1,2,3,4-Tetrachloronaphthalene: An In-Depth Look
Historical Development
Back in the early days of 20th-century industrial chemistry, scientists started exploring naphthalene derivatives for their stability and resistance to breakdown. The push for more robust insulating materials, lubricants, and dielectrics in electrical and mechanical engineering set off a wave of research that led to the synthesis of polychlorinated naphthalenes like 1,2,3,4-tetrachloronaphthalene. Laboratories and chemical companies recognized chlorination’s impact on increasing resistance to chemical degradation, which helped boost production in industries focused on utility over ease of degradation. With more electric grids and combustion engines around, manufacturers leaned on these compounds for their specialized qualities, but knowledge about their risks lagged far behind enthusiasm for their commercial benefits.
Product Overview
1,2,3,4-Tetrachloronaphthalene forms one component in the mix of polychlorinated naphthalenes known to industry. It stands out with four chlorine atoms bonding to the naphthalene core, giving it greater persistence and toughness on both a molecular and macro scale. Workers across different sectors—insulation, electric manufacturing, chemical intermediates—found themselves handling materials with it mixed in. Given the push in the 1960s and 1970s for better-performing flame-resistant and chemical-resistant substances, its role expanded in both formulation and end-use applications, including specialized coatings, some lubricants, lab reagents, and previously in capacitor dielectrics. Its broad profile connected it to many practical industrial problem-solving efforts.
Physical & Chemical Properties
As a white or pale-yellow crystalline solid, 1,2,3,4-tetrachloronaphthalene resists most acids, bases, and oxidizers. Its structure gives it a melting point comfortably above many everyday plastics, making it popular for blends and formulations that get exposed to heat or electrical arcing. Poor water solubility but strong affinity for organic solvents characterizes it—anyone trying to wash it off with water usually failed, but certain solvents do the job. Those chlorines crank up its thermal stability and decrease its reactivity toward most standard reagents. In the air, it can settle on surfaces and linger for months; this makes lab clean-up or industrial spills a true hassle for anyone on duty.
Technical Specifications & Labeling
Working in chemical inventory or regulatory compliance brings a list of technical details. Purity levels often clock in over 97%, and the labeling must match international guidelines, with hazard pictograms and warnings about skin contact, ingestion, and environmental toxicity. Precise molecular weight clocks in at 247.95 g/mol, though most product labels also highlight its boiling and melting points, and detail appearance for quality checks. Regulatory needs mean a clear CAS number—this anchors compliance checks and data tracking. Imports and exports face paperwork that confirms origin, hazard category, appropriate use, and proper disposal channels, as chemical safety authorities in many countries track potential hazards connected to persistent organic pollutants.
Preparation Method
Industrial production has relied on direct chlorination of naphthalene under controlled pressure and temperature, using iron or aluminum chloride as a catalyst. The process demands a tightly controlled environment to manage exothermic reactions, hazardous chlorine gas, and the creation of side products. Batch reactors commonly handle the synthesis, and staff monitor for temperature spikes to prevent unwanted explosions or leaks. After chlorination, distillation handles separation and purification, while further refining polishes the final product for industrial buyers. Facilities regularly retrofitted older setups with more advanced scrubbers and off-gas management, as environmental awareness ramped up over the past decades.
Chemical Reactions & Modifications
Chemists attempting modifications often tackle nucleophilic substitutions or oxidation, but those four chlorines mean reactions need robust conditions and sometimes exotic reagents. Trying to break down the molecule remains an uphill struggle—nature’s own processes grind on for decades to get even partial degradation. In research, scientists assess shifts in reactivity that follow from swapping out different ring positions or replacing chlorines with other halogens. These procedures uncover minute changes in physical properties, opening the door to new pathways for dechlorination, which might prove key for remediation and reducing toxicity.
Synonyms & Product Names
Different catalogs and chemical suppliers tag 1,2,3,4-tetrachloronaphthalene with an assortment of names. Some list it simply as “tetrachloronaphthalene,” while specialized papers use names reflecting IUPAC conventions. Other trade names arise in old engineering catalogs—those surface from time to time on spare parts or archived documents. Researchers and lab staff track synonyms to navigate regulatory controls and label cross-checks, avoiding surprise issues in purchasing, handling, or waste disposal.
Safety & Operational Standards
Industry veterans remember the days of casual handling, but modern standards require gloves, goggles, fume hoods, and rigorous accounting for every gram brought into a plant or lab. Inhalation or skin exposure risks draw immediate response protocols, with material safety data sheets stressing medical attention if someone takes a hit. Spill kits, air scrubbers, and waste drums shaped by hazardous waste criteria fill storerooms for routine handling. Governments, especially in the US, EU, and Japan, place intense scrutiny on persistent compounds, forcing chemical operations to adopt best practices—tracking batch numbers, certifying safe transfer, and constantly checking compliance with local law.
Application Area
Tooling up factories for insulation, transformer production, or advanced resins meant working with 1,2,3,4-tetrachloronaphthalene. Its chemical and heat resistance pushed its use in coatings that stop corrosion and loss of performance at high temperatures. Old electrical equipment sometimes still contains it; repair and recycling teams often bump into it decades later, needing special protocols for teardown and reclamation. Some specialized research applications dip into the compound for benchmarking new dechlorination pathways or building calibration standards for pollutant analysis in contaminated soils and waters.
Research & Development
Academic labs and industrial R&D teams increasingly chase ways to break down chlorinated naphthalenes or substitute safer materials in legacy applications. Research pivots toward greener chemistry—using bio-based catalysts, less toxic synthesis pathways, and clean-up technologies like photolysis or advanced oxidation. Surface chemistry and analytical detection teams build new standards using tetrachloronaphthalene for contamination tracing, giving insight into legacy pollution and new production streams. Cross-disciplinary projects pair chemists with toxicologists, engineers, and remediation specialists, pushing faster solutions to minimize impact on people and planet.
Toxicity Research
Toxicologists track its pathway through the body, finding persistent buildup in fatty tissues, potential for liver and immune system disruption, and links to developmental harm in animal models. Workers with repeated exposure show increased risk of skin, lung, and liver issues. Regulatory reviews regularly warn about developmental and reproductive toxicity; hands-on cases—like chemical plant spills or improper disposal—spark local studies linking elevated exposure to higher rates of specific cancers or birth defects. This history has forced many manufacturers to switch to alternatives or overhaul protective gear and training on the shop floor.
Future Prospects
With growing regulatory restrictions and deeper awareness of ecological safety, new production of 1,2,3,4-tetrachloronaphthalene finds itself squeezed to specialty applications, usually tightly controlled. Environmental scientists test new breakdown methods—engineered enzymes, photocatalysts, or innovative thermal destruction—aiming to clean up contaminated sites. Some companies have invested heavily in tracking old stockpiles and phasing out legacy equipment. While substitution work chugs along, the compound’s name stays on government watch lists, pushing funding and lab time toward remediation, environmental monitoring, and advanced detection. Over time, practical solutions and tighter laws look to cut risks and shift industry habits toward lower-toxicity materials.
What is 1,2,3,4-Tetrachloronaphthalene used for?
Where You’ll Find 1,2,3,4-Tetrachloronaphthalene
1,2,3,4-Tetrachloronaphthalene doesn’t show up in daily conversation, but it’s worth paying attention to, especially considering its industrial roots. This chemical belongs to a group called chlorinated naphthalenes, which chemists first turned to during the 20th century for practical reasons. It can survive harsh conditions, high temperatures, and exposure to all sorts of other chemicals. Still, that strength often brings risks along with benefits.
Why Industry Chose It
Factories started using 1,2,3,4-tetrachloronaphthalene for two main jobs: insulation and lubrication. In electrical gear, old timers valued these chemicals because they kept transformers cool and wires well protected. That heat resistance mattered as machines grew stronger and more complex. I used to work at a plant where we handled old switchgear, and seeing how these oils kept things running smoothly was impressive—until you looked at the safety manual.
Besides electrical work, companies put this chemical in lubricants for heavy-duty machines—think large presses, weaving looms, turbine engines. The makers liked it because it didn’t break down easily. This meant you could run equipment longer between oil changes and service stops. As someone who’s spent time in maintenance shops, I appreciate fewer breakdowns. You could handle twice as much work with half the fuss. Unfortunately, this durability comes with a price.
Environmental Cost and Safety Concerns
After years of use, the evidence piled up: chlorinated naphthalenes make a mess in the environment. Substances like 1,2,3,4-tetrachloronaphthalene resist breaking down in soil and water. They can end up in wildlife and, eventually, move up the food chain. The EPA and other agencies have flagged these chemicals as persistent organic pollutants. I remember reading reports that mentioned how even small spills stuck around for decades. Add the health warnings—possible liver problems, effects on the nervous system, skin issues for workers—and it gets even harder to overlook the downside.
Rethinking Old Habits
Regulators started clamping down in the 1970s and 80s, and many countries put strict rules on importing, producing, and handling chlorinated naphthalenes. In some places, these chemicals didn’t just face regulation—they were outright banned. These days, companies zero in on removing or safely storing the old stuff left behind in transformers or industrial tanks. From personal experience, the challenge isn’t just swapping out the chemicals. Cleaning up past spills or leaks calls for specialized crews, protective gear, and a good budget. Nobody wants to cut corners where lives and ecosystems are on the line.
Alternatives and Solutions
Modern industries keep searching for safer replacements. Synthetic oils, silicone-based fluids, and biodegradable lubricants now do the heavy lifting. None of them solve every problem, but each step away from toxic legacy chemicals counts. Sharing data between companies, following best practices, and investing in green tech help prevent the next round of chemical headaches. By learning from past mistakes, manufacturing doesn’t have to pick between performance and the planet.
Is 1,2,3,4-Tetrachloronaphthalene hazardous to health?
The Reality Behind a Chemical Name
1,2,3,4-Tetrachloronaphthalene doesn’t sound familiar to most people. It’s not on the tip of anybody’s tongue, but its chemical cousins and industrial use bring it closer to our lives than we think. This substance belongs to a group of chlorinated naphthalenes that have been around since the 1930s. Used mainly in electrical insulators, dyes, cable coatings, and lubricants, these chemicals show up where industrial legacy lingers. It looks like a waxy solid and doesn’t dissolve well in water, which means it can stick around on surfaces and in the environment for years.
What Happens in the Body?
Let’s get real about exposure. If someone breathes in dust, touches contaminated surfaces, or comes across old transformers or waste, 1,2,3,4-Tetrachloronaphthalene can enter the body. Lab studies with chlorinated naphthalenes suggest these substances build up in fat tissue and may hang around in the liver or kidneys for long periods. This tendency to stick with us can push the risks higher, especially if a person works in old manufacturing plants or handles outdated equipment.
The Health Evidence
The biggest concerns come from studies with industrial workers and animal lab tests. Breathing these chemicals or absorbing them through the skin leads to problems like chloracne—a severe skin condition that looks a lot like acne but runs much deeper. Workers exposed for months or years reported issues with their liver and immune system as well. Animal studies didn’t paint a better picture: researchers noticed liver enlargement and signs of toxicity, along with reduced fertility and changes in organ weights. The International Agency for Research on Cancer lists some chlorinated naphthalenes as possible carcinogens. Though data is still developing, these findings point to the need for serious caution.
Environmental Trouble
This chemical hardly breaks down outdoors. It can travel through soil and water, landing far from the original source. What’s disturbing is how it accumulates in the fatty tissues of animals, climbing the food chain and reaching people through fish, dairy, or meat. Research groups tracking polychlorinated compounds in river sediments and wildlife find traces decades after the last major use. Children living near contaminated sites risk eating or breathing contaminated dust—an early pathway for harm in communities that didn’t ask for it.
Solutions: What Can Be Done?
Laws in the US and Europe already sharply restrict production and disposal of chlorinated naphthalenes. Still, old landfill sites, industrial dumps, and outdated equipment sit out there, forgotten in some corners. Cleaning up—removing contaminated soil, incinerating hazardous waste, and monitoring old sites—helps bring down exposure risks. Workers handling the chemical need strong protective gear and clear rules on safe handling. People living near legacy pollution sites deserve regular updates, soil testing, and medical screening if contamination is found. When companies make decisions about disposal or cleanup, open communication builds trust and lets everyone play a role in protecting health.
Public Awareness and Practical Choices
Responsible action doesn’t just fall to regulators. Knowing what’s in old building materials, transformers, soil, or water near industrial sites helps families and community groups make safer choices. Reporting any signs of pollution or chemical dumping keeps attention on the issue before small spills grow into long-lasting damage. Simple steps—washing hands, cleaning dust at home, and pressing for cleanups—offer protection on the ground level. As scientists gather more information, everyone deserves up-to-date facts to guide decisions and protect future generations from the mistakes of the past.
What are the storage requirements for 1,2,3,4-Tetrachloronaphthalene?
Understanding the Chemical
1,2,3,4-Tetrachloronaphthalene comes from a broader group known as polychlorinated naphthalenes. These substances played roles in older industrial products. Because of persistent and toxic traits, many countries ban or restrict their use. Anyone working with this chemical needs a clear plan to manage its hazards and store it without taking chances.
Risks Worth Considering
From personal experience in a lab setting, few things cause as many headaches as poorly stored hazardous chemicals. 1,2,3,4-Tetrachloronaphthalene releases harmful dust and fumes, especially during spills or if exposed to high heat. Skin contact brings irritation, and inhalation may spark much worse health effects. NIOSH and OSHA emphasize keeping human contact close to zero for a reason. Poor storage habits can lead not only to contamination but to regulatory fines or worse if health impacts show up later.
Best Practices for Safe Storage
It’s easy to get lazy on storage, but this is one compound where sloppiness bites back. Store 1,2,3,4-Tetrachloronaphthalene in a spot where temperature stays cool and steady — think between 2°C and 8°C for most labs, keeping it well away from sunlight and sources of ignition. Too much heat, or even direct light, could break down the substance or increase pressure inside the container. Acidic or alkaline materials sitting too close add unnecessary risk; this chemical may react with both, causing dangerous byproducts.
Choose containers with tight-fitting, chemical-resistant lids — glass with PTFE seals or properly rated HDPE keep leaks to an absolute minimum. A loosely capped bottle in a humid storeroom builds more risk than many realize; fumes can build up, causing not just a workplace hazard but environmental headaches if contamination happens. Keep the original label visible, with all hazard and handling information easy to read, so no one grabs it by mistake or misjudges what’s inside.
A Safe Storage Space
Calls for ventilation might sound repetitive, but inside a chemical storeroom, there’s no such thing as too much air movement when it comes to toxic organics. A mechanical exhaust hood or vented cabinet can keep levels of airborne vapors down, especially if stored in quantity. Store away from exits, aisles, and any area with heavy movement to prevent accidental knocks, and always use secondary containment — a spill tray, for example — to catch surprise leaks.
Response to Spills or Leaks
If something sparks a spill or you notice a leak, have basic spill cleanup gear nearby: gloves made for organics, eye protection, and an approved absorbent. Waste should never go down the drain. It needs to be collected by licensed hazardous waste handlers to avoid harm to water or soil. Keeping that cleanup kit on the same shelf, not buried in another room, makes the difference between managing a small mess and facing a larger disaster.
Improving Safety and Compliance
Staff training really pays off over time. Everyone working near hazardous chemicals should refresh their knowledge twice a year, not just during onboarding. Written records, regular locker checks, and audits from outside experts help catch small lapses before they become big problems. Responsible storage doesn’t just follow the rules; it builds a culture where everyone feels confident that their health and environment matter just as much as productivity.
How should 1,2,3,4-Tetrachloronaphthalene be handled safely?
Why Safety Isn’t Optional
Anyone working around 1,2,3,4-Tetrachloronaphthalene doesn’t need a chemistry degree to know it brings real risk. This chemical lands on hazardous substance lists for a reason. Just catching a whiff from an open container can irritate the nose, throat, and eyes. Spilling a drop on bare skin isn’t something you’ll forget quickly. Inhaling dust or fumes in a closed shop isn’t only uncomfortable—it’s a potential health disaster.
My own time in industrial settings taught me to respect any compound with a long, tongue-twisting name—especially one flagged by multiple safety agencies. 1,2,3,4-Tetrachloronaphthalene has been linked with skin rashes, liver problems, and possible reproductive difficulties after frequent, heavy exposure. Regulatory guidance from places like OSHA and the CDC stress how these risks can creep up. That’s not fearmongering. Stories float around about tanks leaking in older facilities and emergency crews stepping in after workers report burning eyes and difficulty breathing. People learn the hard way: short-term convenience never outweighs long-term health.
Practical Safety You Can Trust
Gloves, goggles, and solid work gear should always form the first line of defense. Not the cheap throwaway stuff, either. Nitrile gloves—heavy grade—hold out against leaching. Face shields or snug safety glasses stop splash hazards. It isn’t overkill to use a lab coat or apron that you leave outside the break room; the tiniest residue can spread onto lunch and into homes.
Ventilation makes a big difference too. Fume hoods and local exhaust fans cut the chance of breathing in dangerous vapors. Open windows and floor fans won’t cut it. Closed systems work best whenever someone transfers, mixes, or measures the chemical—spills and fumes travel fast. Regular equipment checks keep unexpected leaks from turning into full-blown accidents. I’ve seen what happens when workers ignore signs of wear on gaskets or ducting—one loose fitting can cost someone a hospital trip and weeks off the job.
Training Makes a Difference
Those annual safety meetings aren’t just a legal box to tick. New hires, old pros—everyone should practice how to handle emergencies. Knowing how to wash a spill off skin or eyes without panicking matters more than memorizing a material safety data sheet. Active drills prepare teams to work through chemical releases, not just talk about them. The best-run companies don’t treat safety as a chore. They build it into daily routines: clear labels, spill kits in every room, everyone empowered to call out unsafe moves before they become accidents.
Disposal and Environmental Care
Treating waste as trash creates more problems. 1,2,3,4-Tetrachloronaphthalene’s impact on soil and water keeps environmental regulators on edge. Pouring leftover chemicals down the drain or tossing contaminated rags into regular garbage spreads it far beyond the shop floor. The right way finds hazardous waste contractors who know how to neutralize and handle the material without shortcuts. Secure storage in sealed drums and sharp tracking limit the risk of leaks. My opinion? Err on the side of caution. Cleanups cost far more than careful disposal ever will. One carelessly handled drum can poison an entire section of groundwater. Local communities carry the burden long after factory workers go home.
Looking Forward
Avoiding harm from 1,2,3,4-Tetrachloronaphthalene isn’t just for compliance. It’s about respect—for coworkers, the environment, and personal wellbeing. Investing in strong gear, good routines, and professional waste partners means fewer regrets down the line. Real safety runs deeper than any checklist—it comes from everyone deciding, together, that nobody wants to be the subject of tomorrow’s accident story.
What is the chemical formula of 1,2,3,4-Tetrachloronaphthalene?
Chemical Structure and Formula Matters
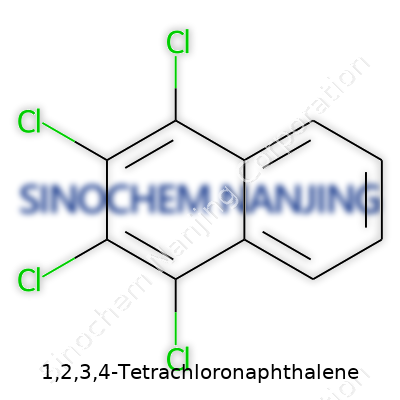
Naphthalene pops up in plenty of science textbooks, especially in those chapters about aromatic hydrocarbons. Tossing chlorine atoms onto a naphthalene ring gives birth to a compound like 1,2,3,4-tetrachloronaphthalene. The chemical formula tells you how many of each atom settle into the molecule. Figure out the parent structure—in this case, naphthalene—which has the formula C10H8. Each added chlorine atom takes the place of one hydrogen atom. Swap four hydrogens for four chlorines, and the resulting formula lands as C10H4Cl4.
Environmental Footprint and Human Health
Not many people chat about tetrachloronaphthalenes over breakfast. Still, the real-world impact shouldn’t fly under the radar. PCNs—or polychlorinated naphthalenes—have shown up in everything from electrical insulators to lubricants. These chemicals stick around a long time in the environment. Because of that, they don’t just fade away; they accumulate in soil, water, and even in the food chain. Studies in places like the European Union have listed them as "persistent organic pollutants." Once they make their way into the environment, getting rid of them turns into a headache.
Why Accuracy Counts in Chemical Formulas
High school chemistry taught me that skipping steps comes back to bite you. Get even one digit wrong in a chemical formula, and lab results end up useless. Regulatory filings need spot-on details—think C10H4Cl4, not some vague approximation. Mistakes could mean hazardous substances go unnoticed or unreported. Industry professionals run safety checks, companies draw up usage instructions, and researchers develop new materials based on these exact formulas. In my own work, chemical mistakes have delayed entire projects or—worse—put teams at risk due to misidentified substances.
Balancing Innovation and Caution
Old industrial applications sometimes glossed over health and environmental risks. PCNs were once prized for heat resistance and chemical stability. Today, we know these features allow them to linger where they shouldn’t. Regulatory bodies—think EPA, ECHA, and more—have stepped up to manage their use. Data from legacy manufacturers highlight the importance of knowing exactly what’s in a chemical blend. Leaving out one component, or listing the wrong one, skews toxicity profiles and the path for clean-up or destruction.
Better Monitoring and Safer Alternatives
Tracking harmful substances starts with basic data—formulas, structures, uses, and exposure routes. In workplaces where chemical substances are common, programs for monitoring air and water help keep people safer. Alternatives to PCNs have started to take hold, but their effectiveness only matches the original if industries have accurate records on all parts—starting with simple chemical formulas. My previous involvement in an environmental testing lab showed me how accurate identification of compounds is essential. Decisions about cleanup and remediation sometimes move forward—or get stuck—based on nothing more than the accuracy of a chemical formula on a label or test result.
Learning From the Past
Experience in research and environmental health makes me wary of any chemical that has a complicated name and a history of persistence. Knowing that 1,2,3,4-tetrachloronaphthalene carries the formula C10H4Cl4 isn’t just an academic exercise. This identification shapes how industry, regulators, and the public treat this compound moving forward. The details lay the groundwork for both daily workplace safety and bigger-picture environmental policy.
| Names | |
| Preferred IUPAC name | 1,2,3,4-tetrachloronaphthalene |
| Other names |
Tetrachloro-1,2,3,4-naphthalene 1,2,3,4-Tetrachlornaphthalin |
| Pronunciation | /ˌwʌn tuː θriː fɔːr ˌtɛtrəˌklɔːrəˈnæfθəliːn/ |
| Identifiers | |
| CAS Number | 1335-88-2 |
| Beilstein Reference | 1362207 |
| ChEBI | CHEBI:82144 |
| ChEMBL | CHEMBL227672 |
| ChemSpider | 16211301 |
| DrugBank | DB16084 |
| ECHA InfoCard | 04c504c7-6692-4469-a1d3-310e5cc0cc83 |
| EC Number | 204-308-2 |
| Gmelin Reference | 171943 |
| KEGG | C14383 |
| MeSH | D014445 |
| PubChem CID | 15138 |
| RTECS number | QJ5950000 |
| UNII | GF526020S8 |
| UN number | UN2202 |
| CompTox Dashboard (EPA) | DTXSID3049179 |
| Properties | |
| Chemical formula | C10H4Cl4 |
| Molar mass | 290.91 g/mol |
| Appearance | White solid |
| Odor | aromatic |
| Density | 1.61 g/cm³ |
| Solubility in water | insoluble |
| log P | 4.8 |
| Vapor pressure | 0.00016 mmHg at 25°C |
| Acidity (pKa) | 4.90 |
| Magnetic susceptibility (χ) | -0.000085 |
| Refractive index (nD) | 1.6390 |
| Viscosity | 2.7 cP (20°C) |
| Dipole moment | 2.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 285.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 211.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5157.7 kJ/mol |
| Pharmacology | |
| ATC code | V19CB15 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause respiratory irritation, suspected of causing cancer, toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS08, GHS09 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H351, H410 |
| Precautionary statements | P260, P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P312, P330, P362+P364, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1,2,3,4-Tetrachloronaphthalene: "2-2-0 |
| Flash point | 104°C |
| Autoignition temperature | 550 °C |
| Lethal dose or concentration | LD50 (oral, rat): 2100 mg/kg |
| LD50 (median dose) | LD50 (median dose): >11 gm/kg (oral, rat) |
| NIOSH | PY35700 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,2,3,4-Tetrachloronaphthalene is "0.1 mg/m³ (OSHA TWA)". |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Naphthalene 1,2,3,4-Tetrachlorobenzene Polychlorinated naphthalenes 1,2,3,4-Tetrachlorodibenzofuran |

