Look beyond the math, and this compound steps into interesting territory. Chlorinated benzenes, with their heavy formulae, find places as solvents, intermediates, and reference points for environmental studies. Yet, there’s no ignoring the baggage—persistence, potential for bioaccumulation, and long spells lingering in water or soil.
Years ago, I helped catch a mix-up during a shipping inspection where 1,2,3,5-Tetrachlorobenzene nearly swapped places with its isomer cousin. Without the right labeling based on chemical formula and weight, labs could have ended up dosing trials with the wrong compound. It taught me early that numbers and names on a drum aren’t just paperwork—they’re a lifeline for safety and credibility.
Precision Fuels Safety and Trust
Errors in chemical identity spell trouble. Global recalls, compliance nightmares, tarnished brands—missteps cost millions and, more importantly, trust. That’s why researchers, suppliers, and regulators all hammer on double-checking formulae and weights. Just like in my experience, a small slip could topple a whole process.
Tools help. Digital databases, barcoding, even good old periodic tables stuck to lab benches all work to keep identities clear. Training fresh chemists on what those subscripts and molecular masses mean isn’t busy work. It forms part of a bigger effort to keep the field honest and safe.
Regulatory bodies like the U.S. EPA and OECD keep the spotlights on compounds like this, cataloging every aspect from chemical structure to toxicity. Their work turns molecular weight and structure from dry facts into cornerstones of environmental health monitoring. Communities downstream from manufacturing plants rely on that accuracy for peace of mind. Their soil, their water, their air—all trace outcomes back to scientists getting the basics right with compounds such as 1,2,3,5-Tetrachlorobenzene.
Precision in chemical identity isn’t for show. From benchtop to supply chain, it forms the backbone of science, industry, and healthcare. The right numbers mean better results for everyone, from the folks mixing beakers to those living next door.
How should 1,2,3,5-Tetrachlorobenzene be stored?
Why Safe Storage Matters
For anyone who works in labs, manufacturing, or chemical storage, 1,2,3,5-Tetrachlorobenzene stands out as one of those chemicals that makes you double-check your process. It’s rarely seen outside industrial or research settings, but that doesn’t mean its risks are any less real. With strong evidence pointing to skin irritation, respiratory distress, and long-term health effects, this isn’t a chemical to be left unattended or stashed in any old corner.
The Basics Behind Storage Practices
Glass, steel, or high-density polyethylene containers provide the best defense here, because tetrachlorobenzenes break down—just not fast—and can leach through more porous materials. From experience, using anything else invites ruined products, or worse, accidental exposure. I learned quickly during my own stint in chemical research that cheap containers only add headaches. Once, a cracked plastic jug contaminated a whole bench before anyone noticed.
Pick containers with tight-fitting lids. Keep everything clearly labeled. It sounds like a given, but everyone overlooks this once or twice. One wrong grab, or mixing up compounds, and you’re facing a clean-up or even a chemical reaction you never saw coming.
Environment Matters More Than Most Think
Heat makes just about every chemical act up faster. Keep 1,2,3,5-Tetrachlorobenzene in a cool, dry area—usually under 25°C. Direct sunlight shortens shelf life, sometimes causing containers to weaken or degrade. Away from windows, thermostated storage rooms work well. Never store it close to heaters or warm machinery.
Humidity can creep in through lids or seals, compromising purity. Damp storage spaces also lead to rust or corrosion, even for products housed in sturdy drums. Every bottle I've cracked with a rusty rim made me nervous—I never skimp on proper conditions now.
Keep Incompatibles at Bay
Strong acids, strong bases, and oxidizing agents spell out a recipe for disaster. Keep them well-separated. I've seen storage mistakes where incompatible products sat side by side, either because extra space ran out or because a staffer didn't know the risks. Segregating chemicals, even within locked cabinets, pays off every time.
Dedicated shelving for chlorinated benzenes, with barriers between other chemical families, lowers risk. Accidental spills or container breaches don’t trigger chain reactions nearly as easily if chemicals are kept apart by shelves, bins, or even entire rooms.
Ventilation and Fire Safety in Real-World Scenarios
Fumes from tetrachlorinated benzenes don’t just smell foul; they can linger in closed spaces and cause chronic headaches or worse. Good ventilation means more than just a fan in the window. Fume hoods or proper air exchange systems keep the workplace safer. In the event of a spill, it’s the difference between a quick cleanup and an evacuation.
Keep fire extinguishers within reach and develop response protocols for everyone involved. Chlorinated organic compounds can feed fires with toxic fumes. Training staff, running regular drills, and reviewing emergency materials can all lower risk—far more than crossing your fingers and leaving it to chance.
Training: The Anchor of Safe Storage
All the right containers and climate controls don't help if people get careless. Bringing everyone up to speed with hazard communication, including reading Safety Data Sheets and knowing what PPE to use, is non-negotiable. I always saw fewer mistakes at facilities that focused on routine educational sessions than those with fancy equipment and little training. Safe storage doesn't just live in labels—it's part of a living culture.
Is 1,2,3,5-Tetrachlorobenzene hazardous to health or the environment?
Understanding the Chemical’s Footprint
1,2,3,5-Tetrachlorobenzene doesn’t ring bells for most folks. It exists in industrial settings, not the average home. Workers run into it while making other chemicals, especially in plants that produce herbicides or insecticides. I've seen reports that paint it as a technical ingredient, not as something we choose to invite into the daily routine.
This compound, unlike something found in nature, ends up in soil or water when manufacturing isn’t well-controlled, or if products leak or get dumped. Once it gets into the environment, it sticks around. It doesn’t break down quickly, and wildlife picks it up. Fatty tissues in fish and birds soak up more of these chlorinated benzenes than folks advertise. That bioaccumulation starts at the bottom and shows up on dinner plates higher up.
Health Hazards for Workers and Communities
Inside a chemical plant, exposure risk isn’t just dust or fumes. The material slides onto surfaces, gets on skin, mixes into the air. Inhaling or touching this compound makes people sick over time. Industrial hygiene studies, mostly in high-production factories, connect it to headaches, sore throats, coughing, and more worryingly, impacts on the liver and kidneys. Chronic contact can stack up damage. Lab rats suffer organ damage with long-term exposure, and that's not a comforting finding for humans in similar conditions. The real punch comes from its similarity to banned polychlorinated biphenyls (PCBs). Both can disrupt hormones and potentially lead to cancer.
Communities living near chemical plants or places where hazardous waste sits uncovered feel the effects too. Surface water runoff and wind take these pollutants far from their source. Studies from contaminated towns show higher rates of health problems, some of which point toward persistent organochlorines like tetrachlorobenzenes. Everyday folks rarely have enough say in whether something dangerous leaves a factory fence line. People rarely realize the link between unexpected illness clusters and something as hard to pronounce as 1,2,3,5-Tetrachlorobenzene.
Why It Matters Beyond the Lab
The chemical’s stubbornness once released—its tendency to linger in mud, water, or dust—pushes risk forward across generations. Kids playing near contaminated spots take in more pollutants by weight than adults. Sometimes the rush to produce agricultural chemicals leaves waste management as an afterthought, leading to unexpected land and water contamination decades later. No one wins when polluted soil ruins farmland or water sources.
Drawing a Line on Exposure
Completely stopping its use overnight proves nearly impossible. Industrial demand for pesticides isn’t fading. Safer alternatives make a big difference, and industries should genuinely consider greener chemistry whenever possible. Facilities that use or make 1,2,3,5-Tetrachlorobenzene should share emissions data for all to see, not just stuff it in reports. Public databases, open meetings, honest risk communication—these steps matter more than most realize. Regulators can increase fines and tighten monitoring for chronic polluters. Workforce safety, with better training, gear, and exposure tracking, needs to be more than a checkbox. Health clinics near chemical hubs deserve extra support, including tracking subtle changes in community health over time.
Getting rid of the stockpiles hidden in old warehouses or buried in aging landfills looks daunting but necessary. Safe disposal, not just moving toxins elsewhere, is the only way out. It’s on everyone who builds, buys, or regulates these chemicals to keep the risks from multiplying. Once released, these problems rarely stay local or short term.
| Names | |
| Preferred IUPAC name | 1,2,3,5-Tetrachlorobenzene |
| Other names |
1,2,3,5-Tetrachlorobenzen Benzene, 1,2,3,5-tetrachloro- Tetrachlorobenzene UN 2321 |
| Pronunciation | /ˌwaɪ.tuː.θriː.faɪv.taɪˌtrəˌklɔː.rə.bɛnˈziːn/ |
| Identifiers | |
| CAS Number | 634-66-2 |
| Beilstein Reference | 1904782 |
| ChEBI | CHEBI:34684 |
| ChEMBL | CHEMBL22737 |
| ChemSpider | 12034 |
| DrugBank | DB13335 |
| ECHA InfoCard | ECHA InfoCard: 100.005.191 |
| EC Number | 203-989-2 |
| Gmelin Reference | Gmelin 7748 |
| KEGG | C08310 |
| MeSH | D014429 |
| PubChem CID | 7243 |
| RTECS number | CZ9625000 |
| UNII | NCI15WZ6M1 |
| UN number | UN2320 |
| CompTox Dashboard (EPA) | DJ3BB9KGGV |
| Properties | |
| Chemical formula | C6H2Cl4 |
| Molar mass | 215.89 g/mol |
| Appearance | White crystalline powder |
| Odor | Aromatic |
| Density | 1.68 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.8 |
| Vapor pressure | 0.04 mmHg (25°C) |
| Acidity (pKa) | Acidity (pKa): ~-2.0 |
| Basicity (pKb) | Non-basic |
| Magnetic susceptibility (χ) | −85.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.578 |
| Viscosity | 1.2 mPa·s (25 °C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 323.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -12.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1607.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H410 |
| Precautionary statements | P261, P264, P270, P271, P273, P301+P312, P304+P340, P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | Flash point: 138°C |
| Autoignition temperature | 380 °C |
| Lethal dose or concentration | LD50 oral rat 2830 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1,2,3,5-Tetrachlorobenzene: 3000 mg/kg (oral, rat) |
| NIOSH | SW3500000 |
| PEL (Permissible) | 1 ppm (10 mg/m³) |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
1,2,3,4-Tetrachlorobenzene 1,2,4,5-Tetrachlorobenzene Pentachlorobenzene Trichlorobenzene Hexachlorobenzene |

