1,1,3,3-Tetrachloroacetone: A Deep Dive into Its Chemistry and Role in Research
Historical Development
Chlorinated ketones always catch the attention of organic chemists thanks to their reactive centers and, frankly, their smell lingers in the lab long after the reaction wraps up. 1,1,3,3-Tetrachloroacetone attracted real interest starting in the early 1900s, when chemists explored new paths for halogenating carbonyl compounds. Synthetic chemists quickly realized this molecule did more than just look interesting on a shelf; it could transform starting materials, add halogen atoms where they matter, and become a tool for building more complex chemicals. Through the decades, the demand for specialty chlorinated reagents kept laboratories from Boston to Basel searching for better preparation routes and studying its reactions inside and out.
Product Overview
In practical terms, 1,1,3,3-Tetrachloroacetone delivers value as a halogen-rich acetone derivative. This compound, typically a clear to slightly yellow liquid, supplies four chlorine atoms on a central carbon skeleton. These features make it more than just another reagent in the bottle rack. Professionals value it for both its reactivity in synthesis and selectivity in introducing specific halogen groups to molecules. Whether chemists need to create novel building blocks or tweak the properties of other compounds, tetrachloroacetone stands as a reliable option that consistently delivers what’s expected in a reaction.
Physical & Chemical Properties
Tetrachloroacetone boils at about 148-150°C, which matters for anyone considering purification or storage. Its density hovers around 1.6 g/cm³—significantly heavier than water, so it settles fast in separatory funnels and rarely gets mistaken for lighter solvents. This molecule doesn’t blend well with water; its polarity keeps it apart, so chemists often use nonpolar or slightly polar solvents to coax it along in reactions. As a chlorinated ketone, it's sensitive to bases, and its carbon-chlorine bonds invite nucleophilic attack, supporting a wide range of synthetic transformations. Anyone who’s spilled even a drop can attest to the intense, unmistakable odor that plagues a space for hours, so lab techs quickly learn to use it under a fume hood every time.
Technical Specifications & Labeling
Accuracy in handling any halogenated compound starts with precise documentation. Most suppliers list 1,1,3,3-tetrachloroacetone under its CAS number (918-00-3), and bottles almost always include purity markings—commonly not less than 97%. Detailed labeling cites the molecular formula (C3H2Cl4O), gross weight, and best storage conditions, usually cool and shielded from light. Based on regulatory guidelines, labels display hazard pictograms for acute toxicity, skin and eye irritation, and environmental hazard. Anyone stocking this chemical checks these specs twice, since outdated or poorly labeled bottles can quickly attract regulatory trouble or cause confusion in crowded storage cabinets.
Preparation Method
Manufacturing 1,1,3,3-tetrachloroacetone runs beyond bench-top chemistry. The most common route involves chlorination of acetone with chlorine gas, favoring radical conditions under UV light. Scale-up facilities carefully meter chlorine addition and often push the reaction through a series of traps and condensers to maximize yield while capturing any vented gases. Every run brings its own quirks, and process chemists continually refine purification stages to strip out side-products or over-chlorinated messes. Laboratory-scale preparations still rely on classic protocols, but strict monitoring is in place to keep exposure and by-products under control.
Chemical Reactions & Modifications
If there’s a versatile aspect to tetrachloroacetone, it’s the way those chlorine atoms invite other reagents to the party. This compound works as a potent electrophile; nucleophiles such as amines or thiols replace chlorines, generating substituted products on demand. In cyclization reactions, it provides activated methylene carbon nestled between chlorinated groups, paving way for ring formation and complex frameworks. Professional chemists often leverage its ability to undergo condensation with enolizable compounds, expanding the palette for heterocycle construction. Its shelf life in a busy synthetic lab may be short—keen researchers find new modifications and push the chemistry every year.
Synonyms & Product Names
In catalogs and research articles, 1,1,3,3-tetrachloroacetone appears under alternative names—tetrachloroacetone, 3,3,3,1-tetrachloro-2-propanone, and even as symmetrical tetrachloroacetone. European suppliers sometimes refer to it in German or French translations, but the CAS number links it across all languages. Despite these names, seasoned researchers check structural formulas, not just labels, since isomers and related molecules carry similar names that lead to confusion.
Safety & Operational Standards
Direct exposure to 1,1,3,3-tetrachloroacetone can irritate the skin, eyes, and respiratory tract almost instantly. Standard operating procedures always include gloves, eye protection, and strict fume hood use. Experienced lab managers train staff to recognize the characteristic odor as a sign of leaks, not just inconvenience. Spill response kits in facilities with tetrachloroacetone stock up on absorbents and neutralizers designed for aggressive halogenated solvents. Safety data sheets urge proper segregation from bases, strong acids, and reactive metals, heading off exothermic reactions and possible fume release. Waste handling plays a key role: disposal happens through permitted hazardous waste firms, not down the drain or in ordinary trash. Across industries, regulators keep a close watch on inventory, so documentation and secure storage remain routine.
Application Area
Organic synthesis drives the demand for 1,1,3,3-tetrachloroacetone. Researchers value this compound for launching domino reactions that create rings and chains in medicinal chemistry, crop protection, and material science. Its selective reactivity provides an entry point to introduce halogens precisely, following paths unavailable with simpler acetone derivatives. Contract research organizations and custom synthesis labs keep this compound in stock for heavy-duty transformations, guiding industrial partners through exploratory work and scale-up strategies. Pharmaceutical candidates, specialty agrochemicals, and tailored organic materials draw from chemistry set in motion by tetrachloroacetone’s unique structure.
Research & Development
Innovation does not slow in this segment of chemical research. Development teams in universities and industry push to reinvent classic reactions that use tetrachloroacetone, mindful of regulatory limits and safety pitfalls. New work documents catalysts for milder reaction conditions and greener solvents to replace traditional, more polluting approaches. Research groups publish about selective dechlorination, cyclization, and asymmetric syntheses that rely on the compound. Many studies spring from practical problems: a new molecule fails unless modified with targeted halogenation, or an existing synthetic route requests improved yields using established reagents. The ongoing push to measure toxicological effects and develop better detection techniques, including portable sensors, expands the toolkit for chemists and safety engineers alike.
Toxicity Research
Toxicological studies shine a light on hazards posed by halogenated ketones. Research indicates that exposure to tetrachloroacetone can quickly trigger mucous membrane irritation, coughing, and headaches. Animal trials, historically more common than today, mapped the compound’s acute oral and dermal toxicity, showing dose-dependent effects that guide current workplace exposure limits. Newer in vitro assays fill knowledge gaps about chronic and sub-acute effects, especially on liver and kidney tissues. Detection in groundwater and workplace air gets addressed through sensitive, field-ready analytical methods. Regulatory bodies base recommended exposure limits on published data, and responsible chemical handlers read these reports before opening a new drum or scheduling maintenance in busy facilities.
Future Prospects
The outlook for 1,1,3,3-tetrachloroacetone depends on the shifting landscape of green chemistry and regulatory demands. In fields where every atom counts—like pharmaceuticals and advanced materials—chemists continue to turn to unique reagents for rapid, precise transformations. Researchers work on more sustainable routes to produce and consume halogenated molecules, including tetrachloroacetone, with less impact on air and water. The drive to phase out persistent toxins lifts safer handling practices and rigid waste controls onto the front burner. As synthetic methodologies evolve, and with continued investment in better exposure controls and detection, tetrachloroacetone looks set to retain its place in the chemist’s toolbox, but only as science keeps risks in check and keeps the door open to newer, cleaner alternatives.
What is 1,1,3,3-Tetrachloroacetone used for?
Diving Into 1,1,3,3-Tetrachloroacetone
Walking through the corridors of a chemistry lab, you’ll find shelves lined with chemicals that barely cross the mind of someone outside the field. 1,1,3,3-Tetrachloroacetone doesn’t roll off the tongue. It’s not a household name either. Yet, for those who spend their days in research and development, this compound has a well-marked spot in their work.
Why Chemists Turn to 1,1,3,3-Tetrachloroacetone
What grabs attention about this compound is its unique structure. Packing four chlorine atoms around a three-carbon skeleton, it brings hefty reactivity to the table. Synthesizing new molecules, especially those destined for pharmaceuticals or agrochemicals, often needs strong building blocks that trigger reactions with precision. 1,1,3,3-Tetrachloroacetone delivers that spark.
In organic synthesis, chemists reach for this compound as a strong halogenated acetone. It’s a tool for building carbon-carbon or carbon-heteroatom bonds, sometimes introducing chlorine atoms in places that would otherwise resist chemical change. As a starting point or a waystation toward complicated molecular targets, its uses feel technical, but its impact touches much in daily life—from medication ingredients to crop protection chemicals.
Industry Experience Brings Out Its Practical Value
My own lab experience showed how grueling experiments can become when searching for a way to drop a chlorine atom right where it counts on a molecule. Regular acetone just won’t cut it. Relying on 1,1,3,3-tetrachloroacetone, we isolated what would have been tangled product mixtures or slow reactions with other reagents. It’s a specialty material, but the right specialty compounds make or break advanced research.
Beyond research, industrial chemists scale up these reactions to produce seed compounds for dyes, fungicides, and other specialized organic products. The demand doesn’t feel massive until a company decides to commercialize a new process or fine-tune a product’s performance. Meeting environmental and safety standards turns into a balancing act, since working with heavily chlorinated molecules means keeping close tabs on storage, disposal, and worker safety.
Safety, Ethics, and the Search for Greener Solutions
Every scientist who signs off on an order form for a halogenated compound knows the risks. Chlorinated chemicals earned a reputation for persistence in the environment. Mismanagement in the last century filled rivers and soil in uncomfortably long-lasting ways. Responsible chemists now track reagents from shelf to waste can, ensuring containment and proper disposal. Even in a laboratory equipped with fume hoods and gloves, a drop leaking from a bottle spells a headache. That’s the trade-off: you get a highly useful molecule, but you commit to high standards too.
To reduce dependence on this type of chemical, companies and universities pour funding and brainpower into greener alternatives. Methods that cut down on hazardous waste, or even replace chlorine chemistry with milder strategies, move slowly but surely. Not every use of 1,1,3,3-tetrachloroacetone will disappear, but the long journey toward safer and more sustainable chemistry nudges everyone to look for substitutes where possible.
The Continuing Relevance in Science and Industry
In a world fascinated by new pharmaceuticals and cutting-edge materials, people rarely see what it takes to create these breakthroughs. 1,1,3,3-Tetrachloroacetone may seem like a footnote, though it sticks around as a workhorse for specific reactions that need a heavy dose of reactivity and precision. Its value reveals a deeper truth: progress in chemistry comes not just from famous discoveries, but also from the reliable stepping stones that push innovation forward, even when they never appear in the headlines.
What are the safety precautions when handling 1,1,3,3-Tetrachloroacetone?
Looking at the Risks of 1,1,3,3-Tetrachloroacetone
1,1,3,3-Tetrachloroacetone isn’t some harmless substance you’d stumble across in a household product. In my lab days, I watched even seasoned researchers treat it with deep respect. The stuff is volatile, hits your skin or eyes hard, and can give your lungs a burning memory you'd rather avoid. Anyone in a research setting knows you don’t casually toss this chemical around. Toxicity reports speak for themselves — inhalation or contact leads to immediate, sometimes long-lasting, health problems.
Protecting Yourself: Hands, Eyes, Lungs
Direct contact burns. You wouldn’t handle this one bare-handed, ever. Nitrile gloves, double-layer if they aren’t certified for chemical splash, offer a barrier. A single splash can land you in the occupational health office, or worse. Even robust latex won’t do — chlorine-rich chemicals slip right through. I made a habit of checking for tears in gloves before every use.
It’s tempting to skip eye protection, thinking a face shield is overkill. I knew a colleague who splashed a chlorinated solvent under his goggles, and it took weeks to heal. With tetrachloroacetone, safety goggles alone don’t cut it. Face shields create an extra layer between you and mistake territory. Rubbing your eyes afterwards only spreads the misery.
Breathing the fumes feels brutal. The vapor is heavier than air, so it settles low. Working outside of a chemical fume hood exposes your lungs and the lungs of everyone else around. I always checked the airflow with a bit of tissue paper and made sure nothing was blocking the sash.
Containment: Avoiding Spreads and Spills
Spills don’t just stay in one spot. Tetrachloroacetone evaporates fast, and the fumes expand before anyone notices what’s happening. Catching a whiff means you’ve already been exposed. Only chemically resistant containers should hold this stuff — old glassware or milk jugs don't cut it. Labels should stay clear and unmistakable, with hazard signs that nobody can miss.
In cleaning up spills, ordinary paper towels distribute toxins in the air. I used absorbent pads approved for organochlorine cleanup, and followed up with proper hazardous waste disposal. Nobody wants to walk into a storeroom reeking of chemicals days later because a half-done job left traces behind.
The Value of Team Safety Culture
Trust in your coworkers matters more when chemicals reach this danger level. I learned quickly that corners aren’t cut. Once, someone left a cap loose over a weekend and we returned to a stinging smell and a ruined batch of samples. The experience hammered home why double-checks and team signoffs protect not just health, but the whole workplace.
Having an eyewash station and shower close by makes a real difference. I tested mine regularly, making sure nothing blocked access. You hope never to use them, but small delays can turn a treatable splash into a permanent injury.
Responsible Storage and Training
Every bottle needs a date and owner, and every handler needs the right training. MSDS sheets can’t collect dust in a drawer; a quick scan before each use refreshes your memory on what’s at stake. Waste goes in labeled, sealed containers, sent for proper disposal. Any shortcut can endanger others—something you never forget after your first real scare.
Experience says you can’t predict every outcome with toxic chemicals, but you can stack the odds in your favor. Caution isn’t paranoia. With 1,1,3,3-Tetrachloroacetone, it’s common sense backed by hard lessons.
What is the chemical formula and structure of 1,1,3,3-Tetrachloroacetone?
Examining the Chemistry Up Close
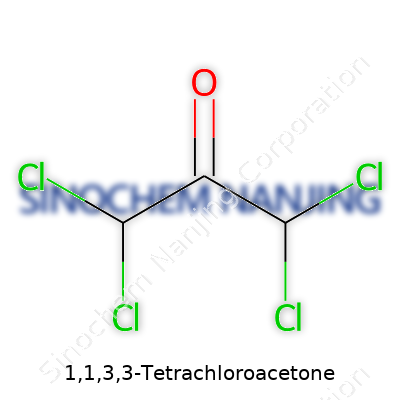
Looking at 1,1,3,3-Tetrachloroacetone, the name alone reveals much about its makeup. You start to see patterns in chemical nomenclature after spending enough time with organic compounds. The "tetrachloro" part means this molecule has four chlorine atoms. "Acetone" marks the basic three-carbon skeleton, centered around a ketone functional group. Put it all together, and the chemical formula becomes C3H2Cl4O.
What's interesting here is how those four chlorines position themselves. The official structure shows two chlorines attached to carbon 1 and another two to carbon 3, leaving the central carbon (carbon 2) paired with the oxygen in a classic ketone double bond. If you draw it on paper, you’ll see a three-carbon chain (CH2-CO-CH2 in acetone), but swap out the hydrogens at positions 1 and 3 with chlorines. So, structurally, this goes:
Cl2CH–CO–CHCl2
Why Structure Matters Beyond the Lab
In practice, details about molecular setup play a key role in both safety and usability. Chlorinated compounds, especially polyhalogenated ketones like this one, often show a mix of high reactivity and significant toxicity. Those extra chlorines make this molecule less flammable than plain acetone. I’ve seen rare ketones like this used in specialized organic syntheses, where their properties let them act as intermediates or building blocks you can’t get any other way.
The flip side, and it's not something glossed over, involves health and environmental risk. Chlorine atoms help the compound stick around in the environment and resist breakdown. You don’t want this stuff getting loose in waterways, and you’ll want good ventilation, gloves, and goggles up in the lab. Hard facts from chemical safety sheets back this up, and so does experience — even a small spill means careful cleanup.
Digging Deeper: Potential and Problems
Lots of efforts in green chemistry aim to avoid or minimize reliance on heavily halogenated chemicals. The persistence and bioaccumulation associated with these compounds have been studied since the 1970s. Policy and research circles keep returning to the idea of replacing or redesigning such chemicals to cut down risk. Sometimes, this means swapping them for less persistent analogues; sometimes, the push is toward entirely new reaction routes.
As a tool, 1,1,3,3-Tetrachloroacetone’s value stays confined to niche reactions. Its usefulness comes up in a handful of places: building more complex molecules or helping researchers understand reaction mechanisms. You won’t see it in household products or commercial manufacturing outside tightly controlled environments.
Moving Ahead With Knowledge
Learning the formula and structure for a molecule like this doesn't just tick off a trivia box. It spotlights the ongoing tension chemists face: balancing innovation with responsibility. Students and professionals alike would benefit from considering not just what a compound can do, but what its existence and usage mean for everyone else. After all, every molecule we make or use leaves a footprint somewhere.
How should 1,1,3,3-Tetrachloroacetone be stored?
Why Storage Choices Matter
Chemical safety isn’t just lab talk; it's about everyday decisions that protect lives and prevent nightmares at work or home. Take 1,1,3,3-Tetrachloroacetone. This stuff carries both opportunity and risk. Industries look to it for specialized synthesis, but one slip can bring on toxic exposure or fire danger. As someone who's handled reactive chemicals in a small university space, I’ve seen how easy it is for one complacent action to create chaos.
Know the Hazards Up Front
This liquid’s volatility is no secret. Volatile organics with multiple chlorine atoms rarely play nice with heat, open flames, or rough handling. That sharp smell isn’t just unpleasant—it signals danger. Eyes, skin, lungs, the whole body can pay a steep price for careless storage. Regulatory guidelines from organizations like OSHA and NIOSH stress controlling exposure to chlorinated ketones, and with good reason.
What Proper Storage Looks Like
Putting this chemical in a random spot or cramming it on a crowded shelf doesn’t cut it. Start with tightly sealed glass containers, since metal packs a risk for corrosion or unwanted reactions. Polyethylene or other high-grade plastics often struggle with solvents like this; ampoules or amber bottles work better if direct light is around. Heat ramps up volatility, so a cool space matters a lot—I've always reached for ventilated flammable storage cabinets set well away from working areas.
Humidity and moisture spell trouble. 1,1,3,3-Tetrachloroacetone hydrolyzes slowly with water, producing acidic byproducts and degrading the original material. Desiccators or climate-controlled cabinets help maintain dryness, especially in tropical or temperate zones that swing between damp and dry climates. Keep incompatible materials away, including strong bases or reducing agents. I once watched an undergrad ruin an entire batch of chlorinated solvent by letting a small leak drip toward an open bottle of sodium hydroxide—nobody forgot the choking fumes anytime soon.
Access Controls and Labeling—No Shortcuts
Mistakes happen when containers aren’t labeled sharply and access is open to everyone. Bright chemical-resistant labels with clear hazard warnings stay readable, even if spills splatter. Regular audits, handled by assigned staff, catch aging bottles and prevent the kind of confusion that leads to dangerous mix-ups. In my experience, a simple barcode inventory system—just a handheld scanner and a Google Sheet—went a long way toward stopping errors or theft.
Training and Emergency Preparedness
People are just as important as precautions. Proper PPE—think nitrile gloves, goggles, and a snug lab coat—keeps contact minimal. And spills shouldn't spark panic if everyone already knows how to deploy absorbent pads, where the eyewash and shower stand ready, and why quick reporting matters more than blame. Annual drills offer peace of mind that written policies hold up in real-world situations.
Small Steps, Big Difference
Effective storage doesn’t always rely on big-budget solutions. Good habits and savvy planning stop one bad day from turning into disaster. By treating 1,1,3,3-Tetrachloroacetone with the respect it demands, anyone can build a safer space for science, work, or study.
What are the physical and chemical properties of 1,1,3,3-Tetrachloroacetone?
Physical Qualities: What You Get With This Compound
1,1,3,3-Tetrachloroacetone shows up as a yellowish liquid, and the stuff gives off a pretty recognizable, pungent smell. This compound boasts a molecular formula of C3Cl4O and weighs in at around 197.85 grams per mole. The density hovers right around 1.6 g/cm3, so it sinks in water rather than floating.
Even at room temperature, you’ll notice it doesn’t evaporate as easily as water, but vapors build pretty quickly in a closed space. The boiling point clocks in close to 120°C, and this means it won’t just start steaming away on a hot day in the lab, but things change if you put it over direct heat.
Transparency matters, but with this liquid’s strong coloring, you won’t mistake it for anything neutral. Spills will stain, and they stand out immediately — a safety plus in fast-paced work. The compound doesn’t dissolve in water with any ease. Instead, it finds comfort in organic solvents like acetone and chloroform.
Chemical Nature: What It Can Do and Where It Can Go Wrong
By structure, two chlorines attach to each side of the central carbon, putting heavy atoms at the edges and one oxygen tucked right in the middle of the molecule as a carbonyl. Chemists know this arrangement cranks up reactivity. Both sides of the molecule can join up with other molecules, and the carbonyl group sitting in the middle makes the compound pretty eager to react with nucleophiles.
One encounter with a strong base, and you’ll see it break down or even kick off a violent reaction. Acids can also spark problems, usually leading to unwanted byproducts. With so many chlorines, hydrolysis spits out hydrogen chloride gas, which can burn the nose and lungs on contact. The electron-withdrawing effect of the chlorines gives this compound real punch in chemical synthesis, especially if you need a strong halogenated building block.
Set it near an open flame or anything really hot, and all those chlorines raise the odds for toxic fume release. Exposure to heat or incompatible chemicals can cascade into danger—so ventilation and protective gear do more than just tick off a safety list. These risks put it on the radar for careful tracking and restricted use.
Risks and the Impact on Workplace Safety
Tetrachloroacetone doesn’t just make life interesting in synthetic chemistry. It adds hurdles for anyone managing chemical waste or designing a safe workspace. If you get any on your skin or in your eyes, expect irritation, and inhaling its vapors goes rough on sensitive airways.
Back in the lab, prepping for spills and keeping antidotes ready isn’t negotiable. Local exhaust and gloves become the frontline defense. Double-checking the material safety data sheet before opening the bottle will save a lot more trouble than it causes. Accidents with this chemical run expensive—not just for cleanup, but for health, reputation, and even compliance.
Where Solutions Start: Handling and Alternatives
Getting safer means rethinking even old habits. Fume hoods and sealed transfer equipment keep the danger out of the room. Storage in cool, labeled containers, away from anything that likes to react with chlorines, keeps things uneventful. Where it’s possible to swap this compound out for less reactive choices, labs see fewer problems down the road.
Knowing the physical and chemical tricks of 1,1,3,3-tetrachloroacetone makes a big difference between smooth work and disaster. Even under the pressure of deadlines, respect for a molecule like this never gets old. The more transparent the information, the safer the outcome.
| Names | |
| Pronunciation | /ˌwʌn,wʌn,θri,θri-tɛt.rəˌklɔːr.oʊ.əˈsiː.təʊn/ |
| Identifiers | |
| CAS Number | 821-35-4 |
| Beilstein Reference | 1209237 |
| ChEBI | CHEBI:82235 |
| ChEMBL | CHEMBL226679 |
| ChemSpider | 20518 |
| DrugBank | DB08181 |
| ECHA InfoCard | 100.007.927 |
| EC Number | 208-911-2 |
| Gmelin Reference | 8639 |
| KEGG | C19190 |
| MeSH | D014025 |
| PubChem CID | 6575 |
| RTECS number | UC6475000 |
| UNII | E2F049U880 |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID9020220 |
| Properties | |
| Chemical formula | C3Cl4O |
| Molar mass | 215.83 g/mol |
| Appearance | Colorless to slightly yellow liquid |
| Odor | Pungent |
| Density | 1.71 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 0.91 |
| Vapor pressure | 0.6 mmHg (20 °C) |
| Acidity (pKa) | 0.86 |
| Magnetic susceptibility (χ) | -52.4e-6 cm³/mol |
| Refractive index (nD) | 1.5000 |
| Viscosity | 15.2 cP (25°C) |
| Dipole moment | 2.02 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 367.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -92.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -622.4 kJ/mol |
| Pharmacology | |
| ATC code | D06BB05 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS05 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335 |
| Precautionary statements | P210, P261, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Flash point | 79 °C (174 °F; 352 K) |
| Autoignition temperature | 265°C (509°F; 538 K) |
| Lethal dose or concentration | LD50 oral (rat) 689 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 580 mg/kg |
| NIOSH | SN9845000 |
| PEL (Permissible) | PEL: 2 ppm (15 mg/m³) |
| REL (Recommended) | 1 ppm (7 mg/m³) |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Acetone Chloroacetone Hexachloroacetone Trichloroacetone |

