1,1-Dimethylhydrazine: A Critical Editorial Look at a Chemical's Place in Science and Industry
Historical Development
The story of 1,1-dimethylhydrazine stretches back to a time when the world looked to chemistry for techno-industrial leaps. Its earliest days tie into rocket engineering during the Cold War, when the race for ever more powerful and reliable propellants shaped the direction of chemical manufacturing. I remember reading how Soviet rocket scientists championed this compound as a game-changer, not just for its raw energy, but also for how it simplified storage and transport problems compared to earlier fuels. Over decades, its use became deeply intertwined with space programs. Even now, the scent of nostalgia clings to the chemical, linking generations of chemists and engineers to shared moments of discovery and disaster.
Product Overview
1,1-Dimethylhydrazine carved out a niche in the world of high-energy fuels. It didn’t just meet the needs of rocket launches – it shaped them. Rarely does a chemical find such a specific purpose. With this compound, the promise came bundled with hazards, and the lessons learned from missteps pushed industries to consider chemical stewardship alongside advancement. For old hands in the field, this product means more than a CAS number or molecular structure; it embodies ambitious leaps, the ever-present threat of danger, and the relentless pursuit for better, safer alternatives.
Physical & Chemical Properties
Meet dimethylhydrazine in person and you'll face a clear, volatile liquid with a sharp, ammoniacal odor. It boils off at relatively low temperatures and lights up quickly in air. In labs, breathing in the vapor leaves a distinct impression—even after all precautions, that scent sticks around. The compound’s reactivity brings it both fame as a go-to rocket propellant and infamy due to its instability and toxicity. In water, it’s fully miscible, which raises serious cleanup issues if mishandled. The volatility that makes it useful also demands respect and strict controls. The chemical’s basic formula—C2H8N2—belies the danger it hides under the surface.
Technical Specifications & Labeling
Engineers and chemists can’t ignore the red tape that surrounds 1,1-dimethylhydrazine. The structure forced regulators all over the globe to set precise limits, from container labeling to how much can be on hand without triggering emergency protocols. Color-coding, hazard symbols, and clear documentation sit at every point along the supply chain. The world has written pages of standards mainly because of bitter lessons learned when labs and launch teams underestimated the risks. Veterans know you don’t cut corners—every drum, every bottle serves as both a tool and a potential threat.
Preparation Method
Factories making dimethylhydrazine work with a synthesis that reacts monochloramine with dimethylamine, producing this propellant as a central product. Handling the process means working with controlled environments: exclusive access, constant monitoring, air handling systems designed for rapid response if a leak occurs. The byproducts themselves call for separate containment and treatment. Over the years, industry pushed to reduce emissions, cut losses, and improve reaction yields, driven by both regulation and the high stakes of failure. No one forgets that a single slip can put lives, property, and the ecosystem at risk.
Chemical Reactions & Modifications
This compound’s value springs from how it reacts. In rocket engines, it pairs famously with nitrogen tetroxide, producing a self-igniting "hypergolic" reaction. Turn the page to organic synthesis, and it becomes a potent methylating and reducing agent. Researchers have poked and prodded at chemical modifications, sometimes searching for safer analogues or for tailored reactivity in synthetic chemistry. These efforts highlight the balancing act: push for utility, push for safety—a tension that defines so much of chemical progress.
Synonyms & Product Names
In shops, storage rooms, and research papers, 1,1-dimethylhydrazine pops up under several names. Unsymmetrical dimethylhydrazine (UDMH) is the familiar shorthand. Some suppliers stick with the IUPAC name or refer to it simply as dimazine. This tangle of synonyms complicates training and regulation, since clear communication keeps accidents at bay. In my own work, cross-checking every shipment and label — sometimes three times — proved worth it.
Safety & Operational Standards
Chemists and operators quickly learn that handling dimethylhydrazine isn’t routine. It demands layers of protection: airtight gloves, respirators, remote handling, spill containment gear. Fire teams drill on specialized extinguishing techniques due to the chemical’s unstable nature. Exposed skin or inhalation can cause immediate harm. The standards for operations exist because so many accidents—some well known, some buried in internal memos—have scarred the industry’s memory. Every improvement in safety comes from real-life close calls. Oversight agencies keep adding more rules, not to tie companies’ hands, but because they know the cost of complacency.
Application Area
This chemical earned its keep mainly in aerospace, where its performance outpaced less volatile alternatives. Military and commercial launch providers leaned on its reliability, especially in legacy systems from the 20th century. Beyond fuel applications, some labs used it for chemical synthesis, taking advantage of its high reactivity. The limited crossover into pharmaceuticals or consumer goods isn’t surprising—its reputation and hazard profile close most doors in those sectors. For those working in rocketry, the material feels like both a legacy and a liability: it delivers unparalleled performance but brings headaches around storage, worker protection, and cleanup.
Research & Development
A steady stream of research still focuses on 1,1-dimethylhydrazine. Scientists want safer substitutes, improved detection methods, and techniques to limit environmental impact. Some projects explore stabilizing additives or packaging innovations, while others dig into less toxic chemical cousins for future fuel blends. Analytical laboratories work overtime to trace minuscule leaks and long-term breakdown products, looking to close the gap between lab safety and real-world practice. The shared goal isn't just finding cheaper synthesis, but building a legacy where risk fades and performance stays strong.
Toxicity Research
There’s no skirting the issue: dimethylhydrazine carries serious human health risks. Both acute and chronic exposures threaten blood, liver, and nervous system. Reports from field workers, and occasionally from local residents near storage sites, paint a picture of illness ranging from headaches to outright poisoning. Animal studies back up the observed risks, showing carcinogenic and mutagenic effects. The chemical’s well-earned toxicity fears meant that every stakeholder—manufacturers, regulators, environmentalists—kept searching for mitigation. Environmental health teams track groundwater and air samples near known storage areas, armed with sensitive detection gear. These ongoing efforts serve as reminders: chemical progress rarely comes without a human toll.
Future Prospects
Looking forward, 1,1-dimethylhydrazine stands at a crossroads. Space agencies and commercial launch firms weigh its track record against rising pressure for sustainability and worker safety. New entrants aim to retire it in favor of greener propellants that sidestep the health and environmental baggage. The aspirations for climate recovery, cleaner water, and electrified launches will push this chemical into narrower alleyways, likely confining its role to legacy engine support and specialized applications where replacement remains impractical. My own experience suggests the industry will keep learning from dimethylhydrazine’s history—so future progress can avoid its mistakes, not just celebrate its achievements.
What is 1,1-Dimethylhydrazine used for?
An Explosive Story in Science and Industry
Every rocket launch that lights up the sky depends on fiercely reactive fuels. 1,1-Dimethylhydrazine, also known as UDMH, brings raw power and reliability to the equation. It turns liquid rockets into technological marvels, feeding engines that carry satellites, cargo, and even cosmonauts out of Earth’s gravitational grip. Soviet engineers picked UDMH for missiles and rockets because it can be stored for long periods, even in extreme climates. It remains stable in underground silos, ready without prepping or cooling, so it’s perfect for military and space programs that can’t afford a delay.
In practice, UDMH blends with oxidizers like nitrogen tetroxide. Mix them, and there’s no need for a spark—just direct contact triggers a violent chemical reaction, sending exhaust screaming out of the rocket’s nozzle. That self-igniting nature supports fast and reliable launches, a massive advantage, especially in remote regions or in space where quick starts matter.
Health and Environmental Shadows
This isn’t the kind of chemical you want near homes or farms. UDMH releases toxic vapors known to cause damage to organs and even cancer after repeated exposure. Technicians who prepare Soviet-era rockets shield themselves with layers of rubber and strict protocols. Disposal often falls short, especially in countries that still launch outdated rockets. This leaves behind toxic residues, which seep into river basins and taint the soil. Areas in Kazakhstan and Russia report clusters of health issues near launch sites—a warning flagged by environmental justice groups.
The global conversation about fuel safety keeps growing louder. Some companies and agencies now re-engineer rockets to run on liquid oxygen and kerosene or methane, which burn cleaner and produce fewer carcinogens. It’s tough to switch entirely, since legacy vehicles can’t pivot overnight. Still, I see real progress: SpaceX Falcon and new Artemis missions use less hazardous fuels, while several European missions ban hydrazine derivatives altogether.
Balancing Progress and Risk
I’ve watched coverage of rocket launches since childhood, hypnotized by the plume of fire. The flipside to these triumphs often hides in technical footnotes and post-mission reports: workers sent for emergency medical care, clean-up bills that climb into the millions, fields robbed of safe crops for generations. These are problems no amount of spectacle can mask.
Regulations grow stricter as risks become impossible to ignore. Russia and China still use UDMH for older launch systems, but pressure from the public and international partnerships keeps nudging programs toward greener choices. Engineers search for propellants with fewer health hazards and governments fund cleanup of polluted launch sites. My background in science taught me that progress often asks for tough trade-offs, but I believe the future belongs to safer fuels and smarter oversight.
Learning from Experience
Space needs powerful engines, but not at any cost. Choosing what powers rockets shapes more than spaceflight history—it affects lands, waterways, and public health for decades to come. I hope more agencies listen to scientists, medical researchers, and communities living in rocket shadows. Holding launch programs accountable for cleanup and risk reduction drives innovation, turning the dream of space exploration into something everyone can celebrate. As technology evolves, so must the commitment to a cleaner launch.
Is 1,1-Dimethylhydrazine hazardous to health?
What is 1,1-Dimethylhydrazine?
1,1-Dimethylhydrazine, better known as unsymmetrical dimethylhydrazine (UDMH), shows up most often as rocket fuel in space programs. It burns hot and fast, which works well for blasting rockets upward, but shows a much darker side for workers and bystanders. Decades of launching satellites and spacecraft have pumped thousands of tons of this chemical into the ground, water, and air, especially near military and space facilities in Russia, Kazakhstan, China, and other countries.
How Exposure Happens
I grew up near a site where rocket debris sometimes landed. People in such places joke about burnt rubber smells after rocket launches. This scent usually means hydrazine or a close cousin leaked out. You’d think once a launch is done, the risk would drift away with the smoke, but that's not how chemicals like UDMH behave. Contact with this stuff comes through breathing vapors, touching contaminated surfaces, or drinking water tainted by debris and leftover fuel.
Health Hazards: What Do We Know?
Long before lab tests confirmed it, anyone with direct experience noticed UDMH made people sick. Symptoms start with headaches, dizziness, and nausea. Your skin and lungs bear the brunt if you cross paths with it directly. American and Russian toxicologists report that even low concentrations can shut down the nervous system, mess with the liver, and ruin the kidneys. UDMH is classed by the International Agency for Research on Cancer as a possible human carcinogen. Studies with animals showed cancerous tumors following both short-term and chronic exposure.
Children near contaminated rocket crash sites in Kazakhstan grew up with higher rates of blood disorders. Residents there tell stories of mysterious illnesses and livestock dying off after launches. These aren’t isolated events—similar health trends pop up anywhere this fuel gets spilled or dumped.
Environmental Fallout and Long-Term Impact
Soil and water hold onto UDMH for a long time. Because it’s highly soluble, the chemical spreads quickly once it hits groundwater. I’ve seen photos from research trips: local rivers run brown after a fuel spill, and plants wilt for months. Fish and birds in those areas develop growth and reproductive problems. Cleaning up afterward means dealing with stubborn chemicals that resist breaking down. Remediation takes years, and people living around launch sites often lose access to clean water or grazing land.
What Can We Do?
Safe alternatives exist. Newer rockets in the US and Europe run on liquid methane or kerosene derivatives, which break down naturally. A major push to retrofit or retire old rocket classes using UDMH could protect future generations. Local governments should keep people updated about risk zones, push for regular soil and water tests, and hold companies or agencies responsible for cleanup. International watchdogs like the World Health Organization provide safety guidelines, but local investment in health clinics and water treatment matters most. Transparency about spills and routine monitoring grants people the information they need to protect themselves and push for change.
Every time I hear a rocket launch rumble overhead, I think about what’s left behind on the ground. That lingering risk—especially for rural families and children—sticks around long after the trail of fire disappears. Addressing the hazards of 1,1-dimethylhydrazine means more than just better fuel: it means acknowledging the cost that comes with every launch.
How should 1,1-Dimethylhydrazine be stored safely?
Handling a Tough Chemical
Most folks outside the aerospace and chemical industries never see 1,1-dimethylhydrazine up close, and for good reason. This chemical serves as a powerful rocket fuel and a key industrial reagent, but it also brings serious risks. My own brush with it came at a research facility, where even a whiff from a poorly vented cabinet made it clear: mistakes with this substance carry high stakes.
Why the Hazards Call for Respect
1,1-Dimethylhydrazine burns fast and clear in engines, but in storage, the same qualities turn hazardous. It catches fire easily, and its vapors can poison lungs and skin. Facility managers and technicians can’t cut corners here. A study from the U.S. Environmental Protection Agency reports that spills or leaks of this fuel, even in small amounts, create huge safety and environmental problems. The chemical loves to explode when mixed with oxidizers or exposed to open flames, and just touching it without the right gloves can start a chain reaction on the skin.
Down to the Nuts and Bolts of Storage
Safe storage for 1,1-dimethylhydrazine starts with basics that no experienced safety manager ignores. Steel drums lined with special coatings work better than plain steel since the chemical eats through metal over time. All containers need airtight, corrosion-resistant seals—nothing falls into the "good enough" category. Forced ventilation removes stray vapors that could otherwise turn the air inside a storeroom into an emergency.
The storage area counts for a lot. This chemical never belongs near oxidizers, acids, or strong bases. Mixing even vapor traces with these sets up a fire risk, and every fire department hazmat officer I’ve met agrees that separation often saves lives. Stick to cool, dry storage. Heat cranks up vapor pressure inside containers, multiplying the risk of leaks or bursts.
Anyone storing 1,1-dimethylhydrazine needs to gear up with full-face shields, chemical-resistant suits, and heavy-duty gloves. Those aren’t just for spill response—routine inspections can stir up vapors. Regular checks catch corrosion early. From my visits to well-run plants, I’ve seen tight inspection logs and sensors set to alarm at even trace vapor releases. That’s not paranoia; it’s planning.
Culture and Systems Come First
Firms with a safety-first culture get the edge in avoiding accidents. Daily gear checks, emergency drills, and frank talk about risks mean mistakes don’t get hidden. The American Chemical Society points to a strong link between worker training and lower incident rates, especially around chemicals with fast-acting dangers. Stick to labeled storage, keep emergency wash stations close, and train every handler to spot symptoms of exposure.
Solutions: Technology, Design, and People
Online monitoring and leak detection tech add a layer of safety that older generations didn’t enjoy. Smart systems shut off valves and notify emergency crews at the first sign of vapor leaks. Spill berms around storage give extra time for first responders. Double-walled tanks add backup protection for bulk storage, especially in older facilities.
Building storage around the idea that mistakes can happen—using smart sensors, tough containers, and open communication—brings accidents down. In my years seeing what works, it’s always a mix of well-chosen gear and empowered workers that keeps 1,1-dimethylhydrazine locked down and away from trouble, where it belongs.
What are the handling precautions for 1,1-Dimethylhydrazine?
An Unforgiving Chemical
1,1-Dimethylhydrazine stands out as one of those chemicals your average person will never encounter at home but plays a huge role in rocket fuel. It has earned a reputation for being tough to work with and for good reason. The fumes pack a punch—sharp, nose-tingling, and dangerous. People who have spent any time in labs or on pads where this stuff gets stored know that short cuts bring real consequences. Keeping exposure low starts with discipline, not luck.
Staying Ahead: Know the Hazards
This compound can attack fast. It’s flammable, volatile, and toxic whether inhaled, swallowed, or absorbed through skin. Even a splash on the hand can mean irritation, blisters, burns, or worse. Breathing in small amounts can leave you dizzy; higher doses tie into cancer risk. The US Environmental Protection Agency actually lists it as a probable human carcinogen. I remember a case where a friend ended up with a painful rash that stuck around for weeks—just from a contaminated glove. It taught us all to never let our guard down.
Protective Gear: No Room for Half Measures
Walking into a storage or transfer area starts with the right gear. Nitrile gloves, chemical goggles, a face shield, and a lab coat make up the basics, but for bigger jobs you need a full chemical suit and a respirator that can handle air-purifying cartridges specifically rated for organic vapors. No gear stays effective forever, so it matters to inspect everything for tears or wear every time. Tiny leaks spell trouble. Sites that use 1,1-Dimethylhydrazine regularly invest serious money in backup supplies and mandatory fit testing.
Workspaces Built for Safety
You never want to handle it in crowded labs or where fumes can build up. Fume hoods rated for toxic, flammable chemicals give the best shot at keeping vapors away from your face. At bigger sites, you’ll find dedicated transfer rooms with heavy-duty ventilation and multiple sensors to warn about leaks. All transfer lines use sealed systems, and even routine maintenance calls for purging lines with nitrogen. More than once, we rehearsed emergency shut-downs—hoping never to use them, but knowing the drill by heart anyway.
Emergency Prep and Spill Control
Real-world experience shows that spills don’t split hairs over neatness. Absorbent pads, neutralizing agents, and sealed waste containers must be close, not stashed at the far end of the lab. Everyone trained for quick response knows exactly which showers and eyewash stations actually work. You only discover flaws with drills, not the real thing. Calling authorities matters if more than a few milliliters get loose. It’s never a solo fix—spills demand trained teams.
Towards Better Systems
Safer handling starts with cutting back on how much of the stuff moves at once. Robotics or remote-handling gear has reduced human contact with nasty substances like this. Real progress also depends on a culture where workers speak up over near-misses, rather than hide mistakes. Regular air monitoring, personal badge samplers, and smart sensors now track exposure much better than eyeballing a color change or relying solely on alarms. Teams with better training and real backup respond faster and with more confidence.
Why Consistent Vigilance Means Everything
Chemicals like 1,1-Dimethylhydrazine don’t forgive inattention or bravado. They force you to slow down, double-check, talk things through, and never assume the day will run as expected. Legal rules and safety sheets only matter if people care enough to follow them, and if managers make time for drills. In my own work, I’ve seen that good habits and a healthy respect for the risks set apart safe operators from those who get unlucky. Handling precautions work, but only when everyone treats them as real lifelines—not just red tape.
What are the environmental impacts of 1,1-Dimethylhydrazine?
A Closer Look at a Troubling Rocket Fuel
Growing up, I watched televised rocket launches—big, thrilling, noisy events. The aftermath never made the news. What happened out in the fields, rivers, and forests near launch sites stayed mostly out of sight. The chemical at the heart of this worry, 1,1-Dimethylhydrazine (frequently called UDMH), makes rockets fly. It also spreads a toxic footprint long after the roar fades.
Poison in the Launch Path
UDMH helped the Soviet Soyuz, Proton, and other rockets get off the ground. It turned space exploration into an everyday event for some countries, but it brought a silent cost. Every time old rocket stages fell back to earth, they let loose not just bits of scorched metal but also traces (and sometimes pools) of unburned fuel.
Researchers in Kazakhstan and Russia found sharp increases in cancers and neurological issues near old crash sites. UDMH doesn’t just vanish in sunlight. It seeps deep into groundwater, finds its way into crops, gathers in fish and livestock. I read about entire regions around Baikonur that live with the legacy of these spills, and it’s not fiction; this is long-term suffering.
Why UDMH Sticks Around
The fuel doesn’t break down quickly. Its by-products, like N-nitrosodimethylamine (NDMA), rank high on lists of hazardous substances. NDMA turns up in drinking water—sometimes above what health agencies consider safe. Wind and rain move this contamination far beyond impact sites. Where the fuel touches soil, it wrecks plant life. Soil organisms—the tiny backbone workers of agriculture—take a heavy hit, which weakens food chains.
Wildlife doesn’t dodge the bullet, either. Animals drinking from polluted water or grazing on contaminated grasses build up toxins in their bodies. Reports have listed mass die-offs of birds and cattle after major spills. These deaths don’t just affect herders and farmers; they break rural economies and social fabrics.
Better Choices and Making Things Right
Safer rocket fuels do exist—some new rockets use liquid oxygen or kerosene, which don’t bring the same disaster when spilled. The push from commercial operators and private space companies has helped speed up the shift away from UDMH. Stronger regulations around the world force governments and companies to clean up their launch programs. People are asking for transparency, health checks, and quicker drop zones for rocket stages, not just far-off suggestions.
I once believed rocket launches were a clean symbol of progress. Stories from those living under the threat of UDMH showed me a different reality. It’s all too clear: Space technology must advance without sacrificing the health of people and planet. Cleaning up old messes, investing in greener fuels, and paying attention to frontline voices shows real leadership. The future of spaceflight belongs to everyone—not just those with the loudest engines.
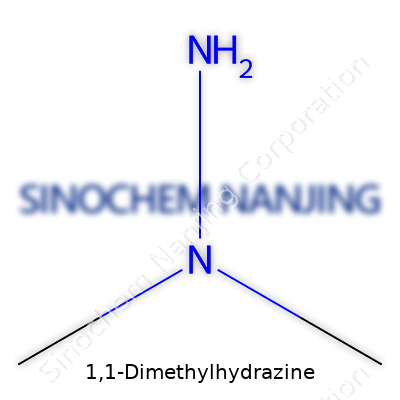
| Names | |
| Preferred IUPAC name | N,N-Dimethylhydrazine |
| Other names |
Unsymmetrical dimethylhydrazine UDMH Aerozine 1 Dimazine |
| Pronunciation | /ˈwaɪlənˌdaɪˌmɛθəlhaɪˈdreɪziːn/ |
| Identifiers | |
| CAS Number | 57-14-7 |
| Beilstein Reference | Beilstein Reference 605349 |
| ChEBI | CHEBI:7476 |
| ChEMBL | CHEMBL1542 |
| ChemSpider | 7461 |
| DrugBank | DB01990 |
| ECHA InfoCard | 19fd507d-b671-4f21-b6f0-c51d54cace15 |
| EC Number | 01-2119475601-38-0001 |
| Gmelin Reference | 8216 |
| KEGG | C01352 |
| MeSH | D002584 |
| PubChem CID | 7516 |
| RTECS number | UD8750000 |
| UNII | 83M8V38O8I |
| UN number | UN 1234 |
| Properties | |
| Chemical formula | C2H8N2 |
| Molar mass | 60.10 g/mol |
| Appearance | Colorless liquid |
| Odor | fishy |
| Density | 0.79 g/cm³ |
| Solubility in water | miscible |
| log P | -0.38 |
| Vapor pressure | 43.4 kPa (at 20 °C) |
| Acidity (pKa) | 7.09 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | -12 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 0.54 cP (20 °C) |
| Dipole moment | 1.62 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 199.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | +66.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2040 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | T59. |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H301, H311, H331, H351, H370 |
| Precautionary statements | P210, P233, P260, P261, P264, P271, P280, P301+P310, P302+P352, P304+P340, P308+P311, P311, P312, P320, P321, P330, P362+P364, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-4-2 |
| Flash point | 0 °C (32 °F; 273 K) |
| Autoignition temperature | 287 °C (549 °F; 560 K) |
| Explosive limits | 2.5–97% |
| Lethal dose or concentration | LD50 oral (rat) 59 mg/kg |
| LD50 (median dose) | LD50 (median dose): 79 mg/kg (oral, rat) |
| NIOSH | NIOSH=UB0175000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,1-Dimethylhydrazine is 0.5 ppm (1 mg/m³) |
| REL (Recommended) | 0.005 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Hydrazine Monomethylhydrazine Tetramethylhydrazine |

