Digging Deeper into 1,1,2-Trichloro-1,2,2-Trifluoroethane: Reflection on Its Journey, Role, and the Road Ahead
Tracing the Substance Through Time
Back in the mid-20th century, chemists chased molecules that could cool our food, clean precision machinery, and power aerosols, all with less risk of catching fire. That’s how 1,1,2-Trichloro-1,2,2-Trifluoroethane came onto the scene. Known among scientists and industry workers as CFC-113, this compound hit its stride during the refrigeration boom and in electronics manufacturing. Early on, it seemed like a golden ticket—no explosive hazards, handy for solvent applications, and reasonably stable under many common conditions. Most folks outside the lab never stumbled on the chemical’s long official name, but they sure reaped the conveniences that came with its spread.
A Look at What 1,1,2-Trichloro-1,2,2-Trifluoroethane Actually Offers
This chemical sits apart due to a sweet spot in its properties. Non-flammable, non-corrosive to many metals, and a relatively low boiling point—these qualities made it a darling in electronics cleaning, refrigeration, and as a carrier fluid for industrial processes. Engineers knew they could count on CFC-113 to do its work without gumming up precision parts or causing spotty evaporation patterns. In my own projects repairing circuit boards, we relied on it to cut away microscopic debris while leaving no traces behind. The clarity and consistency mattered. Anyone who worked in electronics over the past few decades probably remembers the sharp, distinctive scent that hung in the air wherever this chemical got used.
Understanding the Building Blocks
CFC-113 comes as a clear, colorless liquid, boiling at around 47.6°C and carrying a faint sweet smell. Insoluble in water but mixing comfortably with many organic solvents, it handles its job with a mix of volatility and stability. Its density, vapor pressure, and relatively moderate viscosity made it move through fine channels, flow evenly, and evaporate on cue during cleaning or cooling cycles. In hands-on experience dealing with various industrial solvents, CFC-113 offered distinct, predictable behavior across a wide temperature range. Technicians and plant engineers favored it for that reliability, knowing spills or miscalculations wouldn't lead to immediate danger—unlike some rival chlorinated solvents that posed bigger direct risks.
Technical Details and What’s on the Label
Labels and technical sheets for CFC-113 always stressed its chemical makeup and purity levels. Key specs included purity above 99 percent, with tight limits on moisture and acid content—essential for avoiding corrosion in microelectronics. Production batches typically underwent rigorous checks, similar to what I’ve seen in pharmaceutical operations, though here the stakes involved millions of dollars’ worth of delicate circuit boards and hard drives. The synoptic names—Freon 113, CFC-113, or the more tongue-twisting 1,1,2-Trichloro-1,2,2-Trifluoroethane—often appeared together on drums. Anybody in the field quickly learned how to spot them.
The Chemical Craft of Making It
The path to manufacture charted through chlorination and fluorination steps, starting with simpler compounds like tetrachloroethene and feeding them through sequences of reactions. Chemical plants juggled equipment and know-how to build in the right number of chlorine and fluorine atoms. Each batch required careful monitoring, as the byproducts sometimes packed a punch in terms of reactivity or toxicity. I’ve watched plant operators babysit reactors long into the night, knowing a small error could cost them days of clean-up and generate regulatory headaches. Even outside the line, you learn to never underestimate the attention to detail these processes demand.
Where It Shows Up—and How It Hasn’t Disappeared
The main draw for this compound landed in cleaning, cooling, and industrial chemistry labs. Semiconductor fabs deployed it by the drum, not only because it lifted grime but because it didn’t pit metal traces or short-circuit board surfaces. In aerospace, it did double duty in cooling baths for delicate gyroscopes and relays. Solvent extraction processes in pharmaceuticals and pesticide formulation also took advantage of its solvating powers. For folks tackling legacy equipment or vintage electronics, whispers about CFC-113 still pop up in online communities—its reputation for not dissolving insulation or leaving odd residues hasn’t faded, even with newer alternatives on store shelves.
How Chemistry Lets This Molecule Be Modified
The backbone of CFC-113’s chemistry—three chlorines and three fluorines draped onto an ethane frame—means it can sit through many reactions without falling apart. In the lab, researchers poked at it using ultraviolet light or strong oxidizers, watching for ways to nudge it into breaking down or combining with other molecules. Environmental researchers, in particular, focused their attention here, since the molecule’s durability in sunlight let it ride up into the high atmosphere, where it eventually played a part in ozone depletion. Chemical controls and breakdown schemes became practical research topics, as we scrambled to keep both technological gains and environmental responsibilities in balance.
Why Safety Became a Hot Topic
As CFC-113 rolled out worldwide, safety rules tried to keep up. Handling rules focused on keeping exposure low—working in well-ventilated spaces, donning gloves and goggles, treating spills with respect. Chronic exposure posed the risk of dizziness, headaches, and potential liver stress, especially in closed quarters. Regulatory bodies such as OSHA and NIOSH added their guidance, similar to what I’ve seen with other dense, volatile solvents. The stakes for environmental health set the toughest rules of all. Since it survived for years after escaping into the air, its link to ozone breakdown eventually forced manufacturers to dial back production, ramp up containment schemes, and transition to new compounds.
Research, Development, and the Legacy of CFC-113
CFC-113 shaped an entire generation of research by making cleaning and processing more predictable, but its shadow loomed large in atmospheric chemistry textbooks. Laboratories and environmental groups poured resources into understanding its fate in the stratosphere, fueling a wave of findings that led directly to the Montreal Protocol and worldwide policy shifts. Alternative solvents emerged, aiming to ditch the chlorine atoms or to engineer chemicals that broke down faster once released. Yet, taking CFC-113 off the best-tool list left gaps in ease of use and cleaning performance—real challenges for manufacturing sectors used to its unique mix of attributes.
Chasing Safer and Smarter Solutions
The story of CFC-113 touches anybody who’s had to trade off convenience against environmental or health costs. Years ago, I watched a factory retool an entire assembly line to phase out this solvent—an expensive, nerve-wracking project that demanded retraining, new safety drills, and adjustments in product quality. Most new solvents had their own quirks—some came with odder flavors, some left more manufacturing residue, and a few packed hidden health risks. Collaborations between research labs and equipment designers gave us blends and new molecules that chipped away at these drawbacks. Green chemistry principles promised a smarter path: design out hazards, test breakdown products, and keep supply chains honest about what really winds up in the air or water.
Toxicity: A Stubborn Puzzle
Plenty of debate swirled around CFC-113’s direct toxicity in people. Studies in animals showed organs could be stressed at high doses—liver and kidneys in particular. Most workers felt fine with gloves and ventilation, but the uncertainty about long-term, low-level exposure kept policy makers on edge. Some evidence linked chronic workplace exposure to mild nervous system effects, although findings often depended on job conditions. For the millions exposed indirectly through air and water, the far greater worry stayed linked to what the molecule did thousands of feet above our heads—destroying ozone molecules and letting dangerous UV radiation spill through. The debate over its safe use steered the world to act with rare unity.
Where Does the Road Lead for CFC-113?
More than any technical detail, what matters now is how the story of CFC-113 drives change. Companies and labs keep chasing replacements that work as smoothly or as safely, without leaving a heavy contamination tab behind for future generations. In startups and academic chemistry departments, green chemistry isn’t just a buzzword—it’s a rallying cry. New molecules aim to sidestep the sins of the past: breaking down quickly in the environment, posing fewer risks to workers, and delivering the cleaning or cooling punch that high-tech fields crave. Policymakers sharpen their pencils, looking for rules that keep innovation alive without letting another CFC-sized problem slip through.
Final Thoughts on Legacy and Responsibility
1,1,2-Trichloro-1,2,2-Trifluoroethane stands as a touchstone when looking at the tangled thread between chemical discovery, industrial need, and global responsibility. For every benefit it handed workers and industries, it left a challenge for generations to tackle. The scientific community, backed by hard reasoning and honest fieldwork, told the world to act. Today, the lessons ring clear—scrutinize claims, weigh convenience against long-term harm, and invest in smarter chemistry that respects both health and planet. For every new molecule waiting in the wings, that balance stays non-negotiable.
What are the main uses of 1,1,2-Trichloro-1,2,2-Trifluoroethane?
A Chemical With Many Hats
Few chemicals raise as many eyebrows as 1,1,2-Trichloro-1,2,2-Trifluoroethane, better known as CFC-113. Engineers and technicians in manufacturing plants speak about it as both a useful tool and a problem. Decades ago, folks in electronics repair and assembly depended on CFC-113 as a cleaning solvent. I remember working in a neighborhood TV repair shop, watching how easily it stripped away greasy residue from circuit boards. For tasks like this, nothing seemed to beat it.
Main Uses Over the Years
The story of this compound often starts in the electronics and aerospace industries. CFC-113 cleans delicate parts without leaving much behind. Unlike water-based cleaners, it doesn’t cause rust and evaporates quickly. Factories gravitated toward it to clean sensitive mechanical components, including those in aircraft and satellites. Back in the day, airlines scheduled regular deep cleans of gyroscopes and guidance modules with CFC-113 because a dirty sensor meant navigation problems in the air.
CFC-113 proved valuable as a carrier fluid in the production of adhesives or in processes that need stable, non-flammable liquids. Chemists appreciated its low chemical reactivity, a trait that made it a safe choice around unpredictable reactions and flammable materials. Its compatibility with many plastics and metals let technicians work confidently on expensive equipment.
During the early computer age, companies relied on CFC-113 in the manufacture and maintenance of everything from early circuit boards to mainframes. Even medical device makers found it useful for cleaning surgical tools and precision parts. Many readers working in production lines during the 1980s probably remember the prominence of this chemical in daily routines. The smell in the air became a cue for a well-run electronics service bench.
The Environmental Cost
Of course, the effective performance came at a price. Scientists in the 1970s raised alarms about the breakdown of chlorofluorocarbons and their impact on the ozone layer. Lab studies and atmospheric data pointed straight at compounds like CFC-113 as strong contributors to ozone depletion, a risk to both public health and crops. I watched colleagues worry as labels on cans changed from “safe for all uses” to warnings about wearing gloves, goggles, and even reconsidering long-term use altogether.
As regulations tightened, substitutes entered the market. HCFCs and other blends replaced CFC-113 in cleaning and degreasing jobs. Several European companies stopped stocking it, and tech schools began teaching students to use alternatives. Refrigeration and air conditioning also saw changes, since early projects had used CFC-113 as a refrigerant. Now, regulations in most developed countries block CFC-113 use outside of highly controlled applications—destruction of existing stockpiles or research remains the main exceptions.
Moving Forward Safely
Sometimes, people ask whether old cleaning and degreasing standards from CFC-113 days can be matched by newer chemicals. Green chemistry keeps advancing, and several hydrofluoroethers and non-fluorinated options clean delicate equipment effectively, without the legacy risks. Companies no longer need to balance efficiency against atmospheric harm like they once did.
For anyone running into CFC-113 in legacy equipment or forgotten stores, responsible handling and disposal matter a great deal. Organizations can work with certified hazardous waste programs and keep staff trained in safer substitutes. Respecting lessons learned from CFC-113’s rise and fall—which include protecting the global commons as much as the product itself—should guide those choices.
Is 1,1,2-Trichloro-1,2,2-Trifluoroethane hazardous to health?
A Chemical from Another Era
Many folks remember 1,1,2-Trichloro-1,2,2-trifluoroethane—often called CFC-113 or Freon 113—from the heyday of air conditioners, old refrigerators, and certain industrial solvents. Used for cooling and as a cleaning agent, this chemical built a legacy that spread across homes, factories, and even airplanes. Only lately, society sees its shadow on our health and environment. As a parent, a homeowner, and someone who grew up watching aerosol cans fly off the shelves, I pay attention when the World Health Organization and EPA talk about risks tied to these substances.
Direct Health Concerns
Breathing in CFC-113, even in moderate amounts, creates real trouble for the body. This chemical does more than irritate the eyes and nose. Studies show dizziness, fatigue, and even loss of coordination after short-term exposure. After reading OSHA reports and published research, I’ve seen documented cases where workplace spills led to headaches and trouble concentrating—all signs that the brain and nerves don’t cope well with these vapors. Touching the liquid can also hurt skin, causing dryness and cracking, sometimes even frostbite. Workers in cleaning and electronics repair know these dangers firsthand.
Long-Term Risk: Not Just a Buzzword
CFC-113 doesn’t just vanish after use. The body must break it down and, often, stores it in fat. Over time, frequent contact builds up problems in the liver and kidneys. Some animal studies even link high doses to irregular heartbeats and changes in organ tissue. I dug through EPA health assessments that warn of unknowns around cancer risk, though official studies find no solid proof either way. Still, the regulator’s stance grew strict—not because every case leads to illness, but because repeated, low-level exposure becomes a wildcard in crowded factories or poorly ventilated garages.
Environmental Fallout Spells Backlash
CFC-113 set off global alarms not only due to direct health risk but also environmental impact. Released into the air, this compound floats into the stratosphere where it destroys ozone, raising the threat of skin cancer and cataracts for millions. The 1987 Montreal Protocol brought countries together to phase out chemicals like this for good reason. My town swapped old fridges, and at work we traded in cleaning solvents after learning about ozone holes and poisoned groundwater.
Why Regulation Matters—And Where We Go Next
Strong rules changed the game. Cleaner substitutes—hydrofluorocarbons and advanced water-based cleaners—now fill the gap in most industries. They hold their own as less toxic options with less environmental bite. At my workplace, employer-provided safety sheets outline best handling practices, and modern ventilation systems keep the air clear. The EPA and OSHA set workplace exposure limits and require proper labeling. These actions come straight from real-life injuries and contaminated towns. Monitoring storage, investing in proper disposal, and demanding cleaner products lead to safer workplaces and healthier neighborhoods.
Taking Health into Our Own Hands
One lesson from the CFC-113 saga: don’t discount “old” chemicals lurking in equipment or forgotten storage rooms. Before reusing or disposing of anything with unfamiliar names, I look up the product fact sheet, wear gloves, and make sure there’s fresh air circulating. Neighborhoods can take action too—community events that collect hazardous waste give families peace of mind. The more attention we pay to what we bring into our homes and workplaces, the less chance that legacy chemicals quietly chip away at our health. Science moves forward, but it’s up to every one of us to put knowledge into practice.
How should 1,1,2-Trichloro-1,2,2-Trifluoroethane be stored safely?
The Handling Reality
Most folks wouldn’t come across 1,1,2-Trichloro-1,2,2-Trifluoroethane unless they’re working with chemicals or legacy refrigeration. I’ve noticed in my own time at a plant that old stocks show up more often than you’d expect, and there’s a reason anyone working with chemicals should care. This compound, often called CFC-113, brings not only environmental baggage but personal risk. Exposure isn’t a theory — it can irritate skin, eyes, and affect your central nervous system with enough contact or inhalation.
Why Storage Choices Matter
Let’s talk safety—storage isn’t just about following a rulebook. Small oversights stack up fast. A canister with a worn cap, a dark corner near a heat source, a careless placement next to acids; things go wrong when you trust luck. I’ve watched a minor leak during winter suddenly grow with a few degrees’ temperature rise, catching the maintenance team off-guard. If CFC-113 finds a spark, the results may be unpredictable, and nobody wants chemical vapor headaches or an environmental investigation.
Smart, Simple Storage Basics
Anyone storing this chemical needs to keep the container sealed. Not just loosely shut — properly tight, clean, and checked often. Choose containers made of materials that don’t react with chlorinated compounds. Original drums or high-grade stainless steel do the trick; copper and certain plastics don’t hold up, and there’s risk of slow chemical attack that can turn a small spill into a big incident.
Temperature matters a lot. This chemical boils below 50°C, and it gives off vapors that can pool low to the ground. I’ve seen old storage sheds hit 40°C in summer — not ideal. Shelving and storage lockers should be cool, ventilated, and away from sunlight or heat sources. Airflow pulls away vapors and prevents buildup. Any area storing CFC-113 should have exhaust fans, ideally set to switch on automatically if sensors pick up vapors.
Fire Risk and Compatibility Counts
1,1,2-Trichloro-1,2,2-Trifluoroethane catches fire only under high energy, but if it’s close to flames or sparks, things change. Always store away from welding or grinding zones. Bleach, alkali metals, and strong bases are a bad mix — never let these share a shelf. I once saw new staff put incompatible chemicals on the same rack; luckily, someone caught it, but not before they had to air out the entire storeroom. Good separation of chemical families isn’t over-cautious, it just keeps everyone out of trouble.
Better Habits, Fewer Headaches
Personal protective gear for workers grabbing or moving the containers — gloves, chemical goggles, and at least a standard respirator if ventilation isn’t perfect — makes a difference. I’d rather spend five minutes suiting up than risk days recovering. Spill kits and extinguishers should be close by, and labels must be clear. Signs that warn about contents and stress protective steps take away guesswork during emergencies, when time shrinks and mistakes grow.
Room for Improvement
Tools help, but training sits at the core of good practice. Every new staff member using storage rooms should run through a chemical safety drill. Review those steps yearly. Many spills I’ve seen could’ve been stopped in seconds with a clear head and a pair of gloves. If CFC-113 is getting phased out or stored until proper disposal, don’t skip the final check before hand-off. Lax habits and wishful thinking have no place around old chemicals — structure keeps people and the planet safe.
What are the environmental impacts of 1,1,2-Trichloro-1,2,2-Trifluoroethane?
Understanding the Problem Up Close
Many folks remember their first encounter with chemical names in high school science—long, hard to pronounce, and rarely ever explained beyond a test. Yet these chemicals often move beyond the classroom. 1,1,2-Trichloro-1,2,2-Trifluoroethane, or CFC-113, found its way into industrial cleaning, refrigeration, and electronics manufacturing. It seemed almost invisible in the day-to-day shuffle, but its mark on the environment runs deep.
People once praised CFC-113 for its effectiveness as a solvent. It cleans electronic circuit boards without leaving residue and helped build the fragile parts inside early computers. But while technology moved fast, the environment could not bounce back nearly as quick. For those living near facilities using this solvent, the smell used to catch your nose on the way to work. It hung in the air and later, tests started picking it up in soil and water.
Hitting the Ozone Layer Where It Hurts
Ask anyone old enough to remember the 1980s, and they’ll likely recall the fear around the hole in the ozone layer. CFC-113 and its cousins drove that panic. Once released, this compound rises into the upper atmosphere. It breaks down slowly, but sunlight eventually triggers it to release chlorine atoms. These free agents chew through ozone molecules. Less ozone overhead means more harmful ultraviolet rays pounding the earth—raising risks of skin cancer and eye problems. Crop yields take a hit, too, as sensitive plants struggle under the brighter UV. Data from NASA in 2019 shows that while ozone depletion trends have slowed, some impacts linger for decades.
Chemicals Don’t Stay Put
The trouble with 1,1,2-Trichloro-1,2,2-Trifluoroethane doesn’t stop in the upper air. Runoff from factories or leaky storage tanks often seeps into groundwater. My region wrestled with this a few decades ago. Families with backyard wells started smelling chemicals in their tap water. Some later faced health scares after long exposure. Even low levels of this solvent can harm fish and smaller organisms that form the backbone of aquatic life. US EPA lists CFC-113 as a contaminant, warning about its persistence in the environment.
The Burden of Old Machines and Old Habits
Many older appliances or military equipment still contain this compound. Disposing of them means more of it heading back into the air unless handled with care. Even as new laws phase out production, the legacy of earlier use sticks around like a stubborn stain. Replacing and recycling old systems safely doesn’t just need regulation—it needs skilled workers and public education. I’ve watched community groups in my area gather old fridges for “green disposal” days. That kind of grassroots action makes a real difference, but only if supported by proper funding and government backing.
Pushing Toward Cleaner Tools
Alternatives do exist, and industry can switch to less damaging chemicals and processes. In my own work, I’ve seen electronics manufacturers move away from CFC-113 in favor of water-based or alcohol-based cleaners. Sometimes the transition isn’t simple: new solvents require new machines, and small businesses need help to afford the shift. By providing tax credits and grants, governments encourage that move—saving the ozone layer and keeping our water cleaner.
Communities, scientists, and policymakers all face tough choices to handle pollution from the past. Addressing 1,1,2-Trichloro-1,2,2-Trifluoroethane shows what happens when short-term convenience clashes with long-term harm. If we support a healthy mix of technology, education, and policy, the landscape could look very different for future generations.
Are there any regulations controlling the use of 1,1,2-Trichloro-1,2,2-Trifluoroethane?
The Past and Present of a Chemical’s Journey
1,1,2-Trichloro-1,2,2-trifluoroethane sounds like a handful, but for years people called it by trade names or simply referred to it as a refrigerant or cleaning agent. At one time, it showed up in everything from electronics manufacturing to degreasing solutions. I remember working on an old air-conditioning unit, wiping my hands with a solvent, and later finding out I was handling this exact chemical.
The big story here is not just how this chemical worked in the background but what happened after people figured out its environmental effects. By the 1980s, more research showed that chlorofluorocarbons (CFCs) and their cousins played a giant role in harming the ozone layer. From there, you saw governments and scientists rally to put some boundaries in place.
Where Rules and Regulations Stand
Laws shape up when real risks threaten people or the world. With 1,1,2-Trichloro-1,2,2-trifluoroethane, the landmark change happened with the signing of the Montreal Protocol in 1987. Countries agreed to phase out ozone-depleting chemicals, and this substance landed squarely on that list. The Environmental Protection Agency (EPA) put regulations in place that led manufacturers and importers in the U.S. to cut production and find substitutes. Other countries adopted similar bans or introduced permit systems for any remaining limited use.
Across the European Union, the chemical falls under Regulation (EC) No 1005/2009, which restricts production and import. Companies need deep pockets and solid paperwork to even think about using it for scientific purposes or destruction of existing stockpiles. Most businesses left it behind by necessity.
There are also rules from OSHA and other worker protection agencies. If someone comes across an old drum in a factory basement, exposure standards kick in. Workers must have training and protective gear, and spills must be reported. Even landfill managers follow strict guidelines for disposal—otherwise, local authorities can step in.
Why These Controls Matter
It’s tempting to see chemical regulations as red tape, but real lives hang in the balance. Breathing vapors or coming in touch with skin can lead to headaches, dizziness, or long-term health problems. More than one technician over the years has felt the sting of careless exposure. At the same time, millions depend on a healthy ozone layer to shield them from cancer-causing radiation. Science caught up with these dangers by the late 20th century, and governments listened—something not always guaranteed in a fast-moving industrial world.
Room for Improvement and Safer Paths
People sometimes stumble on old barrels in storage, especially in towns with abandoned factories. Disposal and clean-up can turn expensive fast. Some regions still struggle to monitor all chemical stocks because tracking down each source takes time, effort, and public reporting.
Education helps. Communities gain by knowing what’s left behind and how to alert the right agency. Safe recycling programs and chemical take-back initiatives could give workers and neighbors peace of mind. Lawmakers stay on their toes, updating lists as new synthetic chemicals replace the old. All this takes money and commitment, but for those who grew up before these bans, the payoff spreads out over generations—not just for the technicians or the families living nearby, but for the planet’s ozone shield overhead.
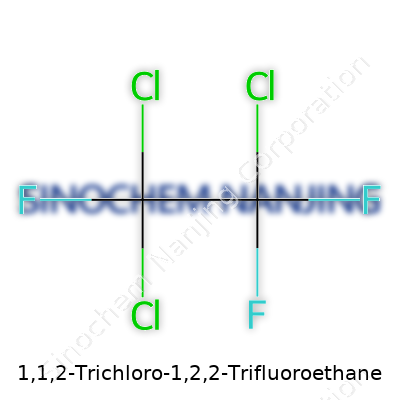
| Names | |
| Preferred IUPAC name | 1,1,2-Trichloro-1,2,2-trifluoroethane |
| Other names |
CFC-113 Freon 113 Halocarbon 113 Refrigerant 113 Arklone Fluorocarbon 113 Genetron 113 Aerosol OT-113 |
| Pronunciation | /ˈwʌn wʌn ˈtuː traɪˈklɔːrəʊ ˈwʌn ˈtuː tuː traɪˈflʊərəʊˌiːˈθeɪn/ |
| Identifiers | |
| CAS Number | 76-13-1 |
| 3D model (JSmol) | `CC(F)(Cl)C(Cl)(F)Cl` |
| Beilstein Reference | 1852296 |
| ChEBI | CHEBI:39147 |
| ChEMBL | CHEMBL45873 |
| ChemSpider | 7346 |
| DrugBank | DB14048 |
| ECHA InfoCard | 03d3ff91-665d-4eb1-93d5-b2d65e3400be |
| EC Number | 602-013-00-6 |
| Gmelin Reference | 73108 |
| KEGG | C01376 |
| MeSH | D014285 |
| PubChem CID | 6570 |
| RTECS number | KI7075000 |
| UNII | K3WOD53H7A |
| UN number | UN1082 |
| CompTox Dashboard (EPA) | DTXSID9020227 |
| Properties | |
| Chemical formula | C2Cl3F3 |
| Molar mass | 187.38 g/mol |
| Appearance | Colorless liquid |
| Odor | Ethereal; faintly sweet |
| Density | 1.569 g/mL at 25 °C |
| Solubility in water | 14 mg/L (20 °C) |
| log P | 2.12 |
| Vapor pressure | 79.9 mmHg (20°C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | pKb = 15.12 |
| Magnetic susceptibility (χ) | -9.54×10⁻⁶ |
| Refractive index (nD) | 1.367 |
| Viscosity | 0.813 mPa·s (20 °C) |
| Dipole moment | 2.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 312.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −993.3 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -1682.6 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N01AB03 |
| Hazards | |
| Main hazards | Harmful if inhaled. Causes skin and eye irritation. May cause drowsiness or dizziness. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS04 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P210, P261, P271, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 1,1,2-Trichloro-1,2,2-Trifluoroethane NFPA 704: "1-1-0 |
| Flash point | 21°C (closed cup) |
| Autoignition temperature | 411°C |
| Explosive limits | Explosive limits: 7.5–15% |
| Lethal dose or concentration | Lethal Concentration (LC50): Inhalation-rat-10,000 ppm/4H |
| LD50 (median dose) | LD50 (median dose): 4,200 mg/kg (rat, oral) |
| NIOSH | NIOSH: KJ3150000 |
| PEL (Permissible) | 1000 ppm |
| REL (Recommended) | 25 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Chloroform 1,2-Dichloro-1,1,2-trifluoroethane Trichloroethylene 1,1,2,2-Tetrachloro-1,2-difluoroethane Chlorotrifluoroethylene |

