Looking Beyond the Lab: The Story and Significance of 1,1-Dichloro-1-Nitroethane
The Journey of Discovery
Folks like to trace the roots of any chemical compound that drifts out of a beaker and into wider industrial use, and 1,1-Dichloro-1-Nitroethane has a story that feels both straightforward and tangled. The earliest references get dusty, buried in mid-20th-century texts as chemists tried to squeeze out new organochlorines and nitro-alkanes for use in synthesizing pesticides, solvents, and intermediates for more complicated molecules. These weren't years when safety sat front and center. Curiosity and commercial promise ran the show. Over time, as more eyes focused on the safer handling and unintended consequences of these chemicals, the push moved from "What can we make?" to "How do we make and use it wisely?" The lesson keeps echoing: the earlier chase for function now shares the spotlight with safety and sustainability.
The Stuff Itself: What Sets 1,1-Dichloro-1-Nitroethane Apart
Walk into a lab where 1,1-Dichloro-1-Nitroethane works as an intermediate, and you meet a pungent, yellow-to-brown oily liquid with a weighty feel and volatility that keeps you alert. The molecule's structure—a two-carbon chain carrying both nitro and dichloro groups on the same carbon—isn’t common, and that sets up its chemical behavior. For chemists, tucked in those nitro and chloro groups are doors to further reactions: reductions, substitutions, nucleophilic attacks. The boiling point sits higher than simpler chloroethanes, though not high enough to relax about inhalation risk or the volatility hazard. As a solvent or precursor for broader chemical synthesis, it's not the stuff you want lingering on skin or in poorly ventilated corners.
Physical & Chemical Realities
Handling this compound, I’ve noticed how its strong odor mandates a cautious approach. Its density and immiscibility with water drive separation in classic extraction operations. It doesn’t mix like alcohols or basic halocarbons, and that complicates cleanup and containment. Combustibility doesn’t typically rank high because it’s hard to ignite, but under breakdown or fire, it drags along both chlorine and nitrogen oxides, neither of which you want set loose indoors or up a vent stack. In the real, gritty details of chemical process work, those physical quirks mean strict standard operating procedures and decent personal protective equipment, beyond a simple gloves-and-goggles policy.
Preparation and Transformation: How Chemists Shape and Change It
The old school way of making 1,1-Dichloro-1-Nitroethane involves direct chlorination of nitroethane, usually in the presence of a suitable catalyst to drive substitution at the right carbon. This reaction can throw off multiple side products like 1,2-dichloro-1-nitroethane or polychlorinated bits, so precision, temperature control, and constant monitoring matter. It’s a balancing act between conversion and selectivity. Add too much chlorine or run the reaction hot, and you cook up a mess that's tedious to clean up. Turn the temperature down and conversion stalls. Any chemist who’s wrestled with old glassware and batch distillation knows that scaling up brings out all the hidden bugs in the process. As for modifications, the presence of two electron-withdrawing groups invites reductions to the corresponding amine, or nucleophilic substitutions that can build out new scaffolds. That flexibility makes it handy for synthesizing niche products in pharmaceuticals or crop protection.
Synonyms and Product Names
One of the annoyances is sorting through a thicket of naming conventions—1,1-Dichloro-1-Nitroethane sometimes crops up as alpha,alpha-dichloro-nitroethane, or under more obscure registry numbers in vendor catalogs. For folks in synthesis or regulatory compliance, clear identification is a time-saver and safety imperative. No one wants confusion when ordering or labeling, especially not when multiple isomers and closely related substances lurk in supplier lists.
Living with the Hazards: Standards for Safety and Operation
You learn early not to get casual around these chemicals. Shortcuts don't work. From the start, handling guidelines call for impervious gloves and splash goggles, and a willingness to treat every drop as potentially harmful until you know exactly what's in your flask. Chlorinated nitroalkanes are reactive, slippery, and energetic when provoked. Ventilation systems should hum, local exhaust rules out, and detectors track vapor. Most labs lean on fume hoods and spill kits for a good reason. Waste management is another puzzle: you need to keep byproducts and solvent remains out of regular sinks, keeping everything above board with waste treatment vendors.
Where It Finds Use
Despite its pungency and challenge, 1,1-Dichloro-1-Nitroethane has elbowed into specialty syntheses, especially where you want a reactive starting material to push toward new heterocycles, or to make stepwise halogenated intermediates. Pesticides, pharmaceuticals, and fine chemicals chemists tap its unique mix of reactive centers, especially as a way to furnish carbon skeletons bristling with functional groups. Its direct use in consumer goods is nearly nonexistent, mostly for reasons of hazard, so its impact stays behind the curtain, woven into the supply chains that churn out more familiar end-products.
The Ongoing Quest for Knowledge: Recent Research and Development
Academic and industrial labs keep poking at safer and greener routes to 1,1-Dichloro-1-Nitroethane. Folks examine lower-emission processes, try to wring higher yields from milder conditions, and parse out routes that cut down on toxic byproducts. Finding catalysts that work at lower temperature or in water instead of organic solvents could mark real progress. Computer modeling opens up new predictions on how to modify the molecule, or how it interacts with biological systems—a step toward evaluating not just what works in a flask, but what impacts could ripple out through the ecosystem or across workers’ health. Some research aims to replace functionally similar compounds with less hazardous profiles, though chemical inertia and equipment cost put real-life adoption on a slower track.
Puzzle Pieces: Toxicity and Human Health
The biggest reason to watch your step with 1,1-Dichloro-1-Nitroethane traces back to its acute and chronic toxicity. Chlorinated nitroalkanes often deliver a one-two punch—both respiratory and liver toxicity present serious risks in exposure incidents. Animal studies flag its ability to cause tissue injury, and the nitro group’s presence rings alarm bells for potential mutagenicity. Regulations still lag behind emerging knowledge, so it's up to chemists, EHS staff, and production workers to keep the risks front of mind. Environmental fate, bioaccumulation, and long-term effects demand open questions and further study. You can’t protect what you don’t understand, and no industry stays honest by brushing off inconvenient toxicology findings.
Peering Ahead: What the Future Holds
Unlike some bulk commodity chemicals, 1,1-Dichloro-1-Nitroethane isn’t vanishing or heading into the chemical sunset; its niche applications remain stubbornly useful. But the horizon tilts toward tighter controls, greater transparency on health impacts, and real innovation in synthetic chemistry. If better, greener alternatives climb the regulatory and economic hurdles, expect the old standards to lose ground. The drive now rewards not just the cleverest synthesis, but the synthesis that accounts for downstream safety and environmental justice. For anyone still working in this corner of industrial chemistry, the future rides on adaptability—balancing utility, responsibility, and the willingness to walk away from yesterday’s convenience when something better stands ready.
What is 1,1-Dichloro-1-Nitroethane used for?
The Role of 1,1-Dichloro-1-Nitroethane in Industry
Many people, including those of us who spend time working in or researching chemistry labs, hear about specialty chemicals like 1,1-dichloro-1-nitroethane and wonder what purpose it serves. While it’s not something the average person runs across in a grocery store, this compound fills a very particular role in chemical manufacturing. Its main use surfaces in the field of organic synthesis, especially as an intermediate in the production of pharmaceuticals and agricultural chemicals. Companies that design synthesis pathways need building blocks tough enough to handle a range of reactions but specific enough to create targeted molecules. In my time talking with colleagues in pharmaceutical development, it’s clear that specialized chemicals speed up drug discovery by allowing researchers to explore new compounds without starting from scratch every time. 1,1-dichloro-1-nitroethane, with its reactive structure, makes it easier to tack on other chemical groups and build out the molecules in demand.
Importance in Research and Innovation
Research teams in both academia and industry need access to obscure chemicals to push the boundaries of what’s possible in synthesis. When a new crop-protecting agent or possible treatment for disease appears in the scientific literature, chances are good that the journey began with steps like halogenation and nitration — the classic moves that 1,1-dichloro-1-nitroethane enables. Some of my most exciting moments as a student came from critically examining these tiny intermediates and realizing their broad consequences. Without them, progress toward new compounds drags, and whole classes of potential products remain left on the drawing board.
Handling and Hazards: More Than Just a Lab Story
Safety matters as much as innovation. This sort of chemical doesn’t belong on a kitchen shelf. Like many nitro and chloro compounds, it brings some risk — including toxicity and environmental persistence. Responsible researchers stress proper storage, careful measurement, and personal protective equipment not because anybody enjoys rules, but because we respect the risks. Improper handling or disposal of 1,1-dichloro-1-nitroethane threatens water, soil, and air. In my experience, clear labeling and regular team safety briefings prevent more accidents than any single warning label. Once, a misplaced sample led to extra time scrubbing down a fume hood, reminding everyone that shortcuts in handling add up to real trouble.
Moving Forward: Best Practices and Regulation
Countless specialty chemicals, including 1,1-dichloro-1-nitroethane, call for ongoing vigilance. Real-world impact comes from rigorous training, transparent incident reporting, and an insistence on compliance with local and international safety guidelines. My own work improved once I started regularly consulting safety data sheets and keeping communication strong with environmental health staff. Companies and university labs can do more by keeping inventories current, switching to greener alternatives when possible, and investing in advanced containment equipment. Regulators should keep up with the shifting landscape, making sure that oversight stays as nimble as the science itself. At the end of the day, safe and responsible use of specialty chemicals makes not only better science, but a safer world for all of us.
What are the safety precautions when handling 1,1-Dichloro-1-Nitroethane?
Understanding the Risks
Anyone who has set foot in a chemistry lab learns pretty fast which chemicals demand a little extra respect. 1,1-Dichloro-1-nitroethane belongs on that list. Spend enough time around chemicals like this, and you start to see the difference between routine and risky. Inhaling its vapors or spilling it on your skin leaves real consequences. Few things wake you up faster than realizing you’ve overlooked a basic rule—like suiting up before opening the container.
Personal Protective Gear Isn’t Optional
Getting lazy with gloves or goggles triggers accidents nobody wants on their resume. Double-layering nitrile gloves makes a difference—latex alone isn’t always enough against harsher organics. There’s a reason lab coats (with closed cuffs), chemical splash goggles, and face shields sit in every well-run work area. I’ve seen what happens when someone skips goggles with this class of compounds: eye irritation that doesn’t wash away quickly, sometimes worse.
Ventilation and Spills
A single whiff lets you know 1,1-dichloro-1-nitroethane isn’t meant for enclosed spaces. Fume hoods aren’t just for show—chemists use them daily to avoid breathing vapors. With solvents and nitro compounds, I keep an open box of activated charcoal handy for quick absorption. On the rare occasion a spill happens, containment beats panic. Industrial absorbent material soaks up much more compared to tissues or towels. Everything gets sealed in a hazardous waste bag—I’ve watched more than one person regret tossing contaminated rags in regular trash.
Storage Practices
You don’t want to fish through a cluttered cabinet with bottles stacked together in chaos. Segregation matters. Chlorinated and nitro compounds ask for their own containers, away from acids or strong bases. Metal cabinets with self-closing doors serve as solid insurance, limiting exposure if something leaks or breaks. I store things straight at eye level, never below waist or above shoulder height, to reduce the chance of knocking something over.
Response Training Saves Time and Trouble
Experience counts for a lot but so does preparation. Reading the safety data sheet takes less than 10 minutes and can save someone from a trip to the ER. I’ve practiced mock spills in workshops—muscle memory means faster reactions. Keeping eyewash stations and safety showers within arm’s reach pays off when seconds make the difference between mild and serious injuries.
Labeling and Communication
Maybe the label faded, or someone borrowed the bottle for an experiment—those are the moments where discipline slips and accidents creep in. Clear, fresh labels and regular reminders about chemical hazards keep the team alert. At my last lab, we ran through a daily whiteboard checklist covering what we all handled that day. That habit flagged possible mix-ups early.
Safer Alternatives and Continual Learning
In some labs, alternatives get tested to dodge the risk entirely. Green chemistry principles guide researchers to less toxic or volatile options. Whenever possible, switching to a safer compound means fewer safety headaches. No matter your experience level, training never stops. Annual refreshers and peer-to-peer reviews keep good habits alive, while sharing stories about close calls reinforces why people take safety seriously.
The bottom line: risk doesn’t disappear through luck or shortcuts. Practice, gear, a solid sense of responsibility, and open communication all raise the odds that people walk out of the lab healthy. That’s a lesson worth repeating, every single day.What is the chemical formula of 1,1-Dichloro-1-Nitroethane?
Looking Beyond a String of Letters
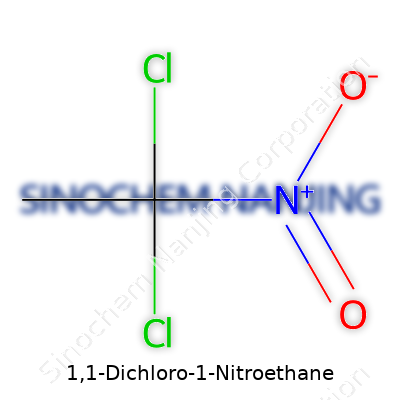
I remember sitting in high school chemistry, staring at endless lines of compounds, trying to make sense of twisting names and cryptic formulas. Teachers rattled off examples, but nobody stopped to ask how those names came to mean so much. The formula for 1,1-Dichloro-1-Nitroethane, for example, is C2H3Cl2NO2. Folks encounter these names in textbooks or obscure online articles, but few realize why such a chemical ends up being more than the sum of its atoms.
Why 1,1-Dichloro-1-Nitroethane Is Worth a Closer Look
C2H3Cl2NO2 spells out more than just the makeup of a compound. The structure matters, especially if you work in fields ranging from pharmaceuticals to environmental labs. Two chlorine atoms attach to the same carbon (that’s what the “1,1-” means), and a nitro group connects to that very spot as well. What's left is a methyl group, and all of it hung together by single bonds. This arrangement shapes how this compound reacts, breaks down, and influences its risk profile—a detail sometimes brushed under the rug in the race to find the next new material.
The Human Side of Handling Chemicals
Years back, I worked with a small research group studying chlorinated nitroalkanes. The labs were stuffy, and the risk assessments never felt complete. A formula like C2H3Cl2NO2 suddenly means you’re juggling volatile ingredients. The chlorine and nitro groups boost both reactivity and toxicity. We needed thick gloves and extra ventilation, not just to check regulatory boxes, but to protect ourselves from headaches and nausea.
Plenty of students and workers forget that behind each compound name there’s a set of health implications. Chlorinated nitro compounds often linger in air and soil, sticking around long after experiments end. Toxicity concerns don’t stop once the waste jug gets sealed.
Fixing Gaps in Chemical Knowledge
Folks often treat chemical formulas as trivia, but the details carry weight. C2H3Cl2NO2 helps point out hazards that may feel invisible. To keep people safe, information sharing must grow—guidelines in plain language, visible signage, and training that shows not just what to use but why. Effective communication matters. It’s easier for people to respect a danger if they truly see it, not just memorize numbers and letters.
Solutions Rooted in Everyday Lab Work
Instead of locking away details in technical jargon, labs and classrooms would do better to connect the dots. Make the chemistry personal: What does this formula mean if it spills? How do its ingredients break apart? Committee-written policies matter less than real conversation in training rooms, where a formula like C2H3Cl2NO2 becomes fuel for discussing responsibility and care. Put the facts out in the open, and respect for the risks and uses will follow.
How should 1,1-Dichloro-1-Nitroethane be stored?
Understanding the Risks
A lot of folks who handle chemicals every day know that carelessness never pays. 1,1-Dichloro-1-nitroethane is no exception. It comes with a reputation—volatile, toxic, a possible threat to both health and the environment. Handling and storing it wrong can spark a string of headaches: health incidents, environmental spills, or even lawsuits.
I’ve seen people in labs treat unfamiliar bottles like household bleach. That comfort can quickly turn into disaster because this compound will not play nice if stored in the open, near heat, or among incompatible materials. Small mistakes accumulate, and even a harmless-looking drum can ruin your week when fumes leak or a spill gets outside your fume hood.
Why Storage Matters
The numbers on workplace injuries all point to poor storage as one of the most common causes. According to the US National Institute for Occupational Safety and Health (NIOSH), thousands of chemical accidents in the past decade started because someone overlooked a label or skipped a safety check. Storing toxic substances in the wrong spot can produce hidden threats—reacting with air, water, or even just the wrong kind of container.
Long before you unscrew the lid, the risk starts in your storage plan. If you’re aiming for a safe lab or warehouse, each step matters. I once watched a colleague pour a solvent into a plastic jug, thinking it would hold up just fine. That mistake brought on chemical burns, lost time, and a visit from the hazmat crew.
Real-World Storage Tips
Keep Containers Tight and SteadyAny bottle of 1,1-dichloro-1-nitroethane needs an airtight seal. Vapors from nitro compounds can build up quick and silently, so tight lids and well-maintained seals are your first line of defense. Store only in manufacturer-approved containers—no improvising or old reused jars. Glass with chemical-resistant caps works best in most lab settings, but always double-check compatibility.
Cool, Dry Storage Reduces TroubleHeat invites trouble with volatile compounds. I’d never trust 1,1-dichloro-1-nitroethane near a window or close to equipment giving off warmth. Aim for room temperature or lower—it hinders evaporation and slows down unwanted reactions. Avoid humidity; moisture can spell corrosion or degradation. A ventilated chemical storage cabinet, away from sunlight and regular foot traffic, usually does the trick.
Separate from IncompatiblesNever stash it with strong bases, oxidizers, or other nitro compounds—mixing can lead to violent reactions. Most labs carry reference charts or digital checklists; take a minute to check them before unpacking anything new. I remember pulling apart a cabinet stuffed with conflicting chemicals. That poor storage plan nearly cost us a serious incident.
Label Everything Clearly
Uncertainty means danger. Always slap a clear, weatherproof label on every container. Include the full chemical name, hazards, date received, and emergency info. Rushed or faded labels cause confusion during emergencies. I’ve seen responders lose time searching for basic information; don’t let that happen on your watch.
Plan for Problems
Even if you follow every guideline, leaks and spills can still happen. Keep spill kits stocked near your storage area—absorbent pads, proper PPE, and instructions for cleanup. Everyone in the area should know evacuation routes and actions to take if a container cracks or vapor alarms sound. Training stays fresh with regular drills, not just a one-time handout.
Legal and Community Responsibilities
Check federal, state, and local rules for hazardous chemical storage. The EPA, OSHA, and local fire departments all set their own requirements. Ignoring these can mean steep fines and damaged trust in your operation. People living nearby count on you to keep hazardous chemicals locked down—clean water, safe air, and health depend on it.
Is 1,1-Dichloro-1-Nitroethane hazardous to health or the environment?
Understanding 1,1-Dichloro-1-Nitroethane
The moment we hear about chemicals like 1,1-dichloro-1-nitroethane, many of us wonder if it’s another one to keep off the shelf at home or in the workplace. This compound belongs to the broader family of chlorinated nitroalkanes, a group known for their industrial uses and, in some cases, their sorted track record regarding health and safety.
Health Impacts at a Glance
Over the years, people working with small but potent molecules like this have raised concerns about breathing problems, skin irritation, and even headaches after exposure. If a person breathes in vapors from chlorinated nitro compounds, or if these chemicals make contact with skin, reactions can follow. Though research into this specific compound isn’t as thorough as with other industrial chemicals, we can draw lessons from related substances. For instance, folks in manufacturing settings have run into dizziness and, after long stretches of contact, even liver troubles with some chemicals in this family.
A big worry comes from long-term exposure. I remember a time touring a chemical facility where they stressed how even small leaks caused staff to cough and leave the area to catch their breath. Chlorinated nitro compounds often slip through the air quickly, carrying risk well beyond a simple spill. Eyes and the respiratory system take the hit first. Consistent protective gear and good ventilation matter a lot.
Environmental Footprint
Let’s turn to the environment. Chemicals like 1,1-dichloro-1-nitroethane don’t simply vanish once used. If they get into soil or water, they linger and can harm aquatic life. A spill near a stream runs the risk of fish kills and disruptions to the surrounding habitat. Chlorinated compounds can break down slowly, meaning rivers, lakes, and groundwater stay at risk for years if contaminated.
Pollution isn’t only a local issue. Once released, chemicals can drift and settle on farms or in neighborhoods miles from the original site. I’ve heard farmers complain about mysterious plant die-offs near drainage ditches. Tracing the issue back to chemical seepage often brings up substances like this—chemical companies hold responsibility for keeping their emissions in check to avoid such unintended effects.
What Can Be Done?
Keeping people safe requires strict control measures. Safe storage, spill-proof containers, and real-time air monitoring ought to be the standard. Workers should know the dangers, with quality goggles and gloves at the ready. Training matters—basic first-aid, quick access to clean air, and the know-how to spot early signs of trouble can save lives.
There’s a push for tighter regulation and for greener alternatives. Green chemistry, with less persistent and less toxic ingredients, offers real hope. Some companies have started switching to chemicals that break down more quickly or don’t linger in human tissue. This transition came not from big legislation right away, but from workers and their families pushing hard for safer choices.
Monitoring doesn’t stop at the gate. Regular sampling of local water, soil, and air works as an early warning system. Without this, small leaks or spills go unnoticed until real harm shows itself. Community oversight and transparent sharing of chemical data build trust and put pressure on users and manufacturers to act responsibly.
Looking Ahead
Dealing with 1,1-dichloro-1-nitroethane means acknowledging its risks and shouldering responsibility, both in the factory and in our backyards. The story repeats with each hazardous chemical: safety starts with good habits, honest communication, and a bit of caution, especially for those living or working near the source.
| Names | |
| Preferred IUPAC name | 1,1-dichloro-1-nitroethane |
| Other names |
1,1-Dichloroethyl Nitrite Ethane, 1,1-dichloro-1-nitro- Nitroethylidene chloride |
| Pronunciation | /ˈwʌn ˈwʌn daɪˌklɔːroʊ ˈwʌn ˈnɪtroʊ ˈɛθeɪn/ |
| Identifiers | |
| CAS Number | 594-70-7 |
| Beilstein Reference | 1209283 |
| ChEBI | CHEBI:140339 |
| ChEMBL | CHEMBL21736 |
| ChemSpider | 197859 |
| DrugBank | DB02837 |
| ECHA InfoCard | 03bc5f2f-c583-4e20-b1e0-2a56ba3deb02 |
| EC Number | 213-082-6 |
| Gmelin Reference | 8329 |
| KEGG | C19287 |
| MeSH | D004055 |
| PubChem CID | 13267 |
| RTECS number | KH8225000 |
| UNII | U6J42K00D6 |
| UN number | UN3385 |
| Properties | |
| Chemical formula | C2H3Cl2NO2 |
| Molar mass | 141.02 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet |
| Density | 1.36 g/mL at 25 °C (lit.) |
| Solubility in water | slightly soluble |
| log P | 0.91 |
| Vapor pressure | 3.2 mmHg (25 °C) |
| Acidity (pKa) | 16.3 |
| Magnetic susceptibility (χ) | -46.2×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.454 |
| Viscosity | 1.191 cP (25°C) |
| Dipole moment | 2.73 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 341.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -171.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -628.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H302, H315, H319, H335, H411 |
| Precautionary statements | P261, P264, P271, P273, P301+P312, P305+P351+P338, P308+P311, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 65°C |
| Autoignition temperature | > 535°C |
| Explosive limits | Explosive limits: 4.4–16% |
| Lethal dose or concentration | LD₅₀ Oral Rat 312 mg/kg |
| LD50 (median dose) | 100 mg/kg (rat, oral) |
| NIOSH | SN2100000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: Ca |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Chloropicrin Nitroethane 1,1-Dichloroethane Nitromethane 1,2-Dichloroethane |

