1,1,3,3-Tetramethyl-1-Butanethiol: A Real-World Look at an Influential Chemical
Historical Development
Back in the early days of organosulfur research, most chemists kept an eye out for compounds with unique atomic scaffolding. 1,1,3,3-Tetramethyl-1-butanethiol showed up as an oddball with a heavily branched structure, catching the attention of process chemists who saw potential beyond simple laboratory uses. The compound’s pronounced thiol odor underscored its role as an unmistakable marker in early gas leak detection systems, shaping the way industries approached both safety and olfactory marking. Over time, improvements in large-scale organosulfur synthesis made this compound more affordable and accessible, pulling it out of the exclusive hands of bench chemists and putting it in toolkits across industrial and environmental sectors.
Product Overview
1,1,3,3-Tetramethyl-1-butanethiol stands out as a colorless to pale yellow liquid with an intense, characteristic thiol scent. Out in the field, anyone who’s worked with mercaptans will recognize its pungency from miles away. It’s a building block for making sulfur-containing chemicals, known for its ability to impart both chemical and olfactory properties wherever added. Producers market it to those who want a persistent, high-impact thiol, either as a reactant or as a proprietary flavor and odorant agent in specialty blends—useful in everything from polymers to industrial leak detection.
Physical & Chemical Properties
This molecule doesn’t hide its personality. With a boiling point hovering around 146–148 °C and a melting point well below room temperature, 1,1,3,3-Tetramethyl-1-butanethiol keeps itself liquid through most lab or plant processes. Its density runs near 0.84 g/cm³, and it brings low solubility in water but mixes well with common organic solvents. As with many thiols, exposure to air or light can lead to slow oxidation, sometimes tarting up storage vessels with traces of sulfoxides or disulfides. The compound’s sulfur-hydrogen bond enables a strong nucleophilic character, a fact that synthetic chemists take advantage of in a range of reactions.
Technical Specifications & Labeling
Every bottle of 1,1,3,3-Tetramethyl-1-butanethiol deserves an honest and practical label. The CAS number, 594-44-5, identifies it for inventory, but more importantly, clear hazard warnings about its flammability and extreme odor need to be front and center. Most suppliers package it in amber bottles or steel drums with vapor-tight seals, and the minimum spec for purity generally sits at 98%, with impurities spelled out for the sake of both safety and downstream chemistry. The Safety Data Sheet has to break down the risks—not just to satisfy paperwork but to ensure everyone involved knows what to expect.
Preparation Method
Manufacturing starts with heavily branched alkanes—typically neopentane derivatives—under stringent, anhydrous conditions. A favorite technique involves chlorination of precursors like 2,2,4,4-tetramethylpentane followed by nucleophilic substitution with hydrogen sulfide or a suitable thiol source. Those with boots-on-the-ground lab experience keep a close watch for runaway reactions, as exotherms and toxic byproducts can spike if moisture sneaks in or temperatures drift. Scaling up isn’t just about bigger vessels; reactor fumes, thiol loss to air, and the mess left by sulfur residues all challenge clean, repeatable production.
Chemical Reactions & Modifications
The strong nucleophilicity of 1,1,3,3-Tetramethyl-1-butanethiol makes it a versatile player in organic synthesis. It cleaves alkyl halides for thioether formation, works in Michael additions, and latches onto electrophilic centers with ease. Oxidation turns it into sulfoxides and sulfones, both of which have their own niches in pharmaceuticals and functional materials. In practical field work, this chemical helps modify polymer matrices, adding flexibility or sulfur-based reactivity to finished products. Thiol-ene "click" chemistry and metal chelation create new ways for engineers to improve selectivity in separation and analysis columns, helping researchers chase purity and yield without breaking the bank on precious reagents.
Synonyms & Product Names
Outside the lab, names morph and proliferate—1,1,3,3-Tetramethyl-1-butanethiol answers to a few. Some catalogs list it as tert-Tetramethylbutanethiol or TMBT. Old school organics texts sometimes refer to it as 2,2,4,4-tetramethyl-3-thiabutane. Industrial circles shorten it to “TM-butanethiol,” depending on the context, but savvy chemists out in the plant know they’re getting the same no-nonsense mercaptan compound with the telltale stench.
Safety & Operational Standards
Working with strong-smelling thiols teaches you lessons fast. Exposure, even with ventilation, triggers headaches and disrupted workflow. Standard process safety rules call for personal protective equipment, robust fume hoods, and careful planning for spills and leaks. Inhalation or skin contact leaves a mark—thiols get absorbed easily, so gloves and goggles aren’t just a formality; they’re a day-to-day requirement. Static discharge near open containers spells disaster given the compound’s flash point. Every plant or academic lab handling significant quantities has an environmental management protocol for air filtration, waste collection, and emergency neutralization containers on hand.
Application Area
Industries leverage this compound for its strong odor, relying on it as a chemical marker in pipeline and tank leak detection. The reliability of its scent lets technicians identify escapes of otherwise odorless gases without expensive electronic sniffers. In the world of specialty polymers and engineering plastics, the reactive thiol brings flexibility or sulfur cross-linking to finished polymers, improving impact resistance or elasticity where traditional cross-linkers won’t cut it. Some flavor and fragrance houses include tightly regulated microquantities in complex scent applications, harnessing its aroma impact as a blending note for sulfurous or “truffle” profiles. Academic research also sees use as a reagent in advanced synthesis, surface chemistry, and materials modification.
Research & Development
Monitoring trends in R&D labs, 1,1,3,3-Tetramethyl-1-butanethiol serves as a benchmark nucleophile for testing new synthetic routes in sulfur chemistry. Its pronounced smells complicate double-blind studies, yet help with rapid identification of successful reactions during troubleshooting. Research teams dig into structure-property relationships, using NMR and mass spectrometry to track modifications or impurities. This compound also features in explorations of green chemistry, where teams seek milder, less odorous alternatives for large-scale synthesis. Its reactivity and structure make it ideal for computational modeling, teaching new chemists how sterics affect chemical reactivity at both undergraduate and professional levels.
Toxicity Research
Thiol toxicity isn’t a question for debate among people who’ve handled 1,1,3,3-Tetramethyl-1-butanethiol outside of textbooks. Contact with skin causes irritation almost immediately, and short-term inhalation leaves technicians with headaches and temporary nervous system effects. In higher doses or with prolonged exposure, animal studies notes liver and kidney stress. Regulators place strict exposure control standards, typically setting allowable air concentrations far below those for less obnoxious volatile organics. While some studies continue to probe its breakdown products or interactions with biological systems, general consensus supports minimizing unnecessary exposure—good ventilation, training, and quick action for spills remain the best insurance.
Future Prospects
Looking ahead, new frontiers in materials science may call for molecules like 1,1,3,3-Tetramethyl-1-butanethiol as flexible intermediates or specialty modifiers. Sustainable synthesis, tighter emission rules, and public demand for odor reduction drive both process improvements and the hunt for alternatives with similar chemical features but less environmental footprint. Emerging tech in sensor calibration, advanced coatings, and high-performance polymer blends keeps this compound on the radar for those pushing chemistry into new spaces. Open questions linger on how to refine production for less waste, ramp up recovery and recycling efforts, and shift use cases toward safer, greener applications as markets and regulations evolve.
What is 1,1,3,3-Tetramethyl-1-Butanethiol used for?
The Job of a Chemical That Smells Awful
Some chemicals turn noses up with just one whiff. 1,1,3,3-Tetramethyl-1-butanethiol ranks right up there as a heavyweight in the odor department. This pungent compound makes its presence known long before anyone takes a reading. Yet, that cutting odor actually shields lives and property. In real-world jobs, nasty smells help keep people safe from hazards you can’t see. This chemical stands as a perfect example.
Why Industry Needs Chemicals Like This
I’ve worked around tanks, valves, and miles of pipe. In those spaces, pipelines carry gases that, left to themselves, don’t warn workers when a leak happens. One property of 1,1,3,3-Tetramethyl-1-butanethiol stands out: it releases a strong smell at very low concentrations. Gas companies take advantage of this by adding it to liquefied petroleum gas. The “rotten cabbage” note signals a leak before invisible dangers can build up. Nobody around a fueling station forgets that scent after their first safety drill.
The Science That Drives Safety
Natural gas, propane, and butane have no smell. That’s a recipe for silent trouble in homes, factories, and workshops. Regulations force suppliers to add odorants. 1,1,3,3-Tetramethyl-1-butanethiol, with its low detection threshold, checks this box. Many North American plants use this molecule so the public can detect leaks at concentrations far below dangerous levels. That quick warning window stops explosions, injuries, and plenty of expensive clean-up.
A Bit Beyond Odorizing Gas
This compound doesn’t only live in the gas industry. Chemical manufacturers sometimes reach for it to build specific molecules. Thiols, in general, open doors for organic synthesis, especially when companies chase new drugs or specialty chemicals. The bulk of 1,1,3,3-Tetramethyl-1-butanethiol heads into odorizing, but a slice lands in research labs or production lines. Each molecule acts as a building block for substances that fuel improvements in medicine and agriculture.
Facing Down Hazards and Looking for Better Solutions
Handling thiols takes some grit. Anyone working in the plant smells it for days, and a careless spill can clear a room. Chronic exposure brings headaches and nausea. Most sites train employees with extra care and install ventilation systems that keep air moving. Tanks always need double-checking for leaks. If spills get out of hand, clean-up teams tackle the mess in layers, using absorbent materials and protective gear. Regular audits cut down on surprises, but there’s always work to do on the safety front.
People in the industry are pushing research into alternative odorants, often hoping for lower toxicity or a less persistent stench. New molecules get tested, but few can beat the detection strength this thiol offers. Until someone builds something better, this workhorse holds its ground, helping utilities and manufacturers protect both workers and the public from harm.
What are the safety precautions when handling 1,1,3,3-Tetramethyl-1-Butanethiol?
Understanding the Risks Up Close
I’ve spent years working with all sorts of chemicals, and strong-smelling, volatile thiols like 1,1,3,3-tetramethyl-1-butanethiol never fail to grab my full attention. Just a whiff lingers for ages and can cause quite a headache. The law treats this chemical seriously because of its low odor threshold, tendency to irritate airways and skin, and flammability. I still remember the sticky feeling on bare skin and that persistent onion stench that clings to lab coats and gloves for days.
Personal Protective Gear: Your Real Shield
In the lab, nothing replaces the right protective gear. Thick nitrile gloves shield skin from nasty sensations and rashes. Loose sleeves or cotton gloves allow vapors to slip through, so they belong in the drawer, not on your hands. A snug-fitting lab coat and chemical splash goggles block splashes and volatile vapors. Some folks shrug off wearing eye protection—a mistake quickly regretted if even a drop finds its way near the eyelid.
Good Ventilation and Containment
This chemical quickly fills the air with its odor, even at tiny concentrations, so good ventilation counts as essential equipment. I always work under a fume hood and keep containers tightly sealed except when pouring or measuring. Fume hoods with proper airflow remove vapors from the workspace, cutting the risk of headaches and keeping coworkers happy. Air filters help only after opening lids becomes necessary. An open window will not clear up the problem; proper containment stops issues at the source.
Smart Storage Practices
Once, I left a flask out overnight instead of returning it to its sealed bottle. The next shift walked into a cloud of thiol vapor, and the entire wing stank for days. I learned quickly to check for tight seals and stash the bottle inside a vented, flammable-safe chemical cabinet, away from heat, sparks, or sunlight. This chemical reacts with oxidizers and acids, so keeping them apart really matters. Label containers clearly—one tiny mistake causes headaches for everyone.
Cleanups and Spills: Plans That Work
Spills happen, even to those who move carefully. I always keep spill-neutralizing agents and lots of absorbent pads handy. If liquid leaks out, I cover it right away with clay-based absorbent and work fast, since vapors spread before you realize it. I never use rags or paper towels—these just spread the odor and raise the risk of fire. After the cleanup, I toss every contaminated glove and wipe in a sealed hazardous waste bin, never regular trash. No one wants mystery odors in the dumpster behind the facility.
Health Precautions: Listen to Your Body
Working around volatile thiols, coworkers sometimes ignore headaches, sneezing, or watery eyes, chalking it up to allergies. In my experience, those symptoms are a warning system. Leave the area, get fresh air, and tell the safety officer if you notice trouble. See a doctor if symptoms don’t fade. Safety training works only if people use it and report issues early. No project deadline excuses risking personal health.
Prioritizing Human Wellbeing over Shortcuts
Protecting yourself and others always trumps saving a few minutes. Consistent habits—suited-up hands, closed containers, fresh air, staying alert—become second nature. It’s not about following rules blindly, but about respecting how dangerous these chemicals prove to be when taken lightly. Overconfidence never ended well in my experience, but well-mapped-out steps kept everyone safe and the workspace odor-free at the end of the day.
What is the chemical formula of 1,1,3,3-Tetramethyl-1-Butanethiol?
Looking Beyond the Name
1,1,3,3-Tetramethyl-1-butanethiol isn’t a name that rolls off the tongue. It sounds like a jumble of numbers and letters, but understanding what those mean opens up a fascinating window into organic chemistry. At its simplest, the formula is C8H18S. Picture eight carbon atoms, eighteen hydrogen atoms, and a single sulfur atom, tightly bound in a very specific structure.
Why the Structure Matters
The structure really matters here. This molecule starts with a four-carbon backbone, known as butane. The ‘1,1,3,3-tetramethyl’ part means that two methyl groups attach to the first carbon, and two more go to the third carbon. Add a thiol group (-SH) to the first carbon, and the shape gets even more defined. This shape isn’t just a matter of trivia. Compact, “branched” molecules like this often behave differently than long, straight ones. The bulk makes them less likely to stick together, and that plays into solubility and boiling point.
Real-World Uses and Concerns
Chemists have a reason for caring about molecules like 1,1,3,3-Tetramethyl-1-butanethiol. Thiols carry a sharp, often unpleasant odor—think rotten eggs—but they serve an important purpose as odorants, tracer chemicals, and intermediates for making other compounds. Fieldwork and personal experience in organic labs show that just a drop of a thiol can change the air in a room. This reactivity isn’t only about smell; sulfur compounds trigger strong responses in living organisms, making them useful for leak detection in fuel gases and for calibrating instrumentation.
Exposure, though, can cause problems. Even trace amounts can set off headaches, respiratory irritation, or allergic reactions. Handling thiols takes good ventilation, reliable gloves, and proper disposal knowledge. Too often, folks ignore safety advice, treating small-scale work as harmless. Lab accidents, once mild and now serious, usually come from ignoring these routines. I’ve seen safety drills dismissed as overkill until that one time someone accidentally spilled a strong thiol. The smell lingered for weeks despite our best cleanup.
Questions Around Environmental and Human Safety
Concerns stick around once a chemical leaves the lab. Accidental releases of smelly thiols trigger community complaints and health worries. Sulfur compounds resist quick breakdown in the environment, persisting in soil and water. Even local wildlife feels the effects, as fish and insects respond strongly to even tiny concentrations. Some countries set strict thresholds for thiol emissions, but enforcement slips in smaller industries or older facilities.
Disposal often falls through the cracks. Instead of following hazardous waste guidelines, some facilities dilute and dump thiols, leading to long-term groundwater and odor issues. Regulations exist, but wider education and regular site checks often do more to keep communities safe. Neighbors know the scent and can pressure for change faster than distant agencies.
Practical Steps Forward
Instituting better on-site chemical management and swift spill response builds trust and stops problems early. Teaching good chemistry habits—double-gloving, labeling, exhaust fans, quick neutralization—keeps both the lab and the wider world safer. Chemistry students benefit most by seeing the real-world impact, not just memorizing formulas.
In the end, understanding C8H18S isn’t just for scientists. Anyone working with or living near labs, plants, or refineries needs to know what’s floating in the air. Community knowledge and transparent safety practices can spare headaches—sometimes literally.
How should 1,1,3,3-Tetramethyl-1-Butanethiol be stored?
Straight Talk About Safety
1,1,3,3-Tetramethyl-1-butanethiol, often used in labs and some industry settings, carries a punch when it comes to both smell and hazard. People underestimate chemicals like this until something goes wrong. Forget the idea that any old storeroom will do. Taking shortcuts or skipping steps turns a tool into a risk. My work around industrial solvents and odorous organics proved that attention to real-life detail keeps people healthy and business running. A leak or careless exposure ruins more than a day—it can ruin lives or reputations.
Not Just Any Container Will Work
Strong fumes and volatility set this chemical apart from simple liquids on a shelf. Vapors escape fast if lids don’t fit snug. Use containers made from metal or high-quality glass with chemical-resistant seals. No cheap plastics here. Some plastics soften, crack, or allow slow leaks, which goes unnoticed until someone smells trouble. Using tight-sealing threaded caps gives better results than snap lids. Store in the original container if possible. Factory-packaged drums and bottles come pre-tested for compatibility.
Let’s Talk Temperature
High temperatures push vapor pressure through the roof. Warm rooms let fumes build up, corroding shelving and joints around the storage area. The best bet—keep the chemical cool, away from direct sun or heat sources. Stay under 25°C (77°F) if possible. Colder is usually better, but avoid freezing conditions that can damage containers or make handling tricky. Beating back temperature swings also stops condensation and keeps seals tight. The fire hazard from volatile organic chemicals remains real, so keep flammable items well separated.
Ventilation and Segregation
Stale, stagnant air traps any vapors escaping a leaky stopper. Ventilated storage rooms help a lot. Even a low-powered exhaust fan reduces toxic buildup. 1,1,3,3-Tetramethyl-1-butanethiol stings the nose and throat—even small leaks become a big deal. Store away from acids, oxidizers, and bases. Accidental mixing sets off noxious, sometimes dangerous, reactions. Over the years I’ve seen more than one facility ignore these simple rules and pay the price in ruined product and worried staff. Well-separated shelves and marked cabinets reduce mix-ups.
Labeling, Logs, and Inspections
Labels on these containers need to stay clear, bold, and complete. Handwriting fades and tape peels, especially in humid environments. Rely on printed chemical-proof labels for peace of mind. Keep a logbook showing when each bottle comes in and goes out. Nothing fancy – just date, amount, initials. Schedule inspections to spot leaks, rust, or loose lids before the nose picks up trouble. Spot-checks take minutes and prevent hours of cleanup or medical evaluation. Staff deserve clear instructions, training refreshers, and the right gear to limit mistakes.
Personal Experience and Solutions
Handling foul-smelling organosulfur compounds early in my career cemented my respect for simple, consistent practices. Safety eyewash stations, respirators, and gloves aren’t a luxury—they’re the cost of working smart. I learned to set spills or vapor clouds as zero-tolerance issues in company policy. A little money spent on proper storage beats long-term costs from accidents or downtime. Consider spill containment pallets and chemical-specific cabinets. Chemical management software alerts teams to expiration dates and volume levels before trouble starts. Organizations who take these cues help employees stay safe, protect the environment, and build trust that lasts longer than any single batch on the shelf.
What are the physical properties of 1,1,3,3-Tetramethyl-1-Butanethiol?
Getting to Know This Thiol
1,1,3,3-Tetramethyl-1-butanethiol probably won't show up in most conversations, yet it plays a bigger role in certain industries than folks realize. Looking at its structure, the bulky four methyl groups sitting on the ends give this molecule its oddball shape and some stubborn qualities. The “thiol” part means a sulfur-hydrogen (–SH) group hangs on, giving it that classic "skunky" smell familiar to those who’ve spent time in labs or near pulp mills.
Properties That Stand Out
Weighing in at 162.33 g/mol, this molecule doesn't float through the air quickly. Instead, the higher molecular weight keeps it hugging the surface a bit longer than lighter things like ethanol or methanethiol. Pour some out at room temperature, and you’d see a colorless, oily liquid that clings to containers. The melting point lands around -40°C, so this isn’t something you’ll see solidify except in a serious deep freezer. The boiling point hovers just above 150°C, so it stays a liquid under most conditions folks encounter in routine work.
The density hits just above 0.82 g/cm³, putting it in line with many organic liquids. That makes spill cleanup tricky — it spreads thin and runs quickly over surfaces. Water doesn't want to mix with this molecule; its hydrocarbon tail shrugs off H2O without a thought. In my time working in chemical formulation labs, anything oily and immiscible demanded extra care with storage and drainage, especially if thiols got involved. The smell lingers, and so does the residue if you aren't careful.
Real-World Impacts
Sulfur compounds like this one turn heads in safety meetings, thanks to their sharp odors and reputation for reacting with metals. Over time, exposure to the air can cause discoloration or even corrosion on some storage vessels. Glass and select plastics work better than metals, which always got flagged during hazard reviews at facilities I've visited.
Flammability sticks out on safety sheets. The flash point of this thiol runs down near 46°C, which means keeping it away from open flames or hot surfaces can't just be a tip — it's a rule in any facility. People sometimes underestimate just how easily vapors catch fire, especially because the heavy sulfur content can also lead to a choking, irritating smoke that throws a wrench in evacuations and cleanups.
Dealing with Challenges
Most places storing thiols have to build extra ventilation and odor controls. Activated carbon filters got the job done for us, trapping stray molecules before they left the fume hoods. On small scales, double-sealing containers helped avoid unwanted attention, and every transfer ran slow, with spill pads at the ready.
Labs that work with organosulfur compounds tend to keep detailed logs. The persistent stench means any mistake or forgotten bottle gets noticed fast, which works as an unintentional safety reminder. Disposal also takes some planning: local wastewater systems can’t handle high loads of thiols, so licensed haulers usually cart off old material for incineration.
Why Pay Attention?
Physical traits might sound dry at first glance, but folks in the field know that boiling points, densities, and flash points often decide how safely and efficiently we use, transport, or contain chemicals. The stubborn nature and sharp scent of 1,1,3,3-tetramethyl-1-butanethiol mean it won’t go unnoticed for long. Proper training, good ventilation, and handling protocols turn what could be a safety headache into just another quiet tool in the chemical toolbox. Ignoring these gritty details can turn ordinary workdays into cleanup operations nobody signs up for.
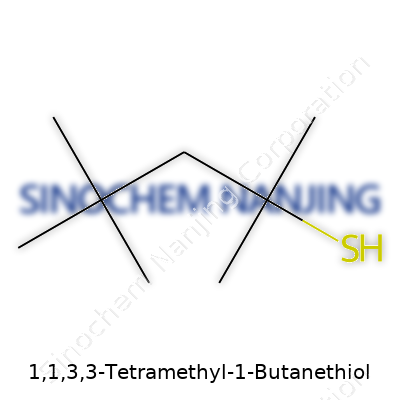
| Names | |
| Preferred IUPAC name | 2,2,4,4-Tetramethylbutane-1-thiol |
| Other names |
2,2,4,4-Tetramethyl-3-thiopentane Trimethylisobutylthiol |
| Pronunciation | /ˈwʌn ˌwʌn ˌθri ˌθri ˈtɛtrəˌmɛθəl ˈwʌn ˈbjuːteɪˌθaɪɒl/ |
| Identifiers | |
| CAS Number | 107-47-1 |
| Beilstein Reference | 1718732 |
| ChEBI | CHEBI:51417 |
| ChEMBL | CHEMBL3736752 |
| ChemSpider | 14240574 |
| DrugBank | DB11261 |
| ECHA InfoCard | 100.181.392 |
| EC Number | 204-698-5 |
| Gmelin Reference | 79021 |
| KEGG | C19206 |
| MeSH | D015685 |
| PubChem CID | 122345 |
| RTECS number | EO1575000 |
| UNII | WCQ21546UN |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID8020423 |
| Properties | |
| Chemical formula | C8H18S |
| Molar mass | 162.34 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | disagreeable |
| Density | 0.834 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 2.92 |
| Vapor pressure | 0.9 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 10.7 |
| Basicity (pKb) | pKb ≈ 3.7 |
| Magnetic susceptibility (χ) | -53.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.481 |
| Viscosity | 1.37 cP (20°C) |
| Dipole moment | 1.35 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -224.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7645 kJ/mol |
| Pharmacology | |
| ATC code | D10AB05 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H225, H301, H311, H331, H315, H319, H335, H400 |
| Precautionary statements | P261, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P403+P233 |
| NFPA 704 (fire diamond) | 3-4-0 |
| Flash point | > 68 °C (closed cup) |
| Autoignition temperature | 232 °C (450 °F; 505 K) |
| Explosive limits | Explosive limits: 1.1–6.9% |
| Lethal dose or concentration | LD50 Oral Rat 237 mg/kg |
| LD50 (median dose) | LD50 Oral rat 890 mg/kg |
| NIOSH | RN1045000 |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
1,1,3,3-Tetramethylbutane tert-Butylthiol 2-Methyl-2-propanethiol 1,1-Dimethyl-1-propanethiol |

