1,1'-Azobis(Cyclohexanecarbonitrile): Exploring a Unique Chemical’s Character and Future
A Glimpse into Its Development
The story of 1,1'-Azobis(Cyclohexanecarbonitrile), often called ACCN or ACHN in the chemical industry, traces back to the early decades when chemists searched for reliable radical initiators to spark free-radical polymerizations. Having handled this compound in a lab packed with textbooks and glassware, I remember reading about the shift from simple azo compounds like AIBN toward molecules with cycloaliphatic modifications. The development of ACCN addressed the growing need for initiators that offered higher thermal stability and generated a strong push in synthetic and industrial chemistry—sometimes outpacing benchmarks established by closely related substances.
What Sets ACCN Apart
Over years in research, ACCN has proven itself as more than just another radical initiator. Colleagues across laboratories know it for its reliability when working at higher temperatures. The compound's structure, featuring two cyclohexane rings anchored to a central azo group, gives it both stability and punch during decomposition, releasing nitrogen gas and free radicals that launch reactions in motion. As a solid, it appears white to yellowish, presenting itself in a non-hygroscopic form that makes storage trouble-free, free from sticky surprises often faced with other solids. The mere fact that ACCN manages decomposition temperatures near 100°C tells a lot about its robustness—this temperature often lands favorably when comparing options in radical polymerization.
Physical and Chemical Highlights
Holding a sample of ACCN feels much like handling other crystalline organic chemicals, yet its higher melting point and relatively low solubility in water show off subtle differences. The compound keeps a low profile when exposed to gentle conditions, resisting breakdown until exposed to heat above 90°C. The emission of nitrogen gas, a key feature of azo initiators, makes the substance attractive for reactions needing an inert and predictable outcome instead of unwanted byproducts. The two cyclohexyl groups push its molar mass well above that of simpler analogs, giving chemists a clear choice when a higher boiling solvent or reaction setup stands planned.
Technical Details in Higher Education and Industry
Back in my academic days, technical specifications of substances like ACCN rarely earned more than a passing glance—until unexpected results or mislabeled bottles crept into experiments. ACCN rolls off lab supply stocks with purity levels often above 98%, free from the confusion of mixed isomers or closely-related impurities. The substance often arrives labeled with its Chemical Abstracts Service Number, a small mark that means a lot when tracking chemical history in research logs. Its decomposition rate, measured in the handfuls of hours at 100°C, becomes a pivotal feature for researchers optimizing yields in polymer labs. Complicated as regulatory standards might seem, ACCN won’t escape scrutiny from workplace labeling systems, and any misstep in storage could translate to regulatory headaches—a lesson hard-learned by some researchers in the field.
The Craft of Synthesis
At its core, preparing ACCN involves coupling cyclohexanecarbonitrile with a diazotization process that introduces the azo bridge—a time-tested approach, familiar to trainers of every generation in organic chemistry. Reactions rely on temperature and pH control, as even minor deviations risk incomplete reaction and troublesome impurities. Organic chemists who allocate hours to purifying ACCN know the sting of missed yields or clumped byproducts; they become attuned to the importance of reaction cleanliness and the role of careful post-reaction workups. There’s a simple pleasure in seeing pure crystals form at the end of a synthesis: it means all those decisions, from reagent grade to timing, paid off.
Chemical Reactions and Modifications
Versatility underpins the worth of ACCN. Its well-behaved decomposition, giving off radicals and nitrogen, launches chain reactions that form polymers from a menu of monomers: styrene, acrylates, and even more specialized functionalized variants. The cyclohexyl side groups set the decomposition kinetics apart from other azo initiators, providing steady control in industrial reactors. Seasoned chemists even push ACCN to new roles with modifications—sometimes tacking on functional groups or using it as a building block for more specialized radical generators. In my stint in a materials science lab, there always seemed to be debate about which azo compound to choose, with ACCN often rising to the top based on outcome predictability.
Name Confusion and Synonyms
If you scan literature, expect synonyms: azobiscyclohexanenitrile, Vazo 88, and others reflect trade names and catalog labels. This confusion isn’t just a linguistic quirk—it can muddle up procurement, data collation, and even regulatory tracking. I’ve seen students order the wrong substance because they missed a catalog synonym, losing days (and sometimes weeks) to delays and corrections. Avoiding such mix-ups helps more than just compliance; it lets professionals focus on their craft instead of cleaning up behind miscommunications.
Safety and Operational Practice
No responsible discussion about ACCN dodges the safety aspect. As with any radical initiator, thermal decomposition can pose hazards—especially if handled in bulk or subjected to heating without adequate ventilation. Most facilities storing ACCN lock it away under cool, dry, and dark conditions, far from reactive materials. Unlike popular perception, these aren’t bureaucratic rules—they’re written in the lessons of past mishaps. Gloves, glasses, fume hoods: such routines separate a routine synthesis from an incident that generates paperwork and anxiety. Regulatory agencies look closely at ACCN’s hazard and precautionary statements, and so do workplace safety trainers. Factoring in thermal and toxic risks doesn’t slow work; if anything, it accelerates discovery by minimizing disruptions from preventable incidents.
Application Landscapes
Every time a new polymer hits the market, chances are good that radical initiators such as ACCN played a supporting role. The world of plastics and rubbers owes a lot to such molecules—whether the final use is insulation, coatings, or specialty elastomers. Companies value ACCN because its higher decomposition temperature grants them a wider processing window, helping to form long, strong chains in resins and composites. Some research outfits explore niche applications: fine chemicals, pharmaceuticals, and even materials for electronics. It’s not lost on me how radical polymerization, powered by reliable initiators, builds modern infrastructure right under our noses.
Research Frontiers
Explorers in academia keep returning to ACCN not just because of tradition but because it delivers new angles in radical chemistry. Recent work harps on ways to further tune its decomposition or pair it with co-initiators that shape molecular weight distribution in end products. Material scientists dig into blends that incorporate ACCN to yield new mechanical or chemical properties, and theoretical chemists model its radical pathways for clues about faster or more efficient syntheses. Real innovation doesn’t always mean chasing the next trendy molecule; sometimes, it means squeezing new performance or insight from a classic.
Toxicity and Environmental Impact
Talk of progress rarely goes far without recognizing the trade-offs. ACCN, like other azo initiators, carries health risks if mishandled. Though its acute toxicity lands in the moderate range, ingestion, inhalation, or improper skin exposure brings hazards—not only for workers but for the environment if disposal protocols get skipped. Waste streams from reactions need close attention, as do residues that might find their way into broader ecological systems. Labs and factories now set up rigorous monitoring for nitrile residues and decomposition byproducts out of both regulatory duty and a sense of stewardship. Serious toxicological research evaluates not just accident risks but also long-term effects, and staying mindful of this helps shape responsible science and industry.
What Lies Ahead for ACCN?
Looking toward the future, ACCN’s role might shift as green chemistry principles spread across industry. Cleaner, safer alternatives always attract attention, but established chemicals like ACCN hold their ground thanks to proven performance, cost stability, and deep user experience. Researchers now look for tweaks—catalytic cycles that recycle breakdown products, or modifications that enable milder operational thresholds. The push for sustainable materials nudges every part of the value chain, asking for initiators that work in aqueous systems or combine lower toxicity with high efficiency. Demand for high-performance polymers continues to climb, especially in sectors like renewable energy, automotive, and medical devices, all asking hard questions about performance and environmental legacy. Treading that balance between innovation and responsibility stands out as the real frontier, and ACCN has proven it can adapt across decades, suggesting it will keep playing its part wherever radical chemistry matters.
What is 1,1'-Azobis(Cyclohexanecarbonitrile) used for?
Why Would Anyone Want 1,1'-Azobis(Cyclohexanecarbonitrile)?
1,1'-Azobis(Cyclohexanecarbonitrile), better known among chemists as ACHN, plays a tougher role in industrial chemistry than people usually notice. At the surface, this white, crystalline powder just looks like any other chemical sitting on a shelf. Dig a bit deeper and it’s the reason some of the plastics and rubbers in daily life show up in the first place.
Polymerization sounds like a word for textbooks, but in practice, it’s pretty simple—the goal is to take small molecules and link them up into chains so they don’t flow everywhere and can actually form real stuff. That’s where ACHN steps up. It breaks apart at the right temperature and creates what’s called free radicals. These are like jump-starters that get those chemical chains building in the first place.
Manufacturers like ACHN because it gives them more control. For example, companies use it in making certain acrylic resins and plastics found in items like paints, adhesives, and even automotive parts. I remember visiting a facility where they made high-performance adhesives. You'd walk in and see powders being combined, temperatures rising, and then magic—the powdered ingredients came together into gummy sheets or plastic pellets. At every stage, a chemical like ACHN set the pace, dictating how fast the polymer formed, and in turn, how the product performed down the line.
Why Safety Beats Speed in These Reactions
There’s a flipside worth thinking about. ACHN isn't exactly a household name, probably because it deserves respect in handling. Thermal decomposition can release gases such as nitrogen, which makes certain polymerization reactions more efficient, but this also means there’s a risk if the process gets too hot. Factories keep a close eye on temperatures and monitor storage conditions for a reason. One incident with runaway reaction and you could end up with a mess that’s both hazardous and expensive.
In the push for safer workplaces, regulatory agencies like OSHA have stepped in, offering guidelines and expecting companies to invest in staff training and protective gear. Experience teaches you quickly that in a job like this, shortcuts invite more trouble than they’re worth. Using sealed reaction vessels, automated monitoring, and pressure relief systems all add layers of protection, helping workers trust that they won’t go home with more than a paycheck.
Learning From the Industry and Looking Ahead
Sustainability always enters the conversation. Waste from unused ACHN or byproducts can become an issue, so now many companies work on capturing emissions or recycling remains. Some research teams test out alternative free-radical starters derived from plants, aiming for the same results without the drawbacks. Progress here comes slow but steady, with teamwork between engineers and chemists. Finding safer chemicals that still let products measure up to the same standards takes time and repeated trials.
The world of specialty chemicals always has an eye on efficiency, cost, and safety. For those of us with a background in industrial processes, the lesson comes down to respect—for both the tools you use and the ways people are affected by them. 1,1'-Azobis(Cyclohexanecarbonitrile) doesn’t show up in glossy ads or TV shows, but it quietly helps build the world you use every day.
What are the storage conditions for 1,1'-Azobis(Cyclohexanecarbonitrile)?
What It Is and Why People Pay Attention
Anyone who’s walked into a chemical storeroom knows the air shifts a little when you’re handling strange, energetic molecules. 1,1'-Azobis(Cyclohexanecarbonitrile) lands on that list every time. It’s a solid, acts as a free radical initiator, and gets dropped into everything from plastics to rubber manufacturing. The story with this chemical always comes down to how temperature influences safety and shelf-life.
Temperature Makes or Breaks It
I’ve watched what some folks call “good enough” storage turn into an expensive lesson. This compound loves to break down if you leave it where temperatures climb above 30°C. It doesn’t just lose punch – decomposition can lead to gas build-up and, given the right conditions, even a fire risk. Keeping storage cool, below 20°C, makes sense not just for science, but for insurance and peace of mind.
If you lean on air conditioning, back it with a digital temperature logger. Don’t trust a window unit to stay steady in July. I’ve seen charts from labs that thought they could guess – peaks at 28°C for days in summer, material ruined and safety team called in. Stability data shows that even mild, repeated heat waves can trigger slow decomposition, even before the techs smell a hint of trouble.
Don’t Get Complacent on Containment
This is a solid powder that wants to stay dry. Moisture sneaks in and speeds up all kinds of unwanted chemical changes. My advice: Always use tightly sealed, chemical-resistant containers. I've worked in places that favored glass bottles, but high-density polyethylene offers tough protection without the breakage worries. You want to stack containers away from heat sources, direct sunlight, and don’t put them beside acids or oxidizers. Mixing up storage zones just creates risks if a spill actually happens.
Labeling, Inventory, and Accountability Save More Than Paperwork
Mislabeling gets people hurt. Label every bottle clearly—date of receipt and, where possible, the date transferred to its present jar. Do a shelf check at least once a month. I’ve walked in on suppliers who simply stack the drums and walk away, only to realize old supplies landed at the front, getting picked much too late. FIFO (first in, first out) should be the baseline, not a best practice.
I’ve seen seasoned techs rely on their “memory” for chemical location, only for someone new to pull the wrong sample. Digital inventories and real logbooks matter. Every spilled scoop, every misplaced container, every “I thought that was the empty one”—it all piles up until someone has to call the fire department instead of just chemistry support.
Training and Routines Cut the Risk
Most fires don’t start from a lack of knowledge—they start because folks skip routines. Embed storage checks into weekly tasks. Don’t let new staff wing it with secondary containment or labeling. Teach everyone what a “hot” storeroom smells like and how to spot pressure build-up. My old mentor always said, "The riskiest storage isn’t the fancy new stuff, it’s the forgotten old stock."
1,1'-Azobis(Cyclohexanecarbonitrile) isn’t an everyday chemical for most people, but get its storage right and you avoid drama, save money, and stay clear of news headlines about accidental fires. A little mindfulness paired with solid systems keeps people and property safe. That’s knowledge worth using any day.
Is 1,1'-Azobis(Cyclohexanecarbonitrile) hazardous or toxic?
What Is 1,1'-Azobis(Cyclohexanecarbonitrile)?
Anyone who’s worked in plastics or polymers might recognize the long-winded name 1,1'-Azobis(Cyclohexanecarbonitrile), sometimes called ACHN. Factories use this substance to help plastics foam up. Chemists know it as an azo compound – that part, the “azobis” in the name, signals it can release nitrogen gas and split apart, a trick that’s handy for industry but tricky for health and safety.
Examining the Risks and Toxicity
My time in labs has left me with a healthy respect for chemicals that decompose with a push. Substances that release a lot of gas or heat can cause a real mess—or worse, a serious accident—if handled carelessly. ACHN draws extra attention because as it breaks down, it does more than just foam up plastics. It releases toxic fumes, including hydrogen cyanide. Some labs take this for granted, but I’ve watched more than one close call play out around compounds that sneak cyanide into the air.
Sifting through research, animal studies flag danger with ACHN. Inhalation, swallowing, or even skin contact can harm health. Effects in test animals center on breathing trouble and organ damage. While these studies use doses bigger than most people ever see, exposure limits set by occupational safety agencies exist for good reason. And the risks don’t vanish just because a factory floor seems clean. Powder can linger on surfaces. Dust can sneak inside gloves or under cuffs.
Why This Matters in the Real World
I’ve met plenty of workers who trust their PPE but underestimate the risks of fine powders. Chemicals like ACHN ask for vigilance, not just lab coats. Overconfidence, or letting safety culture slide, only raises the chances of exposure. For people who live near manufacturing plants, the stakes go beyond the factory gates. Spills make headlines, and even rare leaks leave locals worrying about soil and air safety.
Regulation helps manage some of these dangers. The EPA, OSHA, and similar agencies worldwide put guidelines in place, but rules alone seem toothless without a culture of respect for the chemicals themselves. A proper risk assessment doesn’t just tick boxes; it means asking if handling ACHN remains worth the trade-off, especially as greener alternatives come along.
Safer Practices and Practical Steps
Education shapes the backbone of chemical safety. Workers who see demonstrations about what even a small cyanide exposure does to the body make different choices. Frequent monitoring—air, surfaces, and blood if needed—keeps risks from creeping up without warning. Emergency response drills sometimes get pushed aside for productivity, but my experience says the best teams know what to do long before an alarm ever sounds.
Simple design changes protect people more than the thickest gloves. Closed systems trap powder, keeping it from floating into breathing zones. Good ventilation pulls fumes out and away, instead of pushing them around the room. Waste needs secure handling, too, with disposal teams treating spent material as harmful right until proper neutralization.
Greener Alternatives: A Path Forward
Some in the industry look for other ways to get their plastics to foam. Newer compounds trade a little convenience for a bigger margin of safety. Switching often means higher upfront costs, but for the communities and workers, the health payoff seems obvious. With innovation, companies shoulder less liability and neighbors downwind breathe easier.
What is the molecular formula and structure of 1,1'-Azobis(Cyclohexanecarbonitrile)?
Structure that Shapes Function
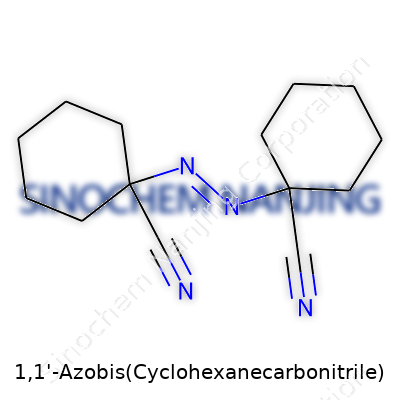
Most chemicals stay out of the spotlight, but 1,1'-Azobis(cyclohexanecarbonitrile) (ACHN) deserves real attention. The basic formula, C14H20N4, doesn’t look like much on paper, but a quick look at its architecture shows a kind of symmetry that reminds you nature and science both love balance. Each molecule builds from two cyclohexane rings, joined by a bridge built from nitrogen at the center, with nitrile (–CN) groups hanging off the rings’ edges. It isn’t a household name, but its structure hints at a purposeful model, tailored for specific uses in the real world.
The Backbone: Breaking Down the Molecular Skeleton
Take any basic chemistry course and you’ll spot patterns like this molecule. Two cyclohexane rings, sturdy six-membered carbon rings, offer stability on both sides. Link those with an azo (–N=N–) bridge in the center: this bridge sets up a weak link ready to snap at the right temperature, releasing nitrogen (N2). Each cyclohexane wears a nitrile group on its first carbon. In shorthand, the molecule looks like this: NC–C6H10–N=N–C6H10–CN.
This isn’t the kind of molecule you’ll find in your kitchen. Chemists look for molecules that break down in a predictable way, sending off steady streams of free radicals. That’s exactly what ACHN delivers. This quality makes it ideal for specific polymerization processes, where a compound’s ability to start a reaction and then step quietly aside defines its usefulness.
Why the Details Matter
If you’ve spent any time mixing polymers or trying to control how they set, the difference between a good initiator and a poor one becomes clear. ACHN cooks off at higher temperatures, around 90–95°C for its half-life, which makes it soar past similar initiators like AIBN (azobisisobutyronitrile) with lower decomposition rates. That difference in thermal stability means it can be used in reactions that demand longer cook times or need stability in heat—like manufacturing specialty plastics. European and U.S. chemical suppliers tout this feature, selling the compound for its reliability in tough jobs.
Research out of Japan and Germany likes ACHN because it makes reactions easier to control without releasing too many byproducts. Nitrile groups add a layer of safety: they hang on and don’t give off smoke or messy side products when the molecule splits, which keeps factory air a little bit cleaner and quality control tighter.
Potential Hazards and Safer Handling
Working with chemicals that generate free radicals always brings risk. I still remember handling similar azobis compounds in grad school, hoping I’d done enough math to keep the temperature under control. Thermal runaway can cause a fireball if a reactor climbs past the safe point. Process engineers watch numbers closely, automate their controls, and use explosion-proof equipment. Staying prepared matters more than cutting corners, especially once you scale up a process.
Newer labs explore safer replacements, but for now, ACHN still fits into many niche roles. Full transparency, detailed handling guides, and regulatory checklists need to be followed. Data from the European Chemicals Agency ranks it as hazardous but manageable if operators stay attentive and avoid dust clouds or loose spills.
Room for Improvement
A push for greener chemistry keeps growing. Academic teams explore if the nitrile side groups or the azo bridge could be “tuned” for less risk, perhaps swapping in renewable cyclohexane rings or finding decomposition products even cleaner than nitrogen. For chemistry teachers and students, studying ACHN offers a chance to see how molecular structure connects to practical solutions in industry. That very relationship, between the nuts and bolts of structure and the real world, lies at the core of every smart process.
What are the handling and safety precautions for 1,1'-Azobis(Cyclohexanecarbonitrile)?
Understanding the Chemical: Why Respect Is Essential
The minute you start working with 1,1'-Azobis(Cyclohexanecarbonitrile), the stakes rise. This compound comes with a storied reputation among polymer manufacturers and lab workers, mostly due to its high energy and instability under heat or shock. At room temperature, it looks tame—white crystalline powder, nothing scary. But stories circulate in industry circles about energetic decompositions, splattering bench tops, and causing real harm. Having spent years around such chemicals, you learn not to underestimate any azo initiator.
Basic Precautions: Setting Up a Safe Environment
Every safe handling protocol starts with a respect for detail: lab coat, chemical splash goggles, proper gloves—nitrile ones work best. Good ventilation isn’t just a convenience, it stops vapors from lingering. Lab managers always stress use of a fume hood. Procedures must include fire extinguishers within arm’s reach and clear, unobstructed exits. One overlooked spill on a hotplate or contact with a spark, and you remember for good.
Storage: Fight Heat, Ignore Carelessness
If you store this chemical near any heat source, you court disaster. Dry, cool cabinets with locked doors and real, physical separation from oxidizers and acids keep people safe, not just materials. Use only containers meant for incompatible chemicals—they should carry tight lids, easy-to-read labels. Lab workers often forget not every storage cabinet is safe; I once had to clean up after a rookie mistake, finding initiators stored above a radiator. Luck kept it from becoming front-page news.
Safe Work Habits: Preventing Accidents Starts Here
Don’t let powder accumulate on scales or surfaces, and don’t rush measuring or transferring. Spilled particles can ignite with friction. I remember how a bit of powder left on the neck of a bottle ended up sparking a near miss for a colleague. He didn’t lose his hand, but he gained a permanent respect for slow, deliberate motions around dangerous materials.
Never handle more than you actually need. Batch size equals risk. I’ve seen enough to know the old saying: “A gram in the trash, a kilogram in the morgue.” Take only what the procedure calls for—never round up.
Emergency Response: Readiness Beats Regret
Accidents happen, even in the best-run labs. Emergency showers, eye wash stations, and spill kits should feel as familiar as a favorite tool. Team drills matter—nobody wants to read the instructions for the first time during an emergency. I’ve participated in simulated spills and those minutes spent practicing made the difference when a real one came along.
Training and Community Learning
Nobody can memorize every rule, but routine safety training keeps knowledge fresh. The best labs encourage a culture of openness, where talking about uncertainties or close calls isn’t a weakness. Sharing near misses trains the group much harder than a dry rules review. Experience taught me: speaking up about a misjudgment or a shortcut means you help everyone—not just yourself—avoid injury or worse.
Knowledge grows when people tell the truth about what can go wrong. Handling high-energy chemicals like 1,1'-Azobis(Cyclohexanecarbonitrile) demands that honesty and preparedness, every single day.
| Names | |
| Preferred IUPAC name | 2-(1-Cyano-1-cyclohexylethylidene)cyclohexanecarbonitrile |
| Other names |
Vazo 88 ABCN 1,1’-Azobis(cyclohexanecarbonitrile) Azobis(1-cyclohexanenitrile) AZCN |
| Pronunciation | /ˈwʌn.wʌn ˈeɪ.zəʊ.bɪs ˌsaɪ.kloʊˈhɛk.sən.kɑːrˈboʊ.nəˌtraɪl/ |
| Identifiers | |
| CAS Number | 2094-98-6 |
| Beilstein Reference | 1466122 |
| ChEBI | CHEBI:51845 |
| ChEMBL | CHEMBL373242 |
| ChemSpider | 17045 |
| DrugBank | DB02592 |
| ECHA InfoCard | ECHA InfoCard: 100.012.348 |
| EC Number | 208-912-9 |
| Gmelin Reference | 303496 |
| KEGG | C18257 |
| MeSH | D000368 |
| PubChem CID | 13182 |
| RTECS number | GR6300000 |
| UNII | BH8M9B6M67 |
| UN number | 3236 |
| Properties | |
| Chemical formula | C14H20N4 |
| Molar mass | 276.39 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.06 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.62 |
| Vapor pressure | 0.06 mmHg (at 25 °C) |
| Acidity (pKa) | no data |
| Basicity (pKb) | 12.85 |
| Magnetic susceptibility (χ) | -61.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.520 |
| Viscosity | Viscosity: 10 mPa·s (20 °C) |
| Dipole moment | 4.58 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 371.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -3261 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H242, H302, H317, H410 |
| Precautionary statements | P210, P220, P221, P234, P235, P240, P241, P261, P264, P270, P271, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P306+P360, P311, P330, P333+P313, P337+P313, P342+P311, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-3 |
| Flash point | 79°C |
| Autoignition temperature | 270 °C |
| Lethal dose or concentration | LD50 oral rat 740 mg/kg |
| LD50 (median dose) | LD50 (median dose): 4000 mg/kg (oral, rat) |
| NIOSH | NA9106 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m3 |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Azo compounds Azobisisobutyronitrile Azobisisobutyramidine Hydrazine |

