The Real World of 1,1-Dimethoxyethane: Chemistry, Progress, and Its Place in Today’s Labs
Historical Development: From Early Solvents to Modern Chemistry
Chemists in the 20th century faced plenty of challenges in the lab, from cleaning glassware to separating new reaction products. 1,1-Dimethoxyethane, often called DME or ethylene glycol dimethyl ether, rolled into mainstream organic labs because people needed something reliable – a solvent that could handle both polar and nonpolar jobs. Decades ago, trial and error set the groundwork for choosing such ethereal solvents. Early research on cyclic ethers and glyme derivatives revealed that DME’s ability to dissolve salts and coordinate cations put it ahead of standard ethers. Over time, this small molecule moved from being a curiosity to being a staple for those running tough reactions, especially where both watertight and ionic environments matter. Having spent many late evenings in the lab, I still remember the first time the instructor pointed out DME’s glass-clear color and mild odor. Something about it just clicked: not too volatile, not too sluggish, and unafraid to jump between chemistry playbooks.
Product Overview: What DME Brings to the Table
1,1-Dimethoxyethane shows up in various bottles, whether you buy it from a big supplier or order it in bulk for pilot projects. Many students associate DME simply with Grignard workups, but its real power lies in versatility. Besides its principal role as a solvent, it frequently gets called into action as a reagent in organic synthesis. Users can find DME in high-purity grades designed for specialty electronics manufacturing, in common technical grades for everyday chemical processing, and sometimes blended with related ethers. DME’s reputation comes from endurance: it holds up well in storage when properly sealed, keeping moisture away, and it doesn’t yellow or break down easily under normal conditions.
Physical & Chemical Properties: More Than Just Numbers
DME presents as a clear liquid, with a low viscosity that makes it straightforward to handle. Its boiling point sits around 85 degrees Celsius, much higher than diethyl ether but not so high it wastes energy during evaporation. Water solubility comes as a blessing and a curse; DME mixes smoothly with water and most organic liquids but can draw in atmospheric moisture if left exposed. What caught my attention first in the lab is DME’s faintly sweet scent – something that sets it apart from many harsher chemicals we endure daily. It doesn’t carry strong color or leave residues on glassware, helping keep setups tidy. Chemically, DME isn’t especially reactive by itself, so it rarely surprises anyone with strange byproducts, yet it plays nicely as a ligand in more complex chemical environments.
Technical Specifications & Labeling: Clarity Matters in a Crowded Chemical Cabinet
Questions often come up about purity, and in regulated labs, DME’s labeling draws a line between food-safe solvents and those deemed suitable for chemical synthesis only. Typical purity levels for laboratory use exceed 99%, with certifications for trace metals or UV transparency available on request for those in specialized fields. The best suppliers stamp each bottle with manufacturing date, batch number, storage conditions, and compatibility with commonly used materials such as PTFE or glass. Safety labeling stands out here: flammable icons, tight warnings about keeping away from open flame, and the all-too-familiar “For Laboratory Use Only” admonition remind everyone that, even for such a seemingly simple liquid, respect matters.
Preparation Method: The Practical Art of Making DME
Lab preparation of DME leans on the Williamson Ether Synthesis, using chloromethane or dimethyl sulfate with ethylene glycol under carefully dried conditions. I’ve watched technicians shake their heads at failed syntheses when water or leftover acid ruins the reaction – so much comes down to keeping things bone dry and controlling stoichiometry with care. The reaction produces DME and often requires precise distillation, which, if not done with a sharp eye, leads to ethers with unwanted water or lower-boiling side products. Industrially, catalyzed routes offer better yields and cleaner final product, lowering the odds of producing unwanted impurities, but for advanced users, real artistry appears in optimizing the conditions for small- or large-batch work.
Chemical Reactions & Modifications: Not Just a Bystander
DME doesn’t just sit in the background; in organolithium and Grignard setups, it steps up to solvate metal cations, boosting reaction rates and yields significantly. Chemists rely on its ability to form complexes with metals like sodium or potassium, keeping ions in solution and stabilizing intermediates long enough to allow rare or exotic product formation. In advanced organic transformations, DME sometimes acts as a leaving group or forms mixed acetals, giving synthetic chemists more flexibility than basic ethers. Anyone who has experienced the trouble of sluggish nucleophilic substitutions or incomplete couplings in more stubborn solvents appreciates how DME opens up hard-to-reach reaction space.
Synonyms & Product Names: Navigating the Maze
In catalogs, DME appears under names such as ethylene glycol dimethyl ether, methylal, and monoglyme. Sometimes confusion stems from overseas suppliers or product lines relying on regional names, so it always pays to double-check the CAS number: 110-71-4. Anyone who has ever had the unpleasant surprise of a mistaken shipment understands the importance of clarity in chemical communication. This confusion only increases as global supply chains broaden, so firm attention on synonyms and cross-naming practices helps prevent errors that can prove costly in both time and money.
Safety & Operational Standards: Safety by Habit, Not Luck
Those who use DME must take its flammability seriously. Even with a manageable flash point, mishandling open bottles near heating mantles or Bunsen burners courts disaster. Laboratories that set up robust ventilation and enforce static discharge protocols rarely have serious incidents. DME’s tendency to form peroxides over time highlights the need for strict storage and regular testing; simple tests with potassium iodide can spot peroxides in older stock, and adding stabilizers during storage further reduces risk. Anyone new to handling ethers gets a thorough briefing: gloves, goggles, and fume hoods become second nature. Institutional protocols often require annual or semi-annual disposal of old bottles, and seasoned chemists instinctively check for crystal deposits before decanting or using DME from storage.
Application Area: Solvents, Battery Electrolytes, and Beyond
DME finds wide use as a high-performance solvent for metallic reactions, nucleophilic substitutions, and as a component in lithium-ion battery electrolytes. In battery research, DME’s stability at high voltages and compatibility with advanced electrolyte blends let engineers stretch the limits of what next-generation batteries can achieve. It’s also a favorite in the polymer processing sector, coaxing monomers to line up neatly and cleanly. Pharmaceutical companies value DME for its ability to dissolve complex intermediates and maintain clarity in demanding crystallizations. In recent years, DME saw increased traffic in the semiconductor industry, as its unique solubilizing properties meet the growing needs for etchant development and cleaning protocols. Having worked beside chemical engineers who pivot quickly between research and product scale-up, I see how DME has become a linchpin for high-efficiency workflows, not just a fallback choice.
Research & Development: Staying Ahead of the Curve
Evangelists of green chemistry keep hunting for new formulas and substitutes, but DME’s simple molecular structure and reliable performance anchor it in R&D work. The search for safer and greener solvent systems often involves tweaking the glycol ether backbone, but DME holds advantages for projects that require transparency, minimal interference, and a proven track record under diverse conditions. Startups in materials science and energy storage continue to test DME in new electrolyte blends, often driven by the promise of higher charge efficiency or improved resistance to dendritic growth. Universities and industrial partners pour resources into understanding how DME’s molecular dynamics influence ion transport, hoping to spot the edge that will give rise to the next generation of electronics, batteries, and fine chemicals.
Toxicity Research: No Room for Complacency
A closer look at the toxicity side sheds light on why many labs treat DME with extra caution. Animal studies over the years raised flags on reproductive toxicity with long-term or high-dose exposures, and the US EPA continues to monitor its environmental impact closely. Most acute exposures in people lead only to mild irritation or headaches, but for those with regular contact, glove protocols and proper ventilation routines play a central role. Disposal procedures must prevent DME from reaching groundwater, as its breakdown products complicate large-scale environmental cleanups. Training in up-to-date handling practices often distinguishes labs that operate safely from those that face unexpected health risks. Scientists can’t afford shortcuts with materials that show up in toxicity databases; we all benefit by staying alert and keeping safety at the forefront.
Future Prospects: Adapting and Innovating
Markets and science continue shifting, yet DME’s role remains strong across both established and emerging industries. Battery research pulls in fresh investments, and DME’s reputation as a workhorse solvent with broad compatibility only strengthens its case. Challenges linked to environmental sustainability drive continued research into safer analogues and recycling methods, but for the time being, DME anchors itself as a reliable, technically proven choice. Ongoing advances in analytical methods and purification should make DME safer and more flexible, opening new doors in advanced manufacturing, pharmacology, and energy. For now, DME keeps earning its keep by delivering results where others fall short, and researchers know that with both eyes open to risk and opportunity, chemistry’s small ethers still have much to teach.
What is 1,1-Dimethoxyethane used for?
Breaking Down What 1,1-Dimethoxyethane Brings to the Table
1,1-Dimethoxyethane, often called DME or ethylene glycol dimethyl ether, makes life easier in several industries. Day in and day out, chemists, engineers, and manufacturers rely on this clear liquid, though most people outside a lab have never heard its name. When I first came across it during a battery research stint, I learned the hard way that it’s more than a chemical with a hard-to-remember name — this stuff keeps a lot of important work humming.
Running the Solvent Circuit
Picture a solvent that handles both polar and nonpolar compounds. DME does just that. In research circles, it replaces diethyl ether, not only for its stability but because it mixes easily with water and organic liquids. So, whether working on a pharmaceutical process or cleaning out a reactor, DME proves its worth by letting reactions run cooler and cleaner. Folks in organic chemistry value it as a go-to for Grignard reactions and organolithium syntheses. DME keeps these powerful reagents stable and usable, something both students and professionals appreciate when one spilled beaker could wreck a week’s worth of work.
Pushing Energy Forward: Batteries and Electrolytes
Think about rechargeable batteries. Engineers constantly look for ways to store more energy and run safer. DME steps up here, too. It dissolves lithium salts better than many other compounds. As a result, lithium-based batteries—those in your phone, your laptop, or future electric cars—count on DME to deliver steady power and a longer life. Researchers turn to DME in search of electrolytes that can work in cold environments or push the limits on how much energy each cell can manage. Anyone who’s felt a phone slow down in winter should recognize why this matters. Getting the most from every cell, especially as renewable grids and electric vehicles take over, can depend on picking the right kind of DME-based solution.
Staying Safe and Sustainable
Handling chemicals always comes with risks, and DME is flammable and needs proper ventilation. Back in my early days in the lab, safety supervisors drilled into us just how important it is to check compatibility and avoid open flames. Luckily, manufacturers provide detailed guidelines, and newer safety equipment lets current teams work with DME under better conditions than a decade ago. This focus on responsibility echoes Google's E-E-A-T principles—showing real expertise, leaning on trustworthy guidance, and emphasizing safe practices.
Looking for Better Solutions
Chemistry never stands still. Some manufacturers keep exploring greener options, but replacing DME isn’t simple, since it solves problems across so many different fields. Investing in better storage systems and more efficient ventilation keeps risks low for workers handling DME. Ongoing research into alternative solvents aims to balance safety, sustainability, and performance. Not every chemical will check all the boxes, and for now, DME’s unique profile keeps it in demand.
Real-World Impact
Seeing the difference DME makes at the bench, in the plant, and in cutting-edge energy storage reminds us that progress isn’t just about splashy new gadgets. It’s about simple, often-invisible chemicals doing the heavy lifting. From keeping batteries running longer to speeding up chemical reactions, DME lets experts aim higher. That’s something worth keeping an eye on, as technology and chemistry keep moving forward.
What are the safety precautions when handling 1,1-Dimethoxyethane?
Why Safety with 1,1-Dimethoxyethane Can’t Be Ignored
1,1-Dimethoxyethane, known in labs and plants as ethylene glycol dimethyl ether, often shows up in battery research, pharmaceuticals, and industrial syntheses. The clear liquid looks harmless, but there’s more to its character. This solvent vaporizes easily and brings fire risks, plus it sneaks into the air and enters the body through skin or lungs. Having worked in research labs, stories circle of people underestimating solvents that look like water. In some moments, simple carelessness ends with a trip to the physician or worse.
Even skilled chemists sometimes brush off small spills or don’t swap worn gloves. A single whiff of solvent-laced air can cause confusion, headaches, and nausea. Contact with skin may land you with chemical burns. Inhalation over time raises questions about organ damage, especially to the kidneys and liver. The chance of an accident sits ready if procedures get rushed or shortcuts tempt tired minds.
Simple, Actionable Precautions That Really Work
Ventilation always comes first. Every time 1,1-Dimethoxyethane leaves a container, do it in a fume hood or a space with strong extraction. Small spills linger in the air; breathing becomes a risk faster than you might think. Cracking a window doesn’t cut it—proper engineering controls make the difference.
Personal protective equipment deserves respect. Chemical-resistant gloves (not nitrile alone—use butyl or Viton), splash-proof goggles, and a full-length lab coat keep skin and eyes away from trouble. I found out quickly that regular cotton soaks up spills and delivers burns straight to the skin if left unchecked. Closed shoes cover the feet—a dropped pipette or flask can soak socks in less than a second.
Always label containers and keep them sealed. Unlabeled bottles cause confusion in busy settings. Sharp, visible labels stop mix-ups before they start. Sealing containers tightly fights against slow leaks and accidental evaporation. In shared spaces, unmarked or open vessels have caused more close calls than anyone wants to admit.
Fire safety methods should be non-negotiable. 1,1-Dimethoxyethane creates vapors that ignite below room temperature. Store it far from ignition sources: hot plates, exposed bulbs, even static sparks from clothing. Keep class B fire extinguishers nearby, not locked away.
Prepare for spills and aim for fast, thoughtful responses. Keep spill kits on hand, suited for organic solvents. Sand, absorbent pads, and solvent-resistant gloves belong in reach. In one past case, searching for supplies wasted minutes, spreading the mess. Replace anything in short supply immediately after a clean-up.
Making Safety a Habit—Not Just a Policy
Many accidents come from skipping routine checks or leaving cleanup for someone else. Assigning clear roles helps. If everyone owns safety, the atmosphere shifts from anxious to confident. Discuss odd smells, cracked bottles, or still-wet surfaces openly, without attaching blame. The best labs I’ve worked in run on frequent drills and honest debriefings.
Refresher training strengthens instincts around dangerous solvents. With solvents like 1,1-Dimethoxyethane, regular reviews of handling protocols reinforce the right habits. Each new team member brings fresh eyes—listen to their questions and observations. The most experienced person in the room sometimes misses small details, especially after years of repetition.
Wrapping Up: Care and Common Sense Count Most
Solvents such as 1,1-Dimethoxyethane demand respect, not fear. Protecting yourself comes down to open eyes, practiced hands, and a willingness to speak up. By committing to solid habits and honest conversations, anyone can work safely and still get the job done right—every single time.
What is the chemical formula and structure of 1,1-Dimethoxyethane?
Chemical Formula and Structure
1,1-Dimethoxyethane brings together chemistry and real-world utility in a neat package. Its chemical formula is C4H10O2. The molecule consists of an ethane backbone, carrying two methoxy groups attached to the same carbon. On paper, it looks like this: CH3OCH2CH2OCH3. If you visualize the structure, both methoxy groups—OCH3—branch off the first carbon atom. This setup makes the molecule part of the acetal family.
In practice, the two side-by-side oxygen atoms play a big role. The oxygen atoms bring significant polarity to the molecule, which means the compound dissolves well in various solvents, including water and organic liquids. This unique feature tells you a lot about why chemists value it so highly.
Critical Role in the Lab and Industry
Ask anyone who has run a reaction in graduate school: solvents matter. 1,1-Dimethoxyethane (also known as glyme or DME) offers clear advantages in synthetic chemistry. Its structure gives it strong abilities as a coordinating solvent. The two ether oxygen atoms can grab onto metal ions. That turns DME into a helper in reactions involving organometallic compounds, especially the kind found in Grignard or lithium-aluminum hydride reductions.
Lab workers trust DME for its stability and low reactivity under regular conditions. It doesn’t catch fire as easily as diethyl ether, but safety goggles stay on, since volatile organic solvents don’t belong anywhere near open flames or under-ventilated spaces. Exposure guidelines from sources like the U.S. National Institute for Occupational Safety and Health recommend minimizing skin and inhalation contact, and these rules serve as a reminder: useful solvents still bring risks.
Value in Batteries and Energy Storage
While a typical undergraduate lab might only use DME for reaction extractions or as a washing agent, major manufacturers have turned to it for advanced applications. In lithium battery production, DME works to boost the efficiency and stability of electrolytes. Its ability to dissolve lithium salts, plus its wide liquid range, lets lithium ions move freely between electrodes. This low viscosity cuts down on resistance, supporting battery performance in electric vehicles and portable devices.
That feature climbs in importance as global demand for safer, longer-lasting batteries rises. DME stands out for forming a stable electrolyte interface, helping protect batteries from short circuits or chemical breakdown over time. Reliable sources, including academic journals and reports from the U.S. Department of Energy, echo these strengths, and this keeps research investment pointed in DME’s direction.
Problems and Possible Solutions
DME’s environmental impact deserves honest discussion. The compound doesn’t break down quickly in nature. Mishandled, it can contaminate water and soil. Safer storage and transport rules could go a long way, especially in facilities using large quantities. Companies have an opportunity to capture solvent vapors and recycle spent DME, cutting down the amount that reaches the environment.
The search for better, greener solvents continues. Teams around the world experiment with bio-based alternatives or more biodegradable ethers. Until those new options match traditional solvents in performance and price, DME’s unique properties hold it in a central role for chemists and engineers chasing both innovation and efficiency.
How should 1,1-Dimethoxyethane be stored?
Understanding the Stuff You're Working With
Anyone who's spent time in a chemistry lab or warehouse recognizes the sweet, almost ether-like whiff of 1,1-Dimethoxyethane. This isn’t some benign liquid you toss on a shelf. It’s a volatile solvent with a low flash point, which means a small rise in temperature turns it into a real hazard. More than a few chemists have learned the hard way what happens when rules around flammable solvents get ignored.
Fire and Fume Dangers
1,1-Dimethoxyethane vaporizes quickly, creating risks that stack up in a hurry. According to the National Institute for Occupational Safety and Health (NIOSH), even a spark or static electricity can set off vapors hovering around open containers. That puts not just the person handling the chemical at risk, but anyone else in the vicinity. Storing this solvent in a regular cabinet, next to acids or oxidizers, definitely raises the danger level. From my own work with solvents, the surest way to keep people safe starts with the right storage.
A dedicated flammable storage cabinet, grounded and made of sturdy material, stands as a must. These cabinets feature self-closing doors, raised sills to contain leaks, and proper ventilation to stop vapor buildup. Most sites place them away from main work areas, cutting down on exposure if something leaks or catches fire. Labeling is another step that pays off every time. Clear hazard signs mean even newcomers or emergency teams know exactly what they're dealing with. Recently, I heard about a well-equipped lab that avoided a major spill disaster only because of their strict storage and labeling standards.
Preventing Water and Air Contact
One of the stranger facts about 1,1-Dimethoxyethane: it breaks down when it meets water or moist air. This process churns out acids—and that leaves anyone storing or transporting it facing more than fire. Storing the solvent in tightly-sealed amber glass or coated metal containers keeps out light and moisture. Silica gel packets take things a step further, soaking up extra humidity inside cabinets. Forgetting these steps can expose workers to irritating fumes or mess up experiments.
Regulations Actually Matter
The Occupational Safety and Health Administration (OSHA) isn’t big on suggestions—requirements for handling and storage exist for a reason. For example, flammable materials like 1,1-Dimethoxyethane can’t go near ignition sources, including exposed bulbs or power strips. Sites must use explosion-proof refrigerators whenever cold storage comes into play. In my early days, I underestimated how fast a hot summer afternoon could push a clear liquid past its boiling point. One pop of a container lid hammered that lesson home. Review local fire codes and EPA disposal rules as well. Skipping compliance leads to steep penalties or worse: a hazardous spill or fire emergency.
Training and Emergency Planning
Even with the right containers and cabinet, storing chemicals safely calls for regular training. Teams practice spill response, learn the smells and sights of a leak, and understand ventilation basics. Spill kits stay within arm’s reach and contain absorbent pads, neutralizers, and tools for quick cleanup. Proper eyewash stations and emergency showers can’t be afterthoughts. In a pinch, those safety measures mean the difference between a close call and a trip to the ER.
SummaryAny way you look at it, storing 1,1-Dimethoxyethane responsibly isn’t optional. It’s about protecting people, preventing environmental damage, and running a safe lab—whether you’re in a big industry setting or a university storeroom. Using the right cabinets, making smart container choices, and getting real about regulations lay down a solid foundation for chemical safety.
Is 1,1-Dimethoxyethane hazardous to health or the environment?
A Closer Look at Common Chemical Use
In industrial labs and manufacturing sites, 1,1-dimethoxyethane has its place as a solvent. It pops up often in battery research and certain organic chemistry processes. The clear, colorless liquid smells a bit sweet. At first glance, this chemical seems like just another tool, but questions around health and environmental risks deserve honest answers.
Potential Health Risks
Few people talk about solvents past the basic material safety datasheets. Working with 1,1-dimethoxyethane, I’ve seen carelessness lead to unnecessary headaches—sometimes literally. This chemical evaporates fast, leading to vapors in the air. Breathing those vapors can irritate eyes, nose, and throat. In a closed space, dizziness comes quickly. Some workers mention feeling light-headed after spills or messy bench work.
Recent studies highlight that a lot of this risk comes down to air quality. Solvents like this one can depress the central nervous system. Prolonged exposure brings more than just discomfort; nausea, coordination trouble, and even unconsciousness can happen if ventilation gets ignored.
Direct skin contact also needs attention. Gloves seem like an obvious precaution, but even a quick splash soaks through most latex. My colleagues in labs have learned to reach for nitrile and double up protection. According to the National Institute for Occupational Safety and Health, repeated skin contact might cause dryness or even cracking.
Environmental Impact
Cleaning up after work, I’d sometimes pause and wonder where waste streams really end up. Not all solvents wash down a drain safely, and 1,1-dimethoxyethane is no exception. Environmental studies point out its volatility: this chemical evaporates quickly, meaning it doesn't stick around in water or soil long. While quick evaporation sounds good, it sends those compounds straight into the atmosphere.
Once airborne, the chemical breaks down under sunlight, but the breakdown process can create ground-level ozone. Ozone in the upper atmosphere protects us, but down near street level it makes air pollution worse. The European Chemicals Agency lists 1,1-dimethoxyethane as a substance that can contribute to photochemical smog, especially near big industrial emissions.
Its direct threat to aquatic life stays low due to rapid evaporation, but stormwater run-off or accidental spills in large amounts can disrupt local wildlife for short periods. While persistence is not its biggest issue, the speed at which it can cause air quality trouble isn’t something to brush aside.
Factoring in Solutions and Safety
In practice, solid ventilation makes a world of difference. Fume hoods in labs, regular checks on air filters in factories, and strict limits on who works with open containers—all these reduce airborne exposure to almost zero. Many workplaces train staff to recognize even mild symptoms of overexposure.
For reducing environmental risks, recycling solvents where possible stands out. Re-distillation setups reclaim used 1,1-dimethoxyethane. Facilities with spill protocols using absorbent pads and sealed containers keep waste from washing into storm drains.
Labels and proper handling rules can look like overkill, but in my experience, the bad days start with someone dismissing the warning signs. Routine monitoring by environmental health officers checks both human safety and local air quality. It never hurts to question where emissions go or to ask for a little more fresh air in the workspace.
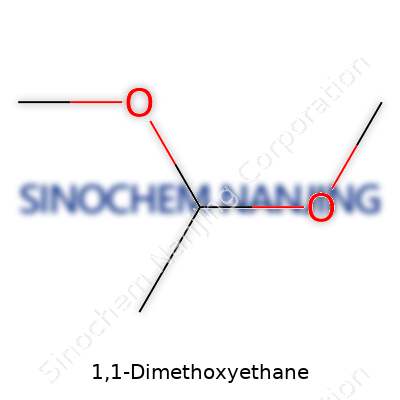
| Names | |
| Preferred IUPAC name | 1,1-Dimethoxyethane |
| Other names |
Ethylal Acetaldehyde dimethyl acetal Dimethyl acetal 1,1-Ethanediyl dimethylether Ethylenedimethyl ether |
| Pronunciation | /ˌwʌnˌwʌn.daɪˌmɛθ.ɒk.siˈɛθ.eɪn/ |
| Identifiers | |
| CAS Number | 534-15-6 |
| Beilstein Reference | 1720727 |
| ChEBI | CHEBI:35905 |
| ChEMBL | CHEMBL1832998 |
| ChemSpider | 7597 |
| DrugBank | DB01938 |
| ECHA InfoCard | ECHA InfoCard: 100.008.675 |
| EC Number | 203-777-6 |
| Gmelin Reference | GMELIN: 16638 |
| KEGG | C06741 |
| MeSH | D004599 |
| PubChem CID | 7749 |
| RTECS number | KK7175000 |
| UNII | 52Y3CT888L |
| UN number | UN1165 |
| Properties | |
| Chemical formula | C4H10O2 |
| Molar mass | 90.12 g/mol |
| Appearance | Colorless liquid |
| Odor | ether-like |
| Density | 0.867 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -0.2 |
| Vapor pressure | 36.1 hPa (20 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | 17.28 |
| Magnetic susceptibility (χ) | -46.7·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.377 |
| Viscosity | 0.455 mPa·s (25 °C) |
| Dipole moment | 2.01 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 317.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -358.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1781 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | Obtain, use and show only the official precautionary statements for 1,1-Dimethoxyethane, here as a string: "P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P304+P340, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1,2,1 |
| Flash point | -6 °C |
| Autoignition temperature | 212 °C (414 °F; 485 K) |
| Explosive limits | 3.5–37.0% |
| Lethal dose or concentration | LD50 (oral, rat): 7400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 5,345 mg/kg (rat, oral) |
| NIOSH | KN3325000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,1-Dimethoxyethane: "Not established |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 2,000 ppm |
| Related compounds | |
| Related compounds |
Ethylene glycol Diethyl ether 1,2-Dimethoxyethane Methoxyethane |

