Looking Closer at 1,1-Difluoroethylene: Where Science, Industry, and Caution Meet
The Road from Laboratory Curiosity to Modern Utility
Scientific progress isn’t always a straight shot from discovery to economic powerhouse. Take 1,1-difluoroethylene, also known as vinylidene fluoride. Back in the early-to-mid 20th century, research chemists started tinkering with fluorinated hydrocarbons. They followed a hunch that adding fluorine to traditional molecules could give them some exceptional properties—things like chemical resistance, temperature resilience, solvent stability. Out of this work, 1,1-difluoroethylene emerged not just as a building block in textbooks, but as the backbone for polyvinylidene fluoride (PVDF), which today shows up in coatings, membranes, battery components, and everyday goods where reliability is prized. Watching this journey unfold, you start to appreciate how deep curiosity and gritty, step-by-step experimentation shape the fabric of modern technology.
Physical and Chemical Identity: Details that Matter
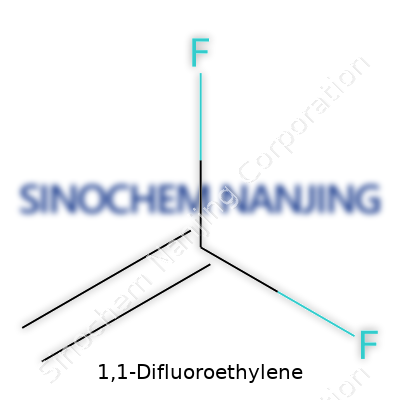
Dealing with 1,1-difluoroethylene hands-on leaves no illusions: chemistry loves specificity. This compound, with formula C2H2F2, sits in the family of gaseous fluorocarbons. Despite being colorless and possessing a faintly ether-like odor, it doesn’t blend into the background. Its tendency to vaporize at room temperature, low boiling point near -85°C, and ability to ignite at elevated concentrations call for respect in every stage of handling. The double bond between the carbons adds reactivity, but the two fluorine atoms tip the scale toward resilience against acids, bases, and many organic solvents. These core features provide the canvas for diverse industrial uses, where being inert at one moment and reactive at another can spell the difference between success and disaster.
Preparation, Modification, and the Path to Polymers
Anyone working in chemical labs knows there’s rarely only one road to a target molecule. Industrial-scale production of 1,1-difluoroethylene usually goes through dehydrohalogenation of 1,1,1-trifluoroethane or hydrochlorination of acetylene—but each route presents its own balance of costs, yields, and waste. Getting this compound from gas cylinders or reactors into safe containment means controlling temperature and pressure with care, because leaks invite fire risks and exposure. The juicy part comes after: subjecting 1,1-difluoroethylene to radical polymerization. This step transforms simple molecules into tough, chemically resistant polymer chains, opening the doors to the prized PVDF plastics found in aerospace, electronics, and filtration systems. On a research note, chemists often tweak the core structure—swapping substituents or mixing in comonomers—hunting for unique properties to match new market demands in areas like lithium-ion batteries or next-generation membranes.
Keeping Labels Straight: Synonyms and Identity Confusion
Names can trip up even seasoned researchers. 1,1-difluoroethylene also answers to vinylidene fluoride (VDF) or by its IUPAC label, 1,1-difluoroethene. Experienced buyers and lab workers quickly learn not to rely on trade names or brand labeling alone, since misunderstandings about chemical identity can spark trouble during ordering, compliance inspections, and waste disposal. Taking the time to double check CAS numbers, molecular formulas, and label warnings makes mistakes less likely. Clarity here beats the cost of mishandled shipments or regulatory fines later down the line.
The Importance of Safety Protocols and Operational Standards
Flammable gases always demand a steady hand and a respect for detail. With 1,1-difluoroethylene, this starts with proper containment—using leak-tested gas cylinders or closed system reactors. Inhaling the gas irritates airways, and accidental leaks build up burn risks fast. Regulations, including OSHA workplace standards and chemical storage codes, direct both employers and employees to wear flame-resistant gloves, keep electrical sources in check, and run continuous ventilation. I’ve watched industrial sites run drills and update Standard Operating Procedures after even minor incidents because once set alight, fluorinated gases can release noxious byproducts, including carbonyl fluoride and hydrogen fluoride. These stand as a wake-up call—adhering to well-established protocols isn’t nitpicking, it’s the line between work as usual and wider crisis.
Reaching Into Applications: Why This Gas Still Matters
Markets place a high value on what 1,1-difluoroethylene does for them. Most notably, its transformation into PVDF underpins durable coatings, high-spec filtration, and key electrical insulation materials. Engineers designing for chemically harsh environments—think offshore oil installations, water treatment, or cleanroom manufacturing—often turn to polymers born from this molecule, avoiding breakdown that could scrap million-dollar equipment. The battery industry, especially with electric vehicles and grid storage getting a modern push, sees these materials as crucial for electrolyte containment, separator consistency, and even flexible casings. From personal experience, digging through the performance specs of PVDF-coated wires versus conventional plastics reveals how the stability, flexibility, and lifespan really justify the specialized chemistry upstream.
Pushing the Research Frontier and Facing Toxicity Questions
Curiosity continues driving innovation, but it’s impossible to ignore safety and environmental health. Years of study have outlined that 1,1-difluoroethylene can cause respiratory distress at elevated concentrations, and research into long-term exposure hazards pushes scientists and regulators to keep better monitoring data on atmospheric levels and waste releases. Unlike some earlier fluorochemicals, VDF generally doesn’t last long in open air, thanks to rapid degradation under sunlight and oxygen, but breakdown products sometimes raise fresh questions. Laboratories now focus on both extending performance—by creating novel copolymers or surface treatments—as well as reducing fugitive emissions. Some research directions include safer process recycling, lower-emission catalyst systems, and alternatives that lower toxicity without sacrificing crucial end-use benefits.
What’s Coming Next: The Fork in the Road
Advances rarely come without trade-offs or uncertainties. The momentum behind 1,1-difluoroethylene keeps growing as global investment in renewable energy, advanced electronics, and medical devices expands. At the same time, tighter chemical safety standards, greater public scrutiny of fluorinated molecules, and international agreements around greenhouse gases press producers to engineer both safer handling systems and greener synthesis. Many research labs now blend chemistry with environmental science—mapping out the full lifecycle impact, rather than only celebrating performance specs. Direct experience on factory floors and in R&D pilot plants teaches that material suppliers willing to innovate—designing for safer production, improving containment, and embracing meaningful transparency—will set the pace for industries still relying on this unique, foundational molecule. The story of 1,1-difluoroethylene keeps evolving, proof that real progress comes from both technical brilliance and hard-earned lessons in stewardship.
What is 1,1-Difluoroethylene used for?
What 1,1-Difluoroethylene Does in Industry
1,1-Difluoroethylene, which some people know as vinylidene fluoride, sets the pace for a lot of advanced material science work. Most of the time, people picture basic plastics when they think of chemicals like this. The story goes much deeper, though. Its biggest role shows up in the world of fluoropolymers, those strong materials that stand up to all sorts of weather, harsh chemicals, and even the slow erosion of time. Industries turn to these polymers for jobs where regular plastic gives up too soon.
Everyday Products and Real-World Impact
Nonstick cookware often relies on 1,1-difluoroethylene's chemical cousins. The same goes for certain wires and cables that live inside cars, airplanes, and computers. Those wires need an outer jacket that won’t crack or melt under heat, current, or accidental exposure to corrosive stuff. The fluoropolymer coatings that come from 1,1-difluoroethylene handle these tough conditions. Without it, many manufacturers would go back to metals, which adds weight, cost, and sometimes risk of corrosion.
It also helps build membranes for filters that clean up drinking water or separate gases in heavy industry. Those membranes last longer and don’t break down from all the chemicals they meet daily—which matters a lot to both industry and the everyday person relying on clean water.
Health and Environmental Stakes
Chemicals like 1,1-difluoroethylene bring up real questions for health and the environment. Years working in a chemistry lab sharpened my respect for safety policies. Even tiny amounts of exposure demand caution: inhaling the gas or letting it touch your skin can irritate or even hurt you long-term. Strict ventilation, gloves, eyewear, and constant leak checks form the backbone of handling this stuff. It only takes one slip to remind everyone these materials aren’t just another batch of ordinary plastic.
Environmental experts watch chemicals like this with good reason. Persistent materials sometimes escape into air or water during manufacturing or disposal. Regulators, like the EPA and their peers in the EU, lay down tight rules to limit emissions and spills. I saw factories install scrubbers and sealed tanks, pushing to keep the workplace and community safer. Researchers keep hunting for ways to break these chemicals down after their useful life ends. The goal remains clear: keep benefits, lose the damage.
Moving Forward: Safer Alternatives and Better Processes
Some research teams experiment with less hazardous replacements for fluoropolymers, but few options match up on heat resistance and lifetime. Most odds favor new chemistry tweaks, safer plant designs, and tighter containment to control risks. I’ve spoken with engineers investing in technologies that recycle these polymers or limit the waste during production. These steps, along with improved training, matter a lot for anyone working near such chemicals or living nearby.
As technology keeps moving forward, the role of 1,1-difluoroethylene will stick around. Its influence shapes everything from safer electronics to reliable cars and medical gear that keeps performing without fail. Balancing those advantages with full responsibility means everyone—from engineers to environmental scientists—needs to pay attention. That’s the cost and the lesson from using powerful materials in modern life.
What are the safety precautions when handling 1,1-Difluoroethylene?
Why Handling 1,1-Difluoroethylene Matters
1,1-Difluoroethylene, known among chemists as vinylidene fluoride, often shows up in places where plastics and specialty coatings get made. The hype around fluoro-based polymers is real, so it’s no surprise this chemical keeps turning up on factory floors worldwide. Anyone who has spent time near tanks labeled with a big, bold “flammable gas” sign knows the uneasy feeling that comes with volatile substances. This colorless gas packs a punch, especially for new workers who may underestimate it because it doesn’t smell that strong.
A Straightforward Look at the Hazards
Direct exposure to 1,1-difluoroethylene has some very real risks. The gas ignites with a spark or open flame, making it a fire risk in any building. Breathing in the fumes can leave someone dizzy, short-winded, or worse. Colleagues in labs have suffered headaches and numb fingers after quick exposure. Any skin contact tends to cause cold burns or irritation, which comes from the gas escaping under pressure and cooling rapidly.
Long-term health impacts raise concerns as well. The International Agency for Research on Cancer has connected similar chlorofluorocarbons with increased cancer risk. The US Occupational Safety and Health Administration restricts exposure to vinylidene fluoride to small amounts: 2 parts per million during an eight-hour shift. So, brushing off safety procedures isn’t just risky, it can cost lives and livelihoods.
Protection Begins With Awareness
Workers should treat 1,1-difluoroethylene the way you’d treat an unpredictable wild animal—don’t take chances. I’ve watched old-timers suit up with goggles, chemical-resistant gloves, and full-face respirators, not because they wanted to look the part but because years on the job taught them not to take unnecessary risks. Good ventilation trumps everything. Fume hoods and local exhaust vents make a big difference in keeping concentrations down.
Smart Habits: Storing and Moving Safely
This gas lives in steel cylinders under pressure. Dropping a cylinder or leaving it near heat isn’t just sloppy—it can cause violent explosions. Storage areas require cool temperatures, no open flames, and clear signage. I learned the value of double-checking for leaks with soapy water, since even small leaks can fill a room quickly if unchecked. Teams need well-marked gas detectors, not just for safety audits but for day-to-day operation.
Training: The Core of Chemical Safety
Every worker dealing with difluoroethylene must know what to do if the gas escapes. My first fire drill in a chemical plant opened my eyes to how chaotic things get—fire extinguishers didn’t work on flammable gases; we had to swiftly vent the area and use the right type of fire suppression. Having a set of clear emergency protocols makes all the difference, especially when seconds count. Regular safety talks keep people alert and prepared.
Long-Term Solutions: Investing in Better Practices
Company leaders who put resources into safer equipment, regular maintenance, and up-to-date training protect not only workers but also their own reputation. That means looking for leak-resistant valves, automating risky steps, and always updating safety data sheets once new findings come out. Regulatory agencies like OSHA and EPA provide strict guidelines; following them should feel like the baseline, not a chore.
The Path Forward
Working with 1,1-difluoroethylene means respecting its dangers every step of the way—from the moment the cylinder arrives until the last bit is used. With common-sense precautions, attention to personal protective equipment, strong workplace habits, and ongoing learning, workers stand a much better chance of keeping themselves and their coworkers safe.
What are the physical and chemical properties of 1,1-Difluoroethylene?
The Basic Makeup
1,1-Difluoroethylene, or vinylidene fluoride, draws a lot of attention for good reason. It's got a formula of C2H2F2. Right away, those two fluorine atoms tell you this isn’t your everyday hydrocarbon. You find it as a colorless gas, which carries a sharp, somewhat sweet smell. The boiling point sits around -82°C—definitely not something you leave unchecked in a warm room. That kind of volatility means handling and storage must be careful, with equipment designed for gases that can evaporate quickly.
Physical and Chemical Behavior
A gas with a low boiling point moves straight into the set of chemicals that call for respect in the lab and on the factory floor. Vinylidene fluoride weighs in with a molecular mass near 64 grams per mole. With its physical makeup, it’s less dense than air—if it leaks, it rises. I’ve seen plenty of folks surprised at how fast gases like this can disappear from a workspace, making proper ventilation a non-negotiable feature.
Flammable gases like this one aren’t just a theoretical safety line on a sheet of paper. 1,1-Difluoroethylene can form explosive mixtures when enough collects in the air. Sparks, flames, even static—any of these could set it off. In practical settings, this keeps teams on their toes, using explosion-proof tools and frequent leak checks. Chlorinated solvents chew it up, but water leaves it alone thanks to the strong carbon-to-fluorine bonds. Moist workplaces don’t break it down, yet you won’t see it forming stable mixtures with lots of other solvents, so dedicated equipment is common in manufacturing environments.
Uses Driven by Chemical Traits
Vinylidene fluoride forms the backbone of polyvinylidene fluoride (PVDF), a heavyweight polymer in the world of pipes, membranes, and electrical insulation. The chemical stability of those fluorine bonds lets it handle aggressive acids and bases—no small feat for modern industry. I’ve come across plenty of water treatment membranes built from PVDF, where long-term reliability really matters.
Electricians and engineers reach for PVDF in wiring because the polymer keeps its cool under heat and doesn’t break down under UV light. Those same fluorines make the plastic relatively inert, so it doesn’t shed dangerous byproducts under most normal conditions. The catch is in the cost and complexity of working with these fluorinated chemicals, which tend to require skilled handling and specialized gear.
Health, Safety, and Paths Forward
Handling 1,1-Difluoroethylene needs safety glasses, gloves, and solid ventilation. Breathing in the gas irritates airways and can act as a mild anesthetic, so regular monitoring matters—especially in closed spaces. The fire risk means teams need emergency plans drilled and ready. In my own experience, people do best learning from real drills, not just posters on the wall.
Accurate sensors and quick repairs reduce escapes into the air, where the gas doesn’t just pose fire concerns but also adds to environmental worries. While the compound itself hasn’t raised the same alarms as some other fluorinated chemicals, ongoing research should follow its use and impact as closely as possible. Working with groups promoting green chemistry might yield next-gen polymers that hold performance but skip the risks linked to fluorinated gases.
Better training, real investment in leak-proof equipment, and staying updated on research drive safer, cleaner workspaces. Industries counting on vinylidene fluoride should keep asking how to balance its strong features with responsibility, keeping both workers and the environment top of mind.
How should 1,1-Difluoroethylene be stored and transported?
Recognizing the Risks with 1,1-Difluoroethylene
1,1-Difluoroethylene draws a lot of attention in the world of chemical safety because it is flammable and under high pressure as a liquefied gas. Just a small leak turns into a big issue fast. Spending part of my career with people who deal with pressurized gases showed me that even seasoned workers get caught off guard by small mistakes. Reports from the U.S. National Library of Medicine point to its potential for explosion in confined spaces and the health threats posed by vapor inhalation. Hearing a regulator hiss can make hearts race; not just for property, but for colleagues on site.
Storage Contingencies—What Works in Daily Practice
Proper containers make all the difference. Metal cylinders built for high-pressure service prove dependable. Regular checks for rust or valve damage give peace of mind. It helps to set these cylinders upright in cool, dry buildings with steady temperatures, well away from sunlight or heat sources. Easy access for inspection and plenty of space around each cylinder—these are steps that might seem basic, but they prevent headaches later. Good storage means keeping them away from chemicals that react with fluorinated gases. Oxygen, for one, stays far from difluoroethylene in any serious facility.
Physical separation isn’t just a suggestion; insurance inspectors and chemical safety boards report case after case of avoidable fires and explosions traced to sharing storage space. This sort of mistake can lead to long investigations and sometimes worse. As a rule, clear signage and locked gates cut down on unauthorized access, which helps if you’ve got newer staff or are dealing with contractors.
Moving 1,1-Difluoroethylene—Precautions That Stand Up to Real Life
Transporting pressurized gas means planning down to the route. Department of Transportation regulations set strict labeling, placarding, and documentation, and for good reason. During field audits, officials check whether those handling these cylinders have had real safety training, not just a cursory video. I once saw a shipment turned away simply because the truck’s storage racks weren’t bolted down, even though everything else looked spotless.
Securing the cylinder so it doesn’t roll or tip reduces risk. Shoulder straps, safety chains, and valve protection caps are common tools. Damage often happens on loading docks, so trained hands and patience save money and lives. Most accidents crop up during transfer from storage to vehicle, so double-checks on valve closure and securement pay off.
Protecting People First
No matter how advanced your safety gear, something as simple as smelling a sweet odor—a sign of a possible leak—or noticing frost near the valve calls for action. Ventilation can’t be an afterthought; proper air flow and exhaust systems in storage areas mean even a leak won’t turn into a fatal hazard. Emergency response kits, eye washes, and clear access to exits are non-negotiable. Safety data sheets posted in plain sight remind everyone about symptoms and steps to take if things go wrong.
Room for Improvement
Automation and remote monitoring help by letting staff see pressure or temperature changes before trouble starts. Better technology doesn’t replace basic vigilance, but it cuts down on the “I didn’t notice” problem. Sharing safety records with local firefighting teams and making sure managers walk storage areas at the start of each shift creates a culture where one person’s mistake doesn’t turn into a company-wide disaster. Chemical safety won’t ever be entirely risk-free, but these routines and a culture of respect for the hazards go further than any rulebook does.
What are the potential health hazards of exposure to 1,1-Difluoroethylene?
Everyday Encounters with an Industrial Chemical
Plastics go in everything these days, from packing cases to coating wires. The drive for cheaper, lighter goods fuels the chemical engine. 1,1-Difluoroethylene, also called vinylidene fluoride, ends up in polyvinylidene fluoride plastics, which engineers like for its strength and chemical resistance. This chemical didn’t show up in any cafeteria lunch menu, though; it’s all business, all industry. But workers making or handling resins, coatings, or solvents run into it in real life, not just safety manuals.
Breathing Risk: Lungs Feel It First
Exposing people to gases they can’t smell or barely notice is always risky. 1,1-Difluoroethylene evaporates at room temperature and drifts into the air before anyone realizes. The National Institute for Occupational Safety and Health (NIOSH) has flagged it as a substance you should handle with care. Inhalation brings the worst effects. Shortness of breath and throat irritation can show up fast. Prolonged exposure could leave people coughing, wheezing, feeling dizzy or disoriented.
One lab study documented lung damage in animals at concentrations above 2,500 parts per million in the air. The U.S. Occupational Safety and Health Administration (OSHA) says workplaces shouldn’t let levels go above 1 ppm on average during a typical shift. That standard comes from cases where workers reported headaches, fatigue, or even chest pain. Long exposure seems to take a toll beyond the nose and throat.
Other Dangers: From Skin to Long-Term Risks
Handling chemicals isn’t just about breathing, either. 1,1-Difluoroethylene comes as a gas or a pressurized liquid, so skin contact can burn or freeze tissue, depending on the way it’s used or spilled. Soaked gloves or boots may trap the chemical long enough to do real damage before anyone notices. Safety data sheets warn about frostbite and delayed skin injury.
Beyond burns and breathing problems, researchers keep asking about long-term risks. The U.S. Environmental Protection Agency (EPA) lists 1,1-difluoroethylene as “possibly carcinogenic,” meaning it might play a role in causing cancer over years of exposure, but definitive human data is still lacking. That doesn’t mean it’s innocent. It reflects how hard it is to get clear answers when exposures change every day on the shop floor. Some animal studies found kidney and liver tumors after heavy, long-term dosing, which grabs attention in industrial health circles.
Solutions Start with Air and Vigilance
Long before regulations arrive, healthy skepticism about “safe” levels leads a lot of factory managers to improve building ventilation. Exhaust hoods, fresh air systems, and sensors can reduce the chance of nasty inhalation events. Supervisors need to make sure everyone who works with gases like this gets good respirators — not just the cheap dust masks — and learns what symptoms point toward exposure.
Lab coats, gloves, and face shields keep emergency room doctors busier when they’re missing. Chemical training that goes deeper than powerpoints can put safety on auto-pilot. Reporting systems for symptom flare-ups or spills help catch problems early, especially for smaller shops or factories that don’t have full-time industrial hygienists. No one should gamble on chemical safety because someone else didn’t bother to check the air filters.
Every person who’s gotten a headache or a skin burn from an “invisible” gas walks away more careful, more skeptical, sometimes angry that warnings weren’t louder. That frustration shapes tough rules and better work habits. Experience and facts — not just chemical formulas — belong in every real conversation about health at work.
| Names | |
| Preferred IUPAC name | 1,1-Difluoroethene |
| Other names |
Vinylidene fluoride 1,1-Difluoroethene DFE VDF |
| Pronunciation | /ˌwʌn.wʌn.daɪˌfluːərəʊˈɛθɪliːn/ |
| Identifiers | |
| CAS Number | 75-38-7 |
| 3D model (JSmol) | `JSMOL="C=CF(F)F"` |
| Beilstein Reference | 1465051 |
| ChEBI | CHEBI:39034 |
| ChEMBL | CHEMBL50238 |
| ChemSpider | 12644 |
| DrugBank | DB14049 |
| ECHA InfoCard | ECHA InfoCard: 100.003.982 |
| EC Number | 206-834-7 |
| Gmelin Reference | 5691 |
| KEGG | C14283 |
| MeSH | D003990 |
| PubChem CID | 6335 |
| RTECS number | KI9625000 |
| UNII | 9Y41106MNB |
| UN number | UN1951 |
| Properties | |
| Chemical formula | C2H2F2 |
| Molar mass | 64.038 g/mol |
| Appearance | Colorless gas |
| Odor | Sweetish, ether-like |
| Density | 0.675 g/mL at 25 °C |
| Solubility in water | slightly soluble |
| log P | log P = 1.65 |
| Vapor pressure | 4620 mmHg (20 °C) |
| Acidity (pKa) | pKa = 12.44 |
| Magnetic susceptibility (χ) | -18.3·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.2055 |
| Viscosity | 0.204 cP (25 °C) |
| Dipole moment | 1.97 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 284.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -483.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1095 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V10BX06 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H220, H280, H351 |
| Precautionary statements | P210, P261, P280, P304+P340, P377, P381, P403 |
| NFPA 704 (fire diamond) | 2-4-2 |
| Flash point | “-119 °C (closed cup)” |
| Autoignition temperature | 455 °C |
| Explosive limits | 5.6% – 14.2% |
| Lethal dose or concentration | LCLo human inhalation 20000 ppm/30M |
| LD50 (median dose) | LD50 (median dose): 4770 mg/kg (rat, oral) |
| NIOSH | KY2625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,1-Difluoroethylene: "1000 ppm (2400 mg/m3) TWA |
| REL (Recommended) | 5 ppm |
| IDLH (Immediate danger) | IDLH: 1,000 ppm |
| Related compounds | |
| Related compounds |
Vinyl fluoride 1,1,1-Trifluoroethane 1,2-Difluoroethane |

