1,1-Difluoroethane: More Than a Simple Chemical
Tracing the Evolution
Long before 1,1-Difluoroethane turned up inside the cans of keyboard dusters or the lines of refrigerant pipes, researchers searching for less dangerous chlorinated refrigerants stumbled over its potential. Chemists in the early twentieth century saw the chase for safer, more stable molecules as more than technical work; they saw societal risk writ large in every CFC production run. The 1930s fluorocarbon revolution pulled a reluctant world away from ammonia and methyl chloride, both widely used in home refrigerators but all too often responsible for fires or poisoning. 1,1-Difluoroethane grabbed attention for its chemical stability and relatively low toxicity. It did not win the first-tier refrigerant crown, but it became deeply entwined with industry in its relentless campaign against global warming and ozone depletion. As society learns more about the environmental costs tied to traditional refrigerants, old chemistry textbooks no longer sit collecting dust, they offer lifelines to safer climate policies.
What Are We Really Handling?
Some folks see 1,1-Difluoroethane as nothing but a colorless, faintly sweet-smelling gas. It’s often described on product packaging as “HFC-152a” or simply “difluoroethane.” Its relative safety compared to older gases might feel reassuring, but catch a whiff and it is impossible to ignore its chemical bite. I recall working in a machine shop where a spill turned the air sharp and cold, the vapor streaming past with a visible white haze. This stuff boils just below minus 25 degrees Celsius, which means a blast from a can drops surface temperatures fast. Chemically, it’s just C2H4F2, but those two fluorines give it new properties—nonflammable at room temperature, resistant to radical formation. Still, read the label’s technical specs. Purity, moisture content, and storage pressures vary across products and not everybody follows the rules. Where someone sees compressed air, another sees potential energy waiting for any leaky valve or static discharge.
Knowing Its Properties Should Change How We Use It
On paper, 1,1-Difluoroethane carries a boiling point around −24°C, its molecular weight just a notch above ethane. It feels lighter-than-air on the tongue but chills if sprayed on the skin due to rapid vaporization. The molecule’s relative inertness compared to older refrigerants reduces the chance of nasty byproducts under normal circumstances. Still, its affinity for oil allows it to carry unexpected residues through pneumatic systems or insulation foam processes. Technically, it falls under hydrofluorocarbons, and no matter how “safe” it’s called in advertising, sales literature rarely highlights its dangerous chemistry when misused indoors. Pressurized cylinders can burst if left in heat, and its tendency to displace atmospheric oxygen doesn’t always come up in discussions about air quality. I’ve seen people treat keyboard dusters as harmless toys. It deserves just as much respect as the freons it replaced.
Digging into Technical Details and Labeling
Product labeling often compresses a host of tech specs into a few cryptic lines: purity percentage, moisture limits, net volume, pressure rating. In practice, these details make or break the difference between an industrial-grade refrigerant and a gas that chokes up an HVAC system. Manufacturers tout “99.9%” as the gold standard for purity, but trace contaminants—like chlorinated solvents or hydrocarbons sneaking in from processing leftovers—threaten equipment reliability and user safety. A can on the shelf may only say “HFC-152a,” “flammable,” and “contains gas under pressure.” I have watched plenty of folks ignore the fine print about non-refillable cans, incompatible storage temperatures, and warnings about intentional inhalation. Fact is, industry sees these warning panels as legal shields instead of guides for best practice. Labels rarely capture the reality of a chemical’s wild possible uses, nor the urgency of respecting its technical boundaries.
Getting Down to the Chemistry: Making and Modifying It
The chemical prep for 1,1-Difluoroethane doesn’t sound glamorous. Manufacturers depend on halogen exchange or direct fluorination of other ethane derivatives; sometimes it starts from vinyl chloride or trichloroethane, treated with hydrogen fluoride under tightly controlled conditions. These reactions lean on catalysts—antimony pentachloride or halides—while industry quietly battles corrosion, toxic intermediates, and erratic yields. Purification steps strip out excess reagents and stabilize the final liquid under pressure. Once isolated, modifications can crank out thinners, hydrofluoroolefins, or building blocks for specialty polymers. Every batch (no matter which plant it comes from) carries the weight of years of tricky chemistry and plenty of environmental headaches. Flaring waste, scrubbing fluorinated byproducts, and cleaning up acid-washed vessels can create more mess than the original synthesis promises to solve. Many folks outside the industry believe chemicals like this spring fully formed into cans. In reality, each can reflects a web of choices about cost, safety, and cleanup nobody else sees.
Chemical Reactions: The Unseen Risks
Handling 1,1-Difluoroethane in a lab feels deceptively safe, and its resistance to spontaneous breakdown makes it seem like a stable solution. Even so, push it hard enough—expose it to flames, sparks, or strong oxidizers—and you unlock a world of toxic breakdown products. Hydrogen fluoride and carbonyl fluoride form with enough heat, both infamous for toxicity and corrosive power. Over the years, eager hobbyists and careless workers have triggered runaway chain reactions, sometimes inside closed HVAC systems. On the flip side, chemists rely on its stability for controlled polymerization, or crash it into other reagents to create fluorinated surfactants or propellants. Its byproduct potential can haunt a process, causing equipment wear and fouling when folks don’t anticipate corrosive traces. In plain terms, nobody respects a molecule until something goes sideways. This one rewards vigilance and punishes shortcuts with invisible toxins and long cleanup times.
Coding and Synonyms: Why Names Matter
Chemistry loves codes: HFC-152a, R-152a, Ethylidene difluoride. Synonyms hide in regulatory paperwork—sometimes it’s “Difluoroethane,” sometimes “1,1-DFE.” Even Material Safety Data Sheets hop between these names depending on international rules. On the shop floor, confusion happens when a worker reaches for a labeled can without realizing the same chemical may show up under a different codename in another department’s manual. For regulators and buyers, tracing synonyms affects everything from safety training to customs checks. Regulatory data links each synonym to rules on transport and handling. I’ve seen confusion slow shipments and sideline worker safety training, because some teams miss cross-references buried deep in documentation. Clear product names backed by universal codes can slash risk and help global trade keep its chemical promises straight.
On Safety and Working Standards
Plenty of people think the strict rules and standards around handling chemicals like 1,1-Difluoroethane don’t apply outside laboratory walls. Hard truth: any gas under pressure, especially with a track record of asphyxiation or fire risk, deserves strict oversight. Industry guidelines set maximum storage temperatures, spell out protective gear for routine maintenance, and demand leak tests for systems using the stuff in bulk. Regulatory boards—like OSHA and EPA—list exposure limits because repeated inhalation can cause central nervous system damage, even if it doesn’t leave you woozy on day one. Emergency response sheets call out frostbite risk on skin or eyes. Still, gaps in training or lack of respect for personal protective equipment lead to injuries every year, especially in less regulated settings like home garages or unauthorized resale markets. I’ve seen folks boast about “huffing” with zero regard for long-term consequences, all because folks warned them only about older, more toxic variants. Sound training and rigorous enforcement stand between regular use and tragic mishaps, not just padded gloves or warning stickers.
Everyday Uses: Where You’ll Find It
Most people cross paths with 1,1-Difluoroethane more often than they realize. The electronics industry banks on its ability to blast dust out of delicate sensors. Insulation foam makers use it as both a blowing agent and pressure carrier for specific panels. The refrigeration sector has pushed it as a transitional refrigerant, especially in older systems avoiding banned CFCs. As a propellant, it finds its way into aerosol cans for personal care and industrial cleaning. Folks in automotive repair lean on its properties for leak detection and safe inflating of tires. But every use carries a fingerprint of risk. Open an aerosol can indoors and your air quality changes in seconds. In well-ventilated factories, its use brings more benefit than danger, but in unventilated spaces, its asphyxiant power moves in fast. Even in consumer markets, the rules seem soft, but the risks are real. People treat it like ordinary air, forgetting its origins as an industrial refrigerant, not a toy.
Pushing Forward: Research, Chronic Risks, and Safer Paths
Lately, research circles buzz about 1,1-Difluoroethane’s long-term impact on health and climate. Toxicologists continue to document cases of frostbite and chemical pneumonitis in both industrial mishaps and accidental “huffing” episodes. The molecule does not pack the ozone-stomping punch of its chlorinated cousins, but its global warming potential still troubles regulators. Some journals link accidental deaths to deliberate inhalation or misuse in poorly designed ventilation systems. Engineers now work on detectors and failsafes that catch leaks in real time. Labs search for ways to neutralize or recycle spent gas instead of flaring it off. Advances in closed-loop recycling and process innovation show promise. The newest batch of fluorinated alternatives keeps coming, each one tested to reduce greenhouse effects or carcinogenic potential. Still, public perception lags. Many people do not realize new foam panels stuffed with HFC-152a trade one environmental issue for another. Industry reform starts with better outreach and education, not just government dictation.
What’s Coming Next?
Chemistry textbooks keep rewriting the story of refrigerants and propellants as climate science evolves. Regulators in Europe and California push toward phase-outs, even as other markets ramp up production for electronics packaging and niche foams. Companies invest in smaller, leak-resistant can designs and automated filling lines that minimize worker contact. Innovators work on bio-derived fluorochemicals designed to degrade more easily in the environment or fade faster from the atmosphere. Researchers also track down metabolic byproducts, worried about slow buildup in human organs after repeated exposures. Community science groups pressure regulators to label cans more clearly and slap warnings on products still popular in online “life-hacks.” As regulations tighten worldwide, 1,1-Difluoroethane’s role will keep adapting in response to safety data, environmental rules, and society’s shifting needs.
What is 1,1-Difluoroethane used for?
Everyday Uses and Industry Roles
I keep seeing 1,1-difluoroethane pop up in so many places in daily life. Most people probably know it from the back of canned air dusters — those sprays folks use to blast crumbs and lint out of keyboards. It works well for that simple job, and hardware shelves everywhere are stocked with it. That’s only the surface, though. This compound shows up in places you wouldn’t guess, finding a way into refrigeration, aerosol products, cleaning supplies, and even as a chemical in the plastics business.
What Makes It So Useful?
The chemistry behind 1,1-difluoroethane sets it apart. Picture a gas that turns to a liquid at the right pressure, evaporates fast, but doesn’t carry a heavy environmental load like older refrigerants, especially those with CFCs or high global warming potentials. Its molecular structure makes it less destructive to the ozone layer than some older cousins, even if concerns about long-term climate impact haven’t disappeared completely.
In refrigeration, it works as a blowing agent for foam and as a refrigerant itself in some systems. Companies choose it over a few other options because it’s safer for the ozone and doesn’t burn as easily as propane or butane. That’s pretty important in workplaces and manufacturing spots where accidents don’t stay small for long.
Aerosols Everywhere
Household sprays pack it in as a propellant. Deodorants, paint sprays, hair products, and cleaning cans depend on gases like 1,1-difluoroethane to get product out smoothly. The can empties without the chemical reacting with the formula inside, which makes it reliable. That reliability keeps factories and brands coming back to it, since recalls and device failures are the last thing they want.
Risks Behind Convenience
The easy availability and the fact that it’s a gas bring some real problems. A few years ago, news started covering a spike in accidental poisonings tied to inhaling canned air. People, especially kids and teenagers, looked for a quick high and underestimated the risks. Breathing it in causes major health trouble — sometimes seizures or even sudden death. The warnings are on every can, but misuse keeps happening.
There’s also pressure from regulators. Some countries pay close attention to any chemicals that threaten air quality or public health. Disposing of cans or managing leaks puts the spotlight on waste, both at home and on the factory floor. Factories have to balance their need for effective chemicals with environmental commitments and community safety.
How to Tackle the Drawbacks
The solution takes more than swapping to something else. Companies and households both have to change habits. Clearer safety education in schools matters, because teenagers often don’t know the risks until it’s too late. Industry groups can keep looking for safer propellants and start cutting back where they can. For people at home, safe storage and proper can disposal shrink risks.
Chemical giants run plenty of research for less hazardous alternatives, especially in new refrigerants and propellants. It’s not a simple task — each swap means retooling equipment and changing manufacturing lines. Consumers and small business owners can push for options with fewer climate worries and tighter safety controls.
The Path Forward
In the end, 1,1-difluoroethane delivers on a promise: easy use, solid safety compared to older options, and lower ozone risk. It’s not perfect. Better habits, more research, and smarter rules may get us closer to a world where we keep the convenience without the health or environmental headaches. The little details in our shopping carts and supply chains add up — and it’s on each of us to stay alert, ask questions, and be open to change.
Is 1,1-Difluoroethane safe to use?
No Silver Bullet for Chemical Safety
A lot of folks ask about 1,1-difluoroethane because you see it everywhere these days. The compressed gas sits inside a can on your desk at work, shooting away dust from keyboards. People see it labeled as an ingredient in products like canned air, and that’s pretty much where they leave their curiosity. But safety questions stick around, because everyone wants to trust the stuff they use at home or in the shop. If you’re wondering whether it’s safe, the truth isn’t black and white.
The Role of 1,1-Difluoroethane
As an ingredient, 1,1-difluoroethane serves as a propellant. Its appeal comes down to performance—it helps force whatever’s inside the can out with a strong blast, making cleaning easy. It’s not considered an ozone-depleting chemical, unlike some old-school propellants. For industrial and consumer use, 1,1-difluoroethane solved the problem of finding something powerful, easy to use, and less damaging to the planet’s upper atmosphere. On paper, this switch sounds like a win for the environment and convenience.
What Science Tells Us About Health Risks
Every chemical deserves a closer look. With 1,1-difluoroethane, in typical, short bursts—like cleaning electronics—it doesn’t usually cause much harm. If you use the product following the instructions, indoors with decent ventilation, the risk of serious side effects goes down a lot. But risks aren’t zero. Some folks have tried inhaling 1,1-difluoroethane on purpose to get a high. This isn’t just misuse; it’s downright dangerous. Inhalation can cause your heart to go haywire. I’ve seen cases in local news where kids ended up in the emergency room or worse after “huffing” canned air. 1,1-difluoroethane cuts oxygen in your lungs and can trigger sudden cardiac arrest, even for someone young and healthy.
Contact with skin doesn’t go unnoticed either. This propellant can freeze your skin or cause frostbite because it escapes the can at a low temperature. If you or someone around you sprays it too close to their body—either by mistake or while goofing around—they come away with skin damage.
Environmental and Fire Hazards
Questions about environmental safety deserve answers. 1,1-difluoroethane leaves a much smaller carbon footprint than CFCs, but it’s still a greenhouse gas. That means releasing a lot of it into the air adds to climate problems over time. Safety in storage also matters. The gas stays highly flammable. A can left near a heater or punctured on accident creates a risk for fire or explosions. You don’t need a lab coat to see the problem—household products should never surprise people with an explosion risk.
Smart Practices for Safer Use
Learning how to use chemical products safely happens in real settings, not just from a label. I make sure to open windows while spraying, keep the can away from heat, and stash it where kids can’t reach. Reading the safety instructions on the can deserves attention too. I toss empty cans in hazardous waste—not straight in the bin.
Better education about what this chemical can and can’t do may keep accidents out of headlines. If manufacturers put clearer warnings front and center, fewer people might treat 1,1-difluoroethane as harmless. Supervising young people, keeping products out of reach, and sticking to careful habits go a long way. Nothing beats real caution and respect for chemicals, even ones with familiar brand names.
What are the health risks of inhaling 1,1-Difluoroethane?
A Closer Look at What We Breathe
1,1-Difluoroethane doesn’t pop up in daily conversation, but flip over an aerosol dust cleaner or gas duster can from any computer store, and there it is. It comes out as a spray, meant to clear dust from keyboards and fans. The trouble doesn’t start with cleaning. Problems show up when folks inhale the stuff on purpose, chasing a quick high or “huff.” That’s when questions about safety aren’t so easy to shrug off.
What Happens in the Body
The lungs draw in 1,1-Difluoroethane, and it quickly passes into the bloodstream. Blood flows straight to the brain and the body’s organs, telling them things are not right. Instead of getting oxygen, tissues start soaking up this chemical. Hearts beat faster or slower at random. In the brain, signals get scrambled, leading to dizziness, loss of coordination, slurred words, and in some cases, loss of consciousness. Even one deep breath can set this off.
Immediate and Short-Term Risks
The main worry with 1,1-Difluoroethane is sudden sniffing death syndrome. The heart can suddenly stop, even in people with no known health problems. This isn’t an urban legend. Emergency rooms have seen teens and young adults roll in, unresponsive, because their heart just gave out after inhaling a single whiff. It can also make breathing difficult by triggering airway spasms or choking.
On top of heart trouble, the high comes with its own risk of injury. Judgment drops, balance fades, and reaction times slow to a crawl. People using this chemical have blacked out, fallen, hit their heads, or even drowned from passing out near water. There’s a real loss of control in those moments—something that seems small but grows dangerous fast.
Long-Term Consequences That Don’t Go Away
Repeated inhalation chews away at the nervous system and damages organs. Studies from poison control centers and hospitals show regular users can experience lasting memory loss, tremors, and problems concentrating. The liver and kidneys get overworked pulling toxins out, but that work comes at a steep cost: tissue damage and scarring build up over time. Skin around the mouth and nose will sometimes develop frostbite, since this gas leaves the can so cold it burns.
What Drives People to Huff?
Accessibility plays a big role. No one checks ID for cleaning products in most stores. The same can doesn't just clean—it tempts kids and teens because it’s cheaper and easier to get than alcohol or tobacco. Peer pressure and lack of good information turn experimentation into tragedy.
Reducing the Risk
Any solution has to start at home and in classrooms, where conversations happen without shame or judgment. Kids need more than horror stories—they need facts, examples, and proof that people care about what they’re facing. Retailers need clear warning labels, and sometimes, these cans get locked up behind the counter. This doesn’t solve everything, but it slows down easy access.
For someone already struggling, mental health resources make a big difference. From my own work with youth counseling, sharing honest stories and opening the door to mental health support helps more than scare tactics ever did. Recognizing the pattern early and helping people find safe outlets can save lives. Focusing only on punishment misses those who most need a hand back onto safer ground.
Bottom Line
Breathing in 1,1-Difluoroethane for a cheap thrill isn’t worth what it takes from the body. Education, caring conversations, and safer access rules matter more than ever with chemicals that slip so quietly into the background of daily life.
How should 1,1-Difluoroethane be stored?
Understanding the Real-World Risks
1,1-Difluoroethane sounds technical, but many folks have crossed paths with it in canned air dusters or refrigerants. Found in workshops, labs, and homes, it stands out as a common product with uncommon dangers. As a writer who’s spent years reporting on workplace safety and chemical incidents, I’ve seen what happens when shortcuts shape the way harmful chemicals get stored. Ignorance isn’t just unsafe—it’s costly, sometimes tragic.
The Chemical Safety Board often points out mishaps tied to flammable gases stored in the wrong spots or with the wrong gear. 1,1-Difluoroethane comes with a flash point below room temperature. That means even a slight increase in warmth can help it leap into flame. Add its knack for quickly building up vapor and you’ve got a recipe for disaster unless folks approach storage with precision.
The Reality of Flammable Gas Storage
I once toured a warehouse with rows of gas cylinders stacked in a dark, locked cabinet near a boiler room. Trouble waited in those shadows. Heat, static, and poor ventilation turned a storage choice into a potential headline. Every year, fire marshals respond to places just like that, where mistakes spring from haste or a lack of info. Flammable gases don’t wait around for teaching moments.
Anyone handling 1,1-Difluoroethane has to keep it clear of sparks, open flames, and heat sources. It sits volatile, so direct sunlight is a no-go. Store it inside a well-ventilated shed or room, away from high-traffic paths. Letting vapors gather spells real danger—these vapors can flow, settle, or build up, ready to ignite at a stray spark.
The Right Containers and Labels Matter
Manufacturers fill those metal cans and cylinders for a reason. They’re rated for pressure, capped against accidental discharge, and marked as hazardous. Swapping original containers for something “more convenient” just courts leaks and confusion. At my first reporting job, I saw what happened after a supervisor transferred chemicals into a soda bottle. No label, no warning… and a trip to the emergency room.
So, keep products in their original cans or pressure-rated cylinders. Never tamper with safety caps or try to transfer the gas. Make sure labels face out and stay clear. A clear label with hazard symbols goes a long way if emergency crews show up. Follow the manufacturer’s sheet—every can comes with one.
Common-Sense Solutions That Save Lives
Take inventory often. Know what gets stored, how much, and where it lives. Post emergency shut-off procedures and keep an extinguisher handy—one rated for chemical fires, not granny’s kitchen model. A few local companies set a strong example by assigning a staff member to walk through chemical storage once a week, checking for signs of leaks or aging containers.
Training everyone—janitors, managers, front-desk staff—means more eyes and quicker action. Spills and leaks need swift response. Proper ventilation, good signage, and routine checks lower the odds of trouble. Often it’s the overlooked, boring little tasks that sidestep scares and lawsuits.
Local fire departments run community inspections all the time. Opening the door for an inspector and listening to their advice beats pleading ignorance in the middle of a crisis. Cities and states lay out rules on hazardous materials for a reason—they’re written in the shadow of past accidents. Follow the rules and lives get safer, work gets easier, insurance rates stay reasonable.
Is 1,1-Difluoroethane flammable?
What 1,1-Difluoroethane Brings to the Table
1,1-Difluoroethane pops up in a lot of places—think compressed air cans for dusting keyboards, refrigerants, and even as a propellant in consumer sprays. Sounds simple, but this stuff calls for respect. People tend to grab a can from any hardware store without much thought. Trouble comes when folks treat it like it’s as harmless as canned whipped cream. That’s simply not the case. If you ever cracked open a can in your garage, you might notice a chemical smell and a blast of vapor that feels colder than an ice cube. Now, let’s talk about flammability, since that question keeps turning up among both hobbyists and people curious about safety at home or work.
Lighting the Facts: Flammability and Real-World Risk
Ask any chemist, and you’ll hear 1,1-difluoroethane absolutely does catch fire. The chemical formula C2H4F2 hints at its cousin, ethane, which anyone who used a Bunsen burner in high school will recognize as highly flammable. The Occupational Safety and Health Administration points out that difluoroethane has a lower flammability limit in air—around 3.9%. That means even a tiny leak in a closed garage or office, followed by a spark, could cause a dangerous flash.
Plenty of injuries happen from people spraying the gas around electronics or open flames, not realizing how easily it can ignite. I’ve seen folks use compressed gas to clean out computers, and if the room doesn’t have enough airflow, static from electronics or a heater can quickly cause a burst of fire. It’s not just a story for safety manuals—there are published incidents and even some news stories about small explosions in homes and workshops due to this exact scenario.
Why the Chemistry Matters in the Real World
1,1-Difluoroethane’s low boiling point means it evaporates faster than water spills on a summer sidewalk, which lets the vapor linger in the air. A spark from a light switch or drop of static electricity sometimes does the trick. Beyond personal injuries, these incidents can also damage property badly. Firefighters deal with burned homes and offices every year because of these fires. A few years back, the Environmental Protection Agency issued warnings after finding that people, especially teenagers, tried to inhale refrigerants or propellants for a quick buzz. That increases risk by letting clouds of vapor form in closed rooms, making an accidental flash fire more likely if someone lights a cigarette or flicks a faulty switch nearby.
Solutions and Smarter Behavior
This isn’t a situation calling for complicated rules only big factories would bother to follow. If you’ve got a can of the stuff, keep windows open and fans running. Leaving electronics unplugged during cleaning and never pointing a stream of gas at open flames—simple steps help. Retailers and manufacturers can do their part with clearer hazard labels and better instructions. Having seen warning labels get skimmed or ignored, I know pictures and direct language work better than paragraphs of legal jargon.
Schools and workplaces should offer quick training on chemical safety, not just for science teachers or lab techs, but for anyone who handles office cleaning. Parents and educators can talk openly with teens about inhalant abuse—not to scare, but to explain real risks without judgment. The science stays the same, but our habits can shift to keep accidents off the front page. Practical steps save lives, properties, and hospital bills. That’s a lesson worth passing on, no matter the setting.
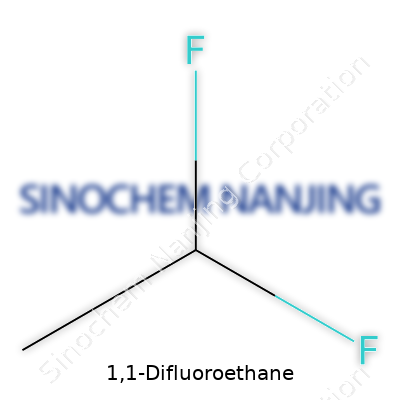
| Names | |
| Preferred IUPAC name | 1,1-Difluoroethane |
| Other names |
Ethyl fluoride HFC-152a Difluoroethane R152a Ethane, 1,1-difluoro- Freon 152a |
| Pronunciation | /ˌwʌnˌwʌn daɪˌfluːroʊˈɛθeɪn/ |
| Identifiers | |
| CAS Number | 75-37-6 |
| 3D model (JSmol) | `JSMOL_MODEL "C(C)(F)F"` |
| Beilstein Reference | 1900225 |
| ChEBI | CHEBI:39037 |
| ChEMBL | CHEMBL137750 |
| ChemSpider | 6616 |
| DrugBank | DB00128 |
| ECHA InfoCard | 03b5e11a-cf33-4cdc-8c14-bc055b8308c3 |
| EC Number | 200-866-1 |
| Gmelin Reference | 1692 |
| KEGG | C06585 |
| MeSH | D000926 |
| PubChem CID | 6368 |
| RTECS number | KI1100000 |
| UNII | 1SPLH8570P |
| UN number | UN1030 |
| Properties | |
| Chemical formula | C2H4F2 |
| Molar mass | 66.05 g/mol |
| Appearance | Colorless gas |
| Odor | Mild ether-like |
| Density | 0.9 g/cm³ |
| Solubility in water | Solubility in water: 1.97 g/L (at 25 °C) |
| log P | 0.75 |
| Vapor pressure | 4250 mmHg (20°C) |
| Acidity (pKa) | 25.0 |
| Basicity (pKb) | pKb: 15.42 |
| Magnetic susceptibility (χ) | -31.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.210 |
| Viscosity | 0.153 cP (25 °C) |
| Dipole moment | 2.41 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | (298) = 292.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -285.03 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1514.7 kJ/mol |
| Pharmacology | |
| ATC code | R20FA12 |
| Hazards | |
| GHS labelling | GHS02, GHS04 |
| Pictograms | GHS02,GHS04 |
| Signal word | Danger |
| Hazard statements | H220, H280 |
| Precautionary statements | P210, P261, P304+P340, P312, P377, P381 |
| NFPA 704 (fire diamond) | 1-4-2 |
| Flash point | -58 °C |
| Autoignition temperature | 454 °C |
| Explosive limits | 3.9% - 16.9% |
| Lethal dose or concentration | LCLo human inhalation 1500 ppm/4H |
| LD50 (median dose) | LD50 (median dose): 86,400 mg/kg (rat, oral) |
| NIOSH | NIOSH: KI1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,1-Difluoroethane: "1000 ppm (TWA) |
| REL (Recommended) | 0.1 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Fluoroethane 1,1,1-Trifluoroethane Chlorodifluoroethane 1,2-Difluoroethane Ethane |

