1,2,3,4-Tetrachlorobenzene: A Comprehensive Commentary
Historical Development
The story of 1,2,3,4-Tetrachlorobenzene stretches back to the early days of synthetic organic chemistry, in a time when the chemical industry expanded rapidly on the backbone of chlorination techniques. Chemists, eager to uncover the properties of aromatic hydrocarbons, found that the halogenation of benzene produced compounds with new physical and chemical characteristics. By the late 19th and early 20th centuries, as industrial demand for chlorinated aromatic compounds climbed, especially in the production of pesticides, dyestuffs, and specialty intermediates, the synthesis of all possible chlorobenzene isomers became widespread. Among them, 1,2,3,4-Tetrachlorobenzene stood out as a challenge in terms of pure isolation and identification, owing to the similarities it shares with other isomers. Its commercial relevance did not truly emerge until industrial-scale chlorination methods took hold, making it accessible as a technical-grade product in both research and manufacturing sectors.
Product Overview
This aromatic compound, built from a benzene ring decorated with four chlorine atoms at the 1, 2, 3, and 4 positions, belongs in a family known for its stability and reactivity in different contexts. 1,2,3,4-Tetrachlorobenzene shows up as a white to off-white crystalline solid at room temperature, earning a spot in laboratory stocks around the world. Its applications often draw from its physical resilience and chemical versatility, yet concern rings out regarding its toxicological effects and environmental persistence. Out in the world, the compound gets manufactured mainly as an intermediate, rarely offered directly to consumers, with purchasing usually going through vetted supply channels and always carrying regulatory baggage.
Physical & Chemical Properties
Handling 1,2,3,4-Tetrachlorobenzene, anyone notices the dense crystals and slight aromatic odor. It boasts a melting point in the range of 128–132 °C, while the boiling point reaches up to 255–260 °C. Low solubility in water contrasts with high solubility in nonpolar organic solvents like benzene, chloroform, or ether. The presence of four chlorine atoms drops its flammability but bumps up its density and chemical inertness under most ambient conditions. Despite its tough exterior, the molecule reacts with certain reducing agents or nucleophilic agents, revealing potential both as a substrate and as a final product in many reaction pathways. Thermal stability extends its shelf life, yet improper storage will invite decomposition and environmental release, particularly under UV irradiation.
Technical Specifications & Labeling
Suppliers provide 1,2,3,4-Tetrachlorobenzene in concentrations purities exceeding 98%, with typical impurities including other polychlorobenzene isomers and trace solvents. Technical data sheets point to CAS number 634-66-2, molecular formula C6H2Cl4, and a molecular weight of 215.89 g/mol. Material Safety Data Sheets demand hazard pictograms relating to environmental toxicity and skin irritancy, urging anyone handling the chemical to wear gloves, goggles, and appropriate lab apparel. Regulatory registers such as REACH or TSCA include this substance, meaning every shipment comes documented with handling precautions, safe storage instructions, and mandatory waste protocols. In the context of shipments, even small samples require substantial paperwork to comply with both national and international chemical safety legislation.
Preparation Method
Industry prepares this compound by controlled chlorination of benzene or its di- and tri-chlorinated derivatives using either liquid or gas-phase processes and chlorine gas under UV light or catalytic conditions. The process, often running at elevated temperatures, demands strict monitoring to avoid over-chlorination or formation of hexachlorobenzene. Fractional crystallization or distillation serves to separate this isomer from others, although high-purity material takes extra steps in chromatography purification. Legacy methods included the use of iron or Lewis acid catalysts, though environmental and safety concerns have shifted practice towards catalytic systems with lower emissions and easier management of by-products. Waste products require robust treatment—thermal destruction or chemical neutralization—due to the persistent nature of chlorinated aromatics.
Chemical Reactions & Modifications
1,2,3,4-Tetrachlorobenzene sits at an intersect of chemical reactivity. Electrophilic aromatic substitution reactions often slow down in the presence of multiple chlorine atoms, yet nucleophilic aromatic substitution opens routes for selective dechlorination, especially under conditions employing hydroxide or amine nucleophiles. Reductive reactions can convert this compound to trichlorobenzenes, while oxidative halogenations yield pentachlorobenzenes. The reactivity offers both opportunity in syntheses—preparing tailored intermediates for agrochemicals or advanced materials—and challenges, most notably the risk of generating toxic by-products such as dioxins, especially under combustion or uncontrolled degradation.
Synonyms & Product Names
Anyone in the lab or industry might encounter a handful of names for this material: 1,2,3,4-Tetrachlorobenzene, Tetrachlorobenzol, or sometimes TCB (with a qualifying isomer label). These aliases, while convenient, sometimes muddy the procurement process, particularly when requesting high-purity samples or referencing regulatory documents that insist on precise IUPAC nomenclature. Some suppliers offer it under proprietary trade names, often with a modifier indicating chlorination degree, intended use, or brand series.
Safety & Operational Standards
Dealing with 1,2,3,4-Tetrachlorobenzene means grappling with real safety concerns. Chronic exposure links to liver and kidney damage, while skin contact causes irritation. The dust can confound respiratory systems, and accidental environmental release lasts for years due to the compound’s persistent nature. Labs and plants handling this material install fume hoods, dust-free dispensing technologies, and containment vessels outfitted for chemical spills. Emergency protocols outline spill response and exposure treatment, with regulatory agencies like OSHA in the United States or ECHA in Europe issuing strict exposure limits and disposal standards. Waste, from lab bench or industrial tank, must not wind up in watercourses or general landfill; incineration within permitted high-temperature units has emerged as the least-worst method on a practical scale.
Application Area
Most batches wind up as feedstock for producing specialty chemicals, intermediates for agricultural chemicals, or even advanced polymers. Some researchers push its use in the synthesis of dyes or chlorinated solvents, leveraging the consistent reactivity profile of its polychlorinated core. In the past, the wider polychlorobenzene family found use as dielectric fluids or insulating media, but environmental persistence dictated sharp downscaling in such applications. Nowadays, the focus drifts towards small-volume, tightly controlled industrial and laboratory work, where traceability matters and every milligram counts.
Research & Development
Ongoing work circles around three core themes: process efficiency, greener synthesis, and remediation. Academic labs try out catalysts based on non-toxic metals or even enzymatic routes, seeking to minimize chlorinated by-products. Waste handling remains a nagging concern, so new destruction and neutralization techniques rank high on research agendas. Analytics teams push for sharper, faster detection of trace contaminants, both for quality assurance and exposure tracking. Public databases swell with new findings every year, while industry keeps data on proprietary processes tightly guarded to maintain competitive advantage. Collaboration between industry and academia, though sometimes fraught, has led to more sustainable protocols and reduced emissions.
Toxicity Research
The push to understand just what this compound does to living systems sparked decades of toxicological research. Early animal studies noted organ damage at moderate doses and persistent bioaccumulation when released into aquatic habitats. Regulatory concern led to long-term monitoring of workplaces handling this chemical, discovering links to elevated cancer risks for substantial exposure and outlining permissible exposure levels. Modern studies leverage genomics and metabolomics, looking for subtle shifts in biological markers following exposure, while large-scale pest management projects examine the ecological impacts further down the food web. Much of this research pushed industry away from bulk production, reinforcing the need for safe handling practices.
Future Prospects
The world’s relationship with 1,2,3,4-Tetrachlorobenzene stands at a turning point. Regulatory pressure grows year on year, pushing producers and users toward strategies emphasizing zero release, closed-loop processes, and safer alternatives. Some hope sits in recovery technologies: advanced oxidation, photodegradation under mild conditions, and robust filtration promise to cut environmental impact. The next decade looks set to bring tough choices: either phase out the most hazardous uses or invest heavily in transformative chemistry that can manage risk, all while supporting the researchers and industries relying on this compound’s unique attributes. Anyone considering future uses cannot ignore the push towards transparency, recyclable intermediates, or advanced monitoring that ties chemical production more closely to ecological responsibility.
What is 1,2,3,4-Tetrachlorobenzene used for?
A Closer Look at 1,2,3,4-Tetrachlorobenzene
Most folks probably haven’t heard of 1,2,3,4-Tetrachlorobenzene unless they work with chemicals or keep a close eye on environmental safety. This compound, a chlorinated form of benzene, often pops up in industry. That doesn’t mean you’ll find it on store shelves. You’re more likely to find it in a chemical plant or lab, where there’s a good reason for using something that sounds straight out of a chemistry textbook.
Where You’ll Find This Chemical
Industries use 1,2,3,4-Tetrachlorobenzene as an intermediate. That means it’s useful for making other chemicals. For example, it plays a role in the production of herbicides. You don’t put it directly on weeds, but it’s a building block behind the scenes. I remember reading reports about certain herbicides that trace their roots back to these kinds of bench chemicals. Chlorinated benzenes, like this one, can crop up in the supply chain for pesticides and fungicides.
It also finds a place in making dyes and pigments. The chlorine atoms in this molecule can turn it reactive enough for companies to turn it into colorants for plastics. It’s not giving you the red or blue itself, but it helps chemists tweak the right molecules to do so. That’s the kind of application you wouldn’t think about unless you dig below the surface of how products get their color. From my old chemistry lab days, I know that precursor chemicals rarely make the final cut in finished products, but everything depends on getting that early chemistry right.
Concerns About Exposure
The problem comes when these chemicals escape outside of the lab or factory. 1,2,3,4-Tetrachlorobenzene doesn’t break down easily. It hangs around in soil or water for a long time. That’s raised eyebrows among folks who’ve studied groundwater contamination. I once interviewed an environmental toxicologist who told me that, even in small amounts, chlorinated benzenes stack up over time. They don’t wash away with a rainstorm.
Concerns extend to work safety, too. Breathing in vapors or handling this compound without proper protection brings risk of skin and eye irritation. Long-term health effects haven’t always been fully understood, which puts extra responsibility on manufacturers to create good workplace safety and waste management routines. Occupational safety data from agencies like NIOSH lay out protective steps—ventilation, gloves, eye protection. I’ve seen firsthand how skipping protocols leads to unnecessary illness or expensive cleanup. Factories that treat this chemical with care see fewer problems down the line.
Pushing for Better Alternatives
Some groups call for alternatives, especially in agriculture, because persistent substances like 1,2,3,4-Tetrachlorobenzene stay in the environment. That’s prompted research into greener herbicide production and safer colorant chemistry. Swapping in less persistent chemicals where it makes sense protects both workers and local waterways.
Government rules keep a close watch. The EPA and similar agencies in other countries track and regulate where this compound ends up. They require careful reporting and handling, which helps encourage good practices. From what I’ve seen, pressure from both regulators and public opinion pushes chemical makers to limit unnecessary use and adopt cleaner technology wherever possible.
What Really Matters
A chemical like 1,2,3,4-Tetrachlorobenzene reminds us that every industrial step leaves ripples. By handling it with care and always looking for safer options, both workers and the environment catch a break. Lessons learned from dealing with persistent chemicals drive innovation in safer substitutes and stronger oversight. Small changes in sourcing and process design cut down risk before problems start. As someone who follows chemical safety news, I see that’s where true progress takes root.
What are the safety precautions when handling 1,2,3,4-Tetrachlorobenzene?
Understanding the Risks
1,2,3,4-Tetrachlorobenzene crops up in chemical labs, some industrial projects, and occasional environmental testing. The first thing I notice with chemicals like this is how quietly they can impact health. It’s a chlorinated aromatic compound, and those don’t mess around. Reports show it can irritate skin, eyes, and cause problems if you breathe too much of it in. Some studies on chlorinated benzenes in general highlight that repeated exposure can affect the liver and nervous system.
Why Proper Protection Makes a Difference
One of my earliest jobs involved weighing out similar compounds. I learned fast that gloves and goggles aren’t just for show. For this chemical, I choose nitrile or butyl rubber gloves — latex doesn’t stand up to chlorinated solvents. Goggles with side protection guard against accidental splashes, and in my experience, a fitted lab coat keeps dust and droplets off everyday clothes.
Not long ago, I watched a new coworker handle a liquid without proper gear. Within a few days, not only did they end up with irritated skin, but they had trouble with eyes watering during simple rinsing. Mistakes like that stick with you.
How Ventilation Saves the Day
These are not the sort of fumes anyone wants to sniff. Even at low levels, chlorinated organics have a sharp, unpleasant smell — a sign of volatile vapors. Lab hoods do more than clear the air; they pull contaminated air away so nothing lingers at mouth or eye level. I never open a bottle of any chlorobenzene product outside of a working fume hood, even if the room looks well-ventilated.
If working in a large plant or warehouse, portable extraction fans and local exhaust hoods add another layer of safety. Air monitors and regular servicing of filters stop lingering fumes and catch leaks before someone feels sick.
Handling and Storage Matter More Than Labels
Locked cabinets marked for toxic or chlorinated solvents serve everybody — not just the person putting the bottle away. I’ve seen what happens when bottles get tucked on the wrong shelf or sit too close to heat sources. This type of chemical can degrade or release vapor if warm, and broken seals let out irritating whiffs no one quickly forgets.
Spills leave a bigger mess if nothing is in place for quick cleanup. I keep absorbent pads and chemical waste containers near any work bench with hazardous products. If a spill happens, don’t just grab paper towels; use pads made for solvent absorption, and wear the same gloves as during handling. Dispose of waste in containers meant for halogenated organics. Kits should always be stocked and clearly labeled so everyone knows where to find them.
Emergency Plans Keep Everyone on Track
Every lab and shop needs training. Regular drills for eye washes, showers, and spill response turn stressful moments into manageable ones. I see the upside in having emergency contacts taped near workstations. If an accident lands someone in trouble, knowing exactly who to call — from a supervisor to a hazmat team — speeds up recovery.
Anyone handling chemicals owes it to themselves and their team to keep these precautions close at hand. No job goes smoother than those where safety comes first, and no warning label stands in for real practice and preparation.
Is 1,2,3,4-Tetrachlorobenzene hazardous to human health?
What’s Lurking in 1,2,3,4-Tetrachlorobenzene?
Most people never hear about 1,2,3,4-Tetrachlorobenzene unless they work in chemical plants or read the fine print on old pesticide labels. The stuff pops up in the production of herbicides and dyes, mostly in heavy industry. Paint remover and insecticide factories might smell it in the air. You probably won’t find it in your kitchen cabinet, but its impact has a habit of creeping far beyond factory walls.
Health Risks That Can’t Be Ignored
Employees in industries using this chemical started asking questions decades ago. Studies found tetrachlorobenzenes lingering in the air, seeping into soil, and tagging along in wastewater. Once in the body, these chemicals don’t exactly pack up and leave right away. The liver and the kidneys, organs that filter nearly everything harmful, bear the brunt. Repeated exposure can lead to serious trouble. Lab studies with animals show damage to their livers and changes in their blood, not far off from what we know happens with other chlorinated benzenes. Reports point to headaches, dizziness, and nausea—pretty standard warnings anytime toxic chemicals are in play.
The U.S. Environmental Protection Agency groups compounds like this among hazardous substances. Governments label it as a possible cancer risk and not without reason. Its structure is similar to other known carcinogens. The World Health Organization links it to disruptions in the endocrine system, raising alarms about reproductive health, developing children, and even risks to immune response. Children playing near chemical dumps or rivers downstream from factories have the greatest risks. The old days of unregulated runoff left some towns cleaning up carcinogens for generations.
On the Job and at Home: Who’s at Risk?
Workers at production sites end up at the frontlines. A cough or rash in the workplace might spark a call to the doctor if a plant uses this chemical. But, there’s personal experience here too: my hometown had an abandoned pesticide plant closed in the early ‘90s. Years later, people still complained about odd smells after a heavy rain, and doctors had their hands full with unexplained illnesses. Parents wondered what was in the river and whether kids should even play outside.
In urban settings, safe drinking water takes attention. Municipalities keep a close eye on the chemical’s levels, since long-term low-dose exposure brings its own slow-moving dangers. For those living near old industrial sites, contaminated soil and dust make gardening and yard work risky. It points to a larger question—can people truly know everything that lingers in their communities after decades of chemical use?
Taking Action: Real Answers, Not Just Reassurances
For real peace of mind, industry has to take full responsibility for emissions and waste, closing leftover dumps and using cleaner tech. Regular health screenings in towns with former chemical plants protect residents, catching issues before they grow worse. Testing isn’t cheap, and cleanup doesn’t happen overnight, but experience shows these steps keep families safer. I’ve seen grassroots groups push for soil and water monitoring, turning a conversation about chemistry into a fight for community health.
Smarter regulations and honest data sharing matter. Families living near industry deserve real answers. Doctors should know what exposures patients might have faced, not just the symptoms. Communities need access to the best testing technology—never guesswork. Problems tied to chemicals like 1,2,3,4-Tetrachlorobenzene only get solved with sharp science, bold policy, and persistent people who refuse to let risks go unseen or ignored.
What is the chemical formula of 1,2,3,4-Tetrachlorobenzene?
Clear Thinking About Chemical Structures
Chemistry makes a big difference in daily life, yet lots of talk around molecules feels mysterious or dry. The chemical formula for 1,2,3,4-Tetrachlorobenzene is C6H2Cl4, a straightforward string that tells you volumes about this compound packed inside a hexagonal ring. Every time I think about the shape of benzene, I picture that sturdy, honeycomb pattern—stable, but not immune to change. Take four of the hydrogens attached to the carbons and swap them out for chlorine atoms, place them on the first four carbons in a row, and you have this tetrachlorinated version.
Interpreting the Formula for Real Life
C6H2Cl4 does more than label a bottle on a shelf; it gives a hint about the way this molecule can interact with the environment and the human body. Four chlorine atoms add weight and bulk to the ring, changing everything from volatility to reactivity. Anyone who's smelled the sharp tang of cleaning products knows chlorine doesn't show up quietly. The formula highlights its potential: as a persistent organic pollutant, 1,2,3,4-Tetrachlorobenzene tends to hang around in soil and water, resisting breakdown. That behavior means chemists and environmental professionals must keep close watch. Extended residency in soil and groundwater means easy movement through ecosystems, something I’ve seen firsthand working on projects tracking industrial contamination.
Health and Environmental Context Matters
The use of tetrachlorinated benzenes has fallen over time. In the past, these compounds got plenty of use for making dyes, serving as intermediates, or acting as dielectric fluids. With mounting evidence about toxicity and stubborn persistence, regulatory agencies started clamping down. The U.S. Environmental Protection Agency and international bodies like the Stockholm Convention categorize these molecules with other dangerous persistent organic pollutants. Workers exposed to chlorinated benzenes often faced headaches and skin irritation, and long-term studies raised questions about liver and immune system effects. Knowing C6H2Cl4’s formula helps predict and test these properties—not just for textbooks, but for guiding actual safety decisions.
Finding Solutions for Contamination—Putting Chemistry to Work
The tough job isn’t just memorizing a structural formula; it’s deciding what to do once that molecule turns up where it shouldn’t be. C6H2Cl4 clings to organic matter in soil, easily leaches into water—smart remediation means matching treatment to properties. Chemical oxidation with hydrogen peroxide or permanganate can break down the ring, but high halogen content brings challenges. Bioremediation efforts using specialized bacteria offer promise, though progress stays slow. I’ve seen land restoration projects where folks relied on a mix of physical removal and clever catalysis—never a quick fix, always demanding close tracking to make sure nothing nasty remains. Fact-based choices for chemical management reduce risk for all who share the land and water.
The Real Value of Precision in Chemistry
There’s no substitute for precise knowledge in science. Being able to trace the structure from C6H2Cl4 gives environmental scientists and safety professionals an edge. The real world doesn’t wait for theoretical perfection—it offers up spills, residues, and waste. Tackling these problems needs a grounded approach, mixing lab facts with practical planning. Following the science, treating these molecules with respect, and watching out for health challenges—this feels less like a chemistry problem and more like everyday responsibility.
How should 1,2,3,4-Tetrachlorobenzene be stored?
Looking at the Facts Around 1,2,3,4-Tetrachlorobenzene
1,2,3,4-Tetrachlorobenzene isn’t new to those working in chemical labs or industrial areas. As someone who’s handled hazardous chemicals, I always remember the trail of rules and mishaps that come with storing substances like this one. This compound comes with its own safety baggage, shaped both by its physical nature and toxicity profile. Many users underestimate chlorinated benzenes, thinking their solid form makes them simple to manage. In reality, anything with chlorines tagging along collects risk—from fire hazards to toxic fumes—especially if storage standards slip.
Storage Location and Environmental Controls
In day-to-day practice, a stuffy corner near an exit never does the job. Good habits include keeping it in cool, well-ventilated spaces that naturally avoid direct sunlight or sources of heat. Some skip this, only to find their containers sweating or getting all sticky when temperatures swing. Temperature swings weaken seals and crank up the odds of leaks. I’ve seen setups where airflow keeps the room fresh, and temperature monitors track the environment, reducing chances of unexpected vapor buildup.
Container Selection Matters
Many suppliers dispatch this chemical inside tightly sealed drums or amber glass bottles. It sits stable under normal conditions, but stored carelessly, contamination or degradation sneaks in. Containers should always be made from glass or chemical-resistant plastic—never some old metal that reacts with chlorinated compounds. I keep an eye out for containers showing any cracks or weaknesses because a tiny split can mean a costly cleanup. Proper labeling is not just a regulatory checkbox, but a lifesaver in emergencies. I’ve found clear, consistent labels prevent mistakes, especially in a busy storage area where dozens of materials compete for attention.
Fire Safety and Spill Preparedness
Chlorinated benzenes don’t catch fire easily, yet if a fire does break out nearby, they produce hazardous fumes, including phosgene and hydrogen chloride. I’ve stood through enough mock emergency drills to appreciate the fuss about fire-resistant storage cabinets. These cabinets stop small incidents from spreading, and closed secondary containment trays under the bottles catch drips or breaks, sparing anyone in the area from bigger contamination headaches. Having a Class B fire extinguisher within arm’s reach gives peace of mind, not just compliance.
Health and Handling Practices
Exposure to 1,2,3,4-Tetrachlorobenzene irritates skin, eyes, and lungs. In my own lab days, I never skipped goggles or gloves while handling such stock. Proper storage includes keeping safety equipment—goggles, nitrile gloves, and spill kits—close by, so accident response isn’t a scramble. Isolation from food, drink, and personal items means storing the chemical in a dedicated chemical area only, never shared with regular storage.
Routine Inspections and Regulatory Responsibility
Responsible storage grows out of routine checks. I make a point to run monthly walkthroughs, searching for degrading containers, improper stacking, and missing labels. Repairs or replacements always follow fast. Waste disposal ties into this as well, since improper handling of leftovers or contaminated material threatens health and the environment. Regulatory agencies like OSHA and the EPA demand strict reporting, but experience teaches you that paperwork is no replacement for common sense, training, and vigilance.
Building a Safer Culture
The real protection flows from respect for the chemical’s risk, peer oversight, and habit. Each worker watching out for the next, double-checking the steps, and treating safe storage as a practice—not just a policy— keeps both people and property secure.
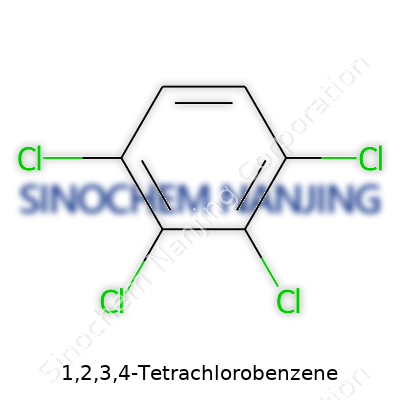
| Names | |
| Preferred IUPAC name | 1,2,3,4-Tetrachlorobenzene |
| Other names |
Tetrachlorobenzene 1,2,3,4-Tetrachlorobenzen Tetrachlorbenzol Benzene, 1,2,3,4-tetrachloro- |
| Pronunciation | /ˌwʌn ˌtuː ˌθriː ˌfɔːr tɛˌtrəˌklɔːrəˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 634-66-2 |
| Beilstein Reference | 636140 |
| ChEBI | CHEBI:36668 |
| ChEMBL | CHEMBL20210 |
| ChemSpider | 9596 |
| DrugBank | DB01595 |
| ECHA InfoCard | 03b75718-71bb-4539-a33e-989a5e5e4efb |
| EC Number | 602-159-5 |
| Gmelin Reference | Gmelin Reference: 41780 |
| KEGG | C06784 |
| MeSH | D013617 |
| PubChem CID | 8549 |
| RTECS number | DC3325000 |
| UNII | VLV56K71Z3 |
| UN number | UN2321 |
| CompTox Dashboard (EPA) | JCHEMID:CHEM0099711 |
| Properties | |
| Chemical formula | C6H2Cl4 |
| Molar mass | 215.90 g/mol |
| Appearance | White solid |
| Odor | aromatic odor |
| Density | 1.50 g/cm³ |
| Solubility in water | Insoluble |
| log P | 4.2 |
| Vapor pressure | 0.0013 mmHg (25 °C) |
| Acidity (pKa) | >2.00 |
| Magnetic susceptibility (χ) | −0.000318 |
| Refractive index (nD) | 1.5705 |
| Viscosity | 4.42 mPa·s (at 45 °C) |
| Dipole moment | 2.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 289.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −28.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -915.8 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ01 |
| Hazards | |
| Main hazards | Toxic by inhalation, in contact with skin and if swallowed. Irritating to eyes, respiratory system and skin. Possible risk of harm to the unborn child. Environmental hazard. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335, H410 |
| Precautionary statements | P261, P273, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1,2,3,4-Tetrachlorobenzene: "2-2-0 |
| Flash point | 110°C (230°F) |
| Autoignition temperature | 380 °C (716 °F; 653 K) |
| Lethal dose or concentration | Lethal dose or concentration of 1,2,3,4-Tetrachlorobenzene: "LD50 (oral, rat): 1650 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,682 mg/kg (oral, rat) |
| NIOSH | CY1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1,2,3,4-Tetrachlorobenzene: Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
1,2,3-Trichlorobenzene 1,2,4,5-Tetrachlorobenzene Pentachlorobenzene Hexachlorobenzene Chlorobenzene 1,4-Dichlorobenzene |

